Clear Sky Science · en
A group 1 hemagglutinin stem vaccine elicits broad humoral responses against influenza in phase 1/2a study
Why This Flu Study Matters
Each year, influenza sweeps around the globe, forcing scientists to predict which viral strains to target in seasonal vaccines. When their guesses are off, protection drops and people get sick. This study tests a different idea: instead of chasing ever-changing flu strains, build a vaccine that teaches the immune system to recognize a stable part of the virus that rarely mutates. If successful, such a vaccine could move us closer to a "universal" flu shot that protects against many current and future strains, including those with pandemic potential like H5N1 bird flu.
A New Target on the Flu Virus
The influenza virus is studded with a protein called hemagglutinin (HA), which looks a bit like a lollipop: a round head atop a stalk-like stem. Most current vaccines train the immune system to attack the highly variable head. That works well when vaccine and virus match, but fails when the virus drifts or shifts into new forms. In contrast, the stem region is much more stable across many group 1 influenza A viruses, including familiar H1N1 strains and dangerous avian viruses such as H5N1. The researchers engineered a protein vaccine that consists only of a stabilized HA stem trimer from the 2009 H1N1 virus, removing the distracting head to focus immune responses on this conserved stalk.
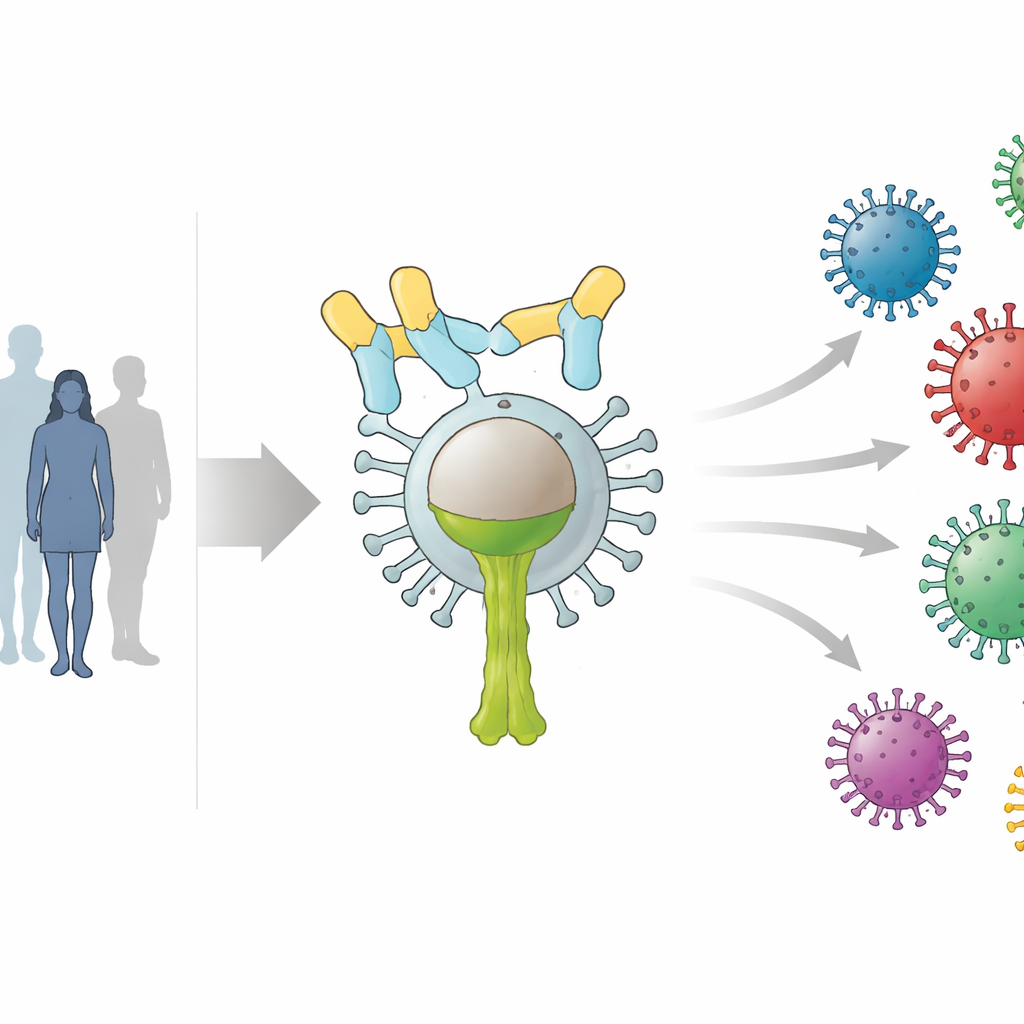
Testing the Vaccine in Volunteers
In this phase 1/2a trial, 170 healthy adults aged 18 to 45 in the United States were randomly assigned to receive one or two doses of the HA stem vaccine at low or high protein amounts, with or without an aluminum-based booster substance, or placebo. The study primarily monitored safety, while also measuring the strength, breadth, and durability of antibody responses in blood. To gauge how this new approach compared with the status quo, the team analyzed samples from a separate group of adults who had received a standard quadrivalent seasonal flu vaccine containing typical head-based antigens.
