Clear Sky Science · en
Sex-specific KDM6A-HNF4A-CREBH network controls lipoprotein cholesterol metabolism and atherosclerosis via epigenetic reprograming of hepatocytes
Why this research matters for heart health
Heart attacks and strokes often begin with slow changes in how the liver handles cholesterol. Men and women are known to differ in their cholesterol levels and risk of clogged arteries, but the underlying reasons are still being uncovered. This study reveals a sex-linked control system in liver cells that helps keep blood fats in a healthier range, especially in females. Understanding this system may point the way to more personalized treatments for high cholesterol and atherosclerosis.
A liver switch on the X chromosome
At the center of this work is a protein called KDM6A, made from a gene on the X chromosome. KDM6A does not carry cholesterol itself; instead, it acts in the cell nucleus as part of the machinery that decides which genes are turned on or off. Because it sits on the X chromosome and can escape the usual inactivation of one X in females, its activity can differ between females and males. The authors asked whether this difference helps explain why the two sexes handle cholesterol and lipoproteins—fat-carrying particles in the blood—in different ways.
What happens when the switch is turned down
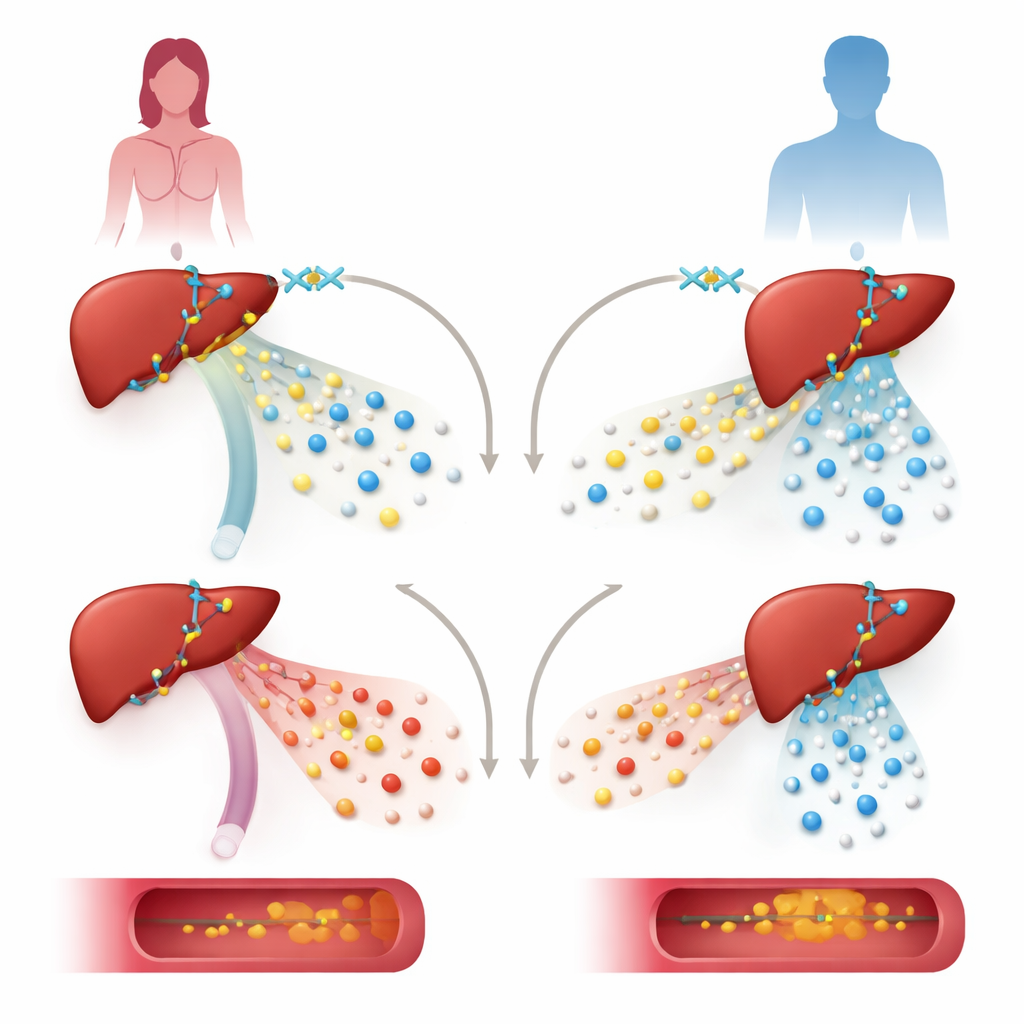
To test KDM6A’s role, the team reduced its levels in human liver cell lines and in mice engineered to lack KDM6A only in hepatocytes, the main liver cells. In liver cells from a female donor, turning down KDM6A strongly lowered the activity of genes that build and process lipoproteins, including genes that support protective high-density lipoprotein (HDL). The same manipulation in male-derived cells affected a different set of genes and did not focus on cholesterol pathways. In female mice, loss of