Clear Sky Science · en
TNF alpha unmasks enteric malate aspartate shuttle dysfunction bridging Parkinson disease and intestinal inflammation
The Hidden Link Between the Gut and Parkinson’s
Many people with Parkinson’s disease first notice trouble in the bathroom long before their hands begin to tremble. Constipation and bloating can appear years ahead of movement problems, hinting that the gut’s own “second brain” – the enteric nervous system – may be involved early in the disease. This study asks a simple but pressing question: how does inflammation in the intestine disturb these gut nerves, and could that disturbance help drive Parkinson’s and other intestinal disorders?
Gut Nerves in a Dish
To explore this, the researchers built a laboratory model of human gut nerves using induced pluripotent stem cells – adult cells reprogrammed back into a stem-like state and then guided to become enteric neurons and support cells called glia. Some stem cell lines carried three copies of the gene for alpha‑synuclein, a protein that clumps abnormally in Parkinson’s disease, while matching control lines had normal gene dosage. After about ten weeks of guided development, these cultures contained a rich mix of gut-like neurons and glia that closely resembled the cellular diversity found in real human intestines. Crucially, the Parkinson’s-like cells produced extra alpha‑synuclein, creating a controllable model of early disease in the gut.
A Preloaded Vulnerability in Cell Energy
Even before any inflammation was added, cells with extra alpha‑synuclein showed widespread signs of strain in their energy systems. Detailed single-cell gene profiling revealed disruptions in pathways that handle fats, cholesterol, and key building blocks for cell fuel. Microscopy showed that their mitochondria – the tiny power plants inside cells – were fewer, smaller, and more fragmented. Communication between neurons and glia was also rewired, with stress-related signaling pathways becoming more prominent. Together, these findings painted a picture of gut nerve cells that look outwardly similar to healthy cells but are already living close to the edge in terms of energy and resilience.
Inflammation Tips the Balance
The team then introduced tumor necrosis factor alpha (TNF), a major inflammatory signal that is elevated both in Parkinson’s disease and in intestinal disorders such as ulcerative colitis. TNF had a strikingly different impact on the two kinds of cultures. In the Parkinson’s-like cells, TNF drove up alpha‑synuclein levels inside both neurons and glia and increased the protein’s physical contact with mitochondria, a combination associated with damage and oxidative stress. Electrical recordings showed that healthy control networks could ramp up their activity after TNF, but the Parkinson’s-like networks remained sluggish, revealing an impaired ability to adapt to inflammatory challenge. At the molecular level, protein and metabolite measurements converged on a key energy-transfer system called the malate–aspartate shuttle, which normally ferries reducing power into mitochondria. Under TNF, this shuttle faltered specifically in the Parkinson’s-like cells, depleting essential molecules and forcing the cells to fall back on burning glutamine as an emergency fuel.
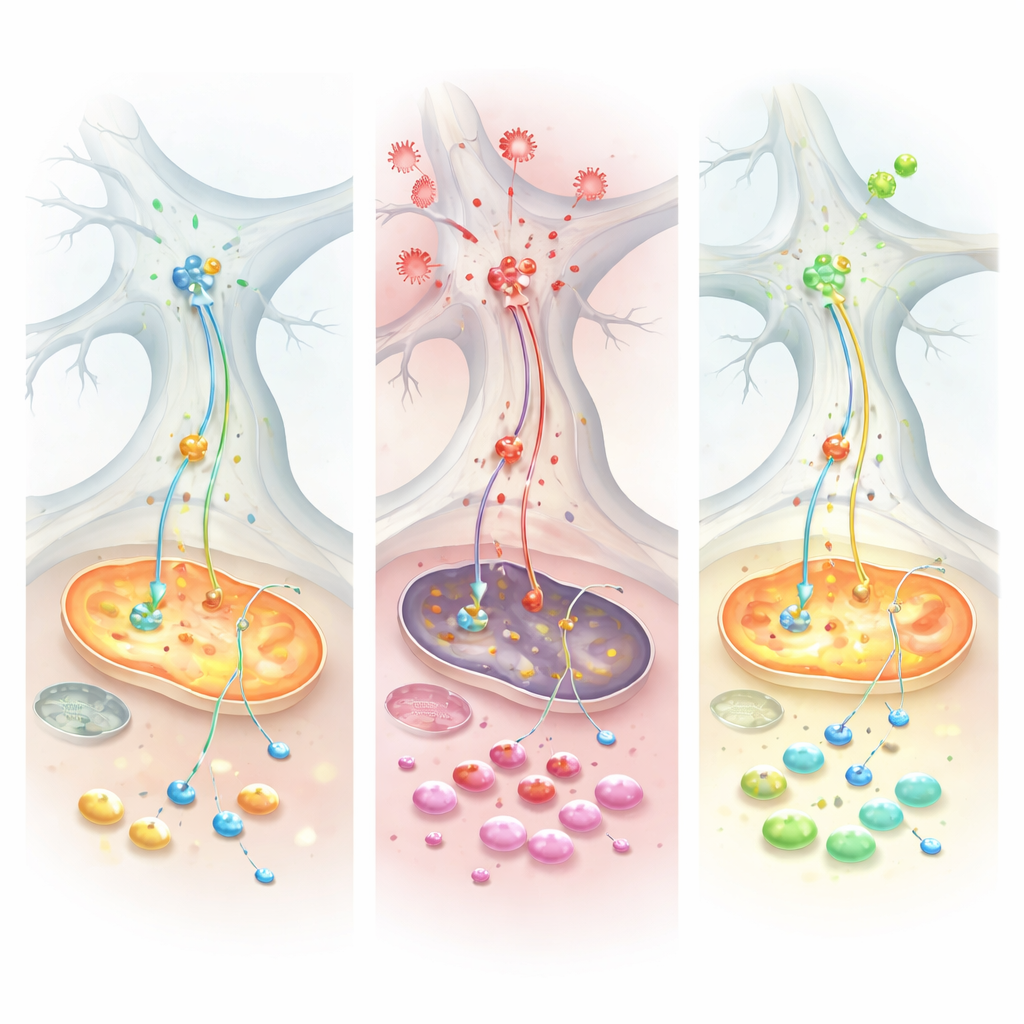
A Potential Rescue and Clues from Patients
When the researchers treated the cultures with Chicago‑Sky‑Blue 6B, a compound that modulates glutamate handling, the picture changed. The drug restored several measures of mitochondrial breathing, reduced the abnormal dependence on glutamine, and partially normalized the energy balance in the stressed cells. To test whether the same metabolic theme appears in real people, the team analyzed gut tissue from patients with ulcerative colitis and large public gene-expression datasets. Across multiple cohorts, they found reduced activity of enzymes that power the malate–aspartate shuttle, alongside increased alpha‑synuclein in inflamed gut tissue. Patients who responded well to anti‑TNF therapy tended to recover expression of these energy enzymes and showed lower alpha‑synuclein levels, suggesting that the inflammatory-metabolic pattern seen in the lab cultures is mirrored in human disease.
Why This Matters for Patients
Put simply, the study suggests that alpha‑synuclein overload makes gut nerves metabolically fragile, and intestinal inflammation, through signals like TNF, exploits this weakness. By crippling a key energy shuttle and pushing cells into a strained backup fuel mode, inflammation increases oxidative stress, encourages alpha‑synuclein to associate with mitochondria, and undermines the normal function of gut neurons. Because similar signatures are seen in both Parkinson’s models and inflamed human intestines, the work points to a shared metabolic weak spot that could be targeted by future therapies. Modulating glutamate and glutamine metabolism – as Chicago‑Sky‑Blue 6B does in this study – emerges as a promising strategy to protect the gut’s nervous system, potentially easing symptoms such as constipation and perhaps slowing the earliest stages of Parkinson’s and other inflammatory gut conditions.
Citation: Ghirotto, B., Gonçalves, L.E., Ruder, V. et al. TNF alpha unmasks enteric malate aspartate shuttle dysfunction bridging Parkinson disease and intestinal inflammation. Nat Commun 17, 3217 (2026). https://doi.org/10.1038/s41467-026-71317-y
Keywords: Parkinson’s disease, enteric nervous system, intestinal inflammation, mitochondrial metabolism, alpha-synuclein