Broad and Lasting Immune Responses
The HA stem vaccine was well tolerated: most side effects, such as injection-site pain, fatigue, and headache, were mild to moderate and transient, and no serious vaccine-related problems occurred. Importantly, even a single dose triggered strong antibody responses against H5 and the matching H1 stem antigen, with average increases of roughly 6.5 to 16 times over baseline within a month—substantially higher than increases seen after the conventional flu shot. When researchers tested blood against a wide panel of different group 1 flu strains, they saw broad boosting of antibodies, including to viruses with pandemic potential. These responses remained elevated for many months and were still above starting levels one year after vaccination, suggesting durable protection. Adding aluminum adjuvant, using a higher dose, or giving a second shot did not markedly improve performance in this adult population.
From Test Tube to Living Protection
Antibody quantity is only useful if those antibodies can actually block or fight infection. The team therefore assessed whether vaccine-induced antibodies could neutralize H5N1 viruses and recruit immune cells to kill infected cells, a process called antibody-dependent cellular cytotoxicity. Both types of functional activity rose sharply after vaccination and remained relatively stable over time, again outperforming the seasonal vaccine. To model real-world protection, the researchers transferred blood serum from vaccinated volunteers into mice and then exposed the animals to a lethal dose of H5N1 virus that differed from the vaccine strain. Two-thirds of mice given post-vaccination serum survived, compared with none of those given pre-vaccination serum, and higher antibody levels strongly predicted survival.
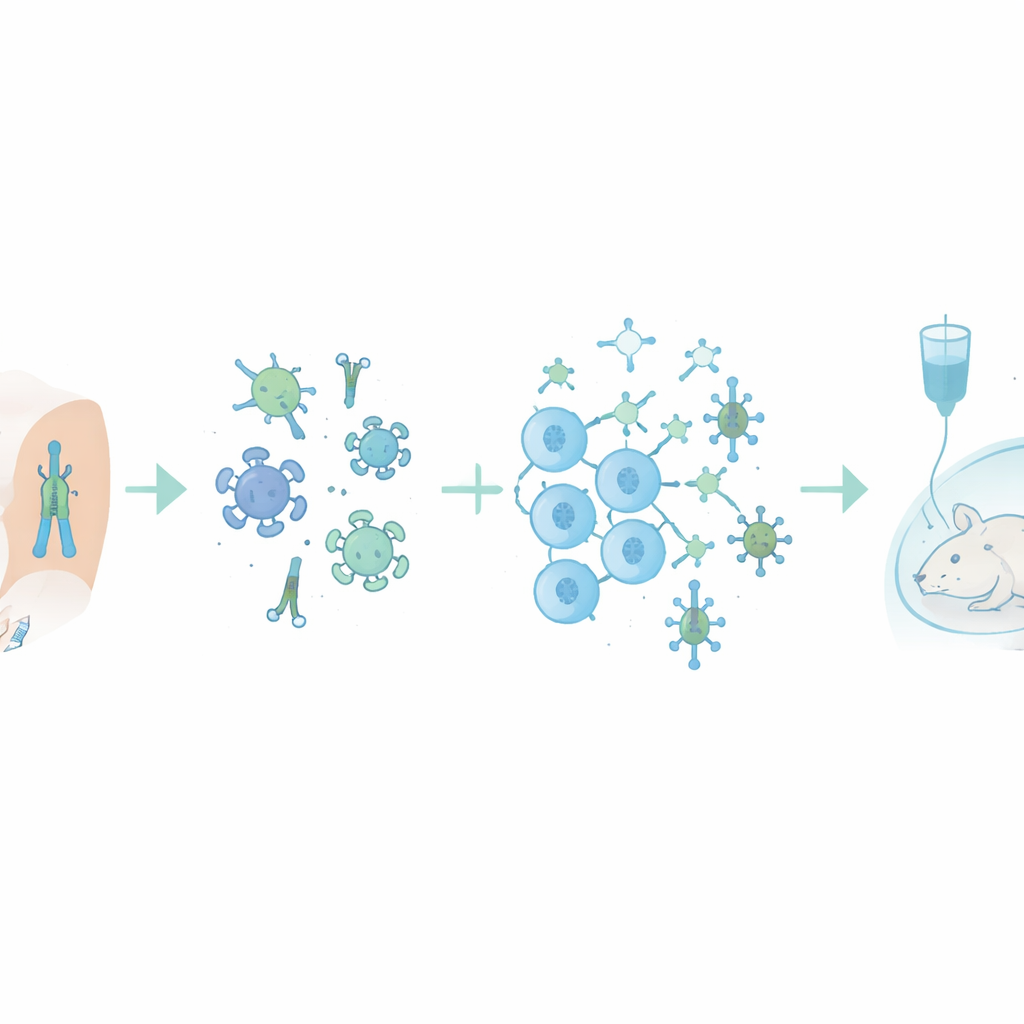
What This Means for Future Flu Shots
This first-in-human study shows that a vaccine aimed solely at the conserved stem of the influenza HA protein can safely trigger strong, broad, and lasting immune responses against many group 1 flu viruses. While it does not yet prove protection in people, it offers compelling evidence that stem-focused immunity can defend animals against deadly, mismatched strains like H5N1. The work supports using such stem-based components as building blocks of a future universal influenza vaccine, which would likely combine group 1, group 2, and influenza B elements. If further trials confirm efficacy, a more universal flu vaccine could reduce annual guessing, improve pandemic preparedness, and lessen the global burden of influenza.
Citation: Hertoghs, N., Tang, C., van Paassen, V. et al. A group 1 hemagglutinin stem vaccine elicits broad humoral responses against influenza in phase 1/2a study. Nat Commun 17, 3451 (2026). https://doi.org/10.1038/s41467-026-70396-1
Keywords: universal influenza vaccine, hemagglutinin stem, H5N1 bird flu, broadly neutralizing antibodies, vaccine clinical trial