KDM6A in the liver led to a more “atherogenic” lipoprotein pattern: very low-density lipoprotein (VLDL) particles became unusually rich in cholesterol, HDL levels fell, and fatty buildup in the main artery increased, especially when the animals were pushed with a high-fat, high-cholesterol diet and an atherosclerosis-driving genetic tool.
How the liver’s gene-control network is rewired
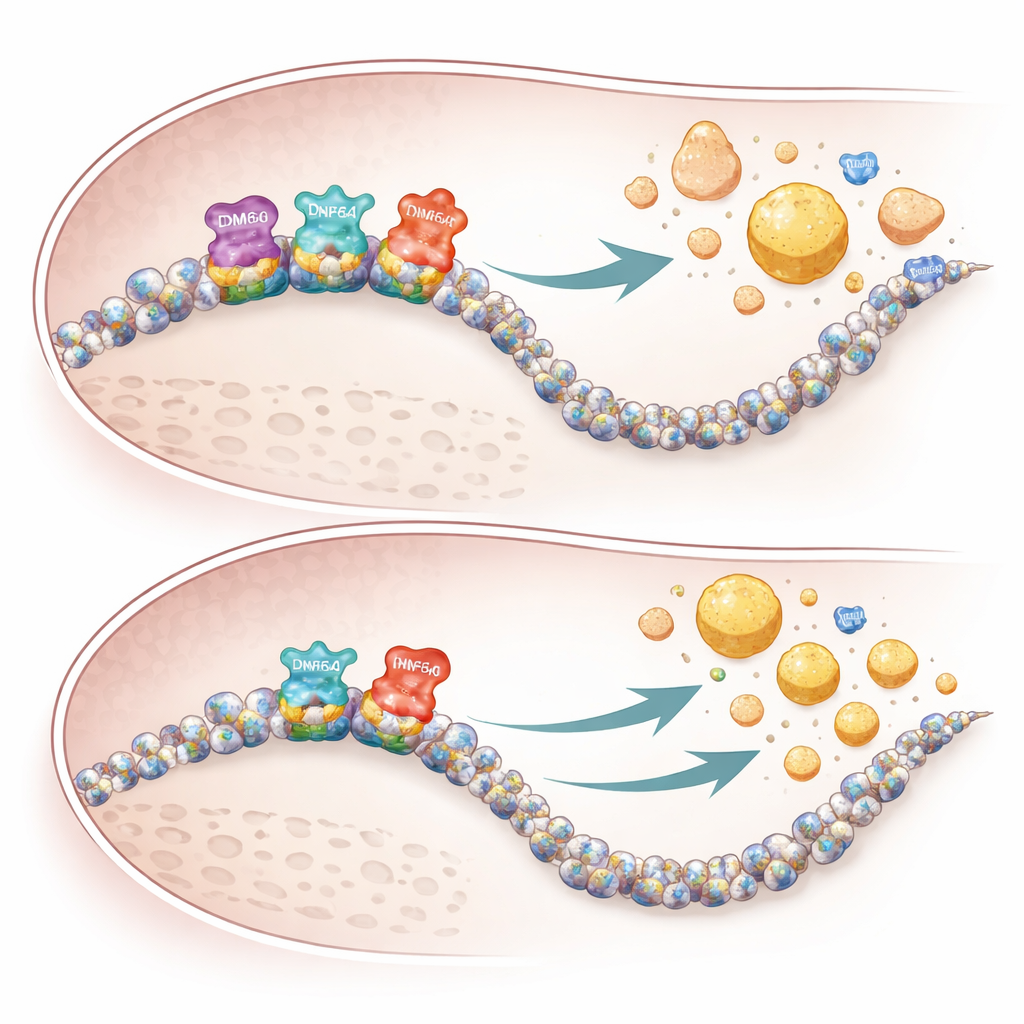
Digging deeper, the researchers mapped how KDM6A sits on the DNA and interacts with other regulators. They found that KDM6A partners with a key liver transcription factor, HNF4A, at control regions near many lipoprotein and cholesterol genes, such as those that make ApoA1, ApoM, and ApoH proteins. Instead of acting mainly through its known chemical “eraser” function on a specific histone mark, KDM6A here behaves more like a scaffold. Together with HNF4A and an enzyme complex called MLL3/4, it helps create open, active chromatin—loosely packed DNA-protein structure—marked by certain chemical flags that favor gene activity. Removing KDM6A weakened these active marks without greatly increasing the repressive marks it usually erases, suggesting a non-enzymatic, structural role.
A third player links the switch to cholesterol output
The study also identifies a third factor, CREBH, a liver stress–responsive protein that directly boosts genes governing triglyceride and cholesterol handling. Using genome-wide binding maps, the authors show that when KDM6A is lost, CREBH binds less strongly to many of its usual target genes involved in bile acid production, cholesterol clearance, and HDL formation. In female mice lacking hepatic KDM6A, these genes were less active, bile acids built up in the liver and blood, and VLDL particles left the liver unusually enriched in cholesterol but relatively poor in triglycerides. This skewed output, combined with reduced HDL-building genes, set the stage for more cholesterol deposition in artery walls.
Why females are more affected
The sex-specific nature of the findings appears to reflect both genetics and backup systems. Because KDM6A is X-linked and more strongly expressed in female tissues, its loss has a larger impact in females. Males carry a related gene on the Y chromosome, called KDM6C, which can partially compensate in liver cells. In male mice, deleting KDM6A in hepatocytes barely changed gene activity or lipoprotein patterns, while similar manipulations in females caused broad reprogramming of cholesterol pathways and striking changes in blood lipids. Human genetic data from large databases echoed these patterns: rare KDM6A variants were associated with lower levels of beneficial ApoA1 and higher levels of atherogenic ApoB in people.
What this means for future treatments
Overall, the work reveals a sex-linked KDM6A–HNF4A–CREBH network that fine-tunes how the liver packages and clears cholesterol. In females, disrupting this network tilts the balance toward more artery-clogging lipoproteins, less protective HDL, and impaired bile acid–driven cholesterol disposal, all of which promote atherosclerosis. By highlighting an epigenetic control hub that differs between sexes, the study suggests that future cholesterol-lowering strategies—and drugs that target chromatin-modifying enzymes—may need to be tailored differently for women and men to avoid unwanted shifts in lipoprotein profiles.
Citation: Chen, L., Kang, Z., Härdfeldt, J. et al. Sex-specific KDM6A-HNF4A-CREBH network controls lipoprotein cholesterol metabolism and atherosclerosis via epigenetic reprograming of hepatocytes. Nat Commun 17, 3945 (2026). https://doi.org/10.1038/s41467-026-70846-w
Keywords: cholesterol metabolism, liver epigenetics, lipoproteins, sex differences, atherosclerosis