Clear Sky Science · en
Ribonuclease 4 Functions in Nociceptor-Mediated Nerve Homeostasis
Why this matters for people living with pain
Pain nerves are often cast as simple messengers that shout when something hurts. This study reveals that some of those pain-sensing neurons, called nociceptors, quietly manage the health of nearby nerve fibers as well. The work focuses on a little-known molecule, ribonuclease 4 (RNase4), and shows that it helps keep pain signals in check while also guiding how damaged nerves break down and rebuild. Understanding this dual role could open new paths for treating chronic pain and improving nerve repair after injury.
A hidden signature inside pain-sensing nerves
The researchers began by mining large single-cell genetic atlases of sensory ganglia—clusters of neurons that relay touch and pain from the body to the spinal cord. They looked for genes switched on specifically in thin, unmyelinated nociceptors, which carry slow, aching pain. Among thousands of candidates, RNase4 stood out as a highly selective marker in mice and showed a matching pattern in human tissue. Microscopy confirmed that RNase4 is found mainly in small-diameter, unmyelinated neurons that belong to the nociceptor lineage, but not in heavily myelinated neurons that handle rapid touch and vibration. This conservation across species suggests that RNase4 marks a specialized subset of pain-sensing cells with a shared biological role.
Shaping nerve behavior from the inside
To probe what RNase4 does, the team engineered mice in which the gene could be deleted either early in development or only in adult nociceptors. When RNase4 was removed during development, the basic wiring of pain circuits and normal heat and touch sensitivity remained largely intact. However, single-nucleus RNA sequencing revealed that many genes changed activity inside nociceptors, including transcription factors, structural proteins, and potassium channel components that help set the electrical firing threshold of neurons. In adult mice, turning off RNase4 only in nociceptors triggered a temporary increase in mechanical pain sensitivity: the animals reacted more strongly to light touch with filaments applied to the paw, while heat sensitivity stayed stable. Over about a month, this heightened mechanical sensitivity faded, hinting that other pathways compensate over time to restore balance.
Sending signals outward to neighboring cells
RNase4 can be secreted outside the cell, raising the possibility that nociceptors influence nearby nerve fibers and support cells. In cell culture, adding RNase4 to a sensory neuron–like line dampened activity in the PI3K–AKT–mTOR pathway, a central growth and survival signaling hub. In living mice, the authors then used a sciatic nerve crush model, which mimics traumatic nerve injury followed by degeneration and regrowth. After injury, RNase4 levels naturally rose across many nociceptor subtypes, especially during the early pain and recovery phases. When RNase4 was absent from nociceptors, behavioral testing showed that mice actually regained mechanical sensation faster than controls, although thermal recovery could lag depending on the deletion strategy. This suggested that RNase4 normally restrains certain aspects of regrowth and sensitivity, and that removing it tilts the system toward quicker tactile recovery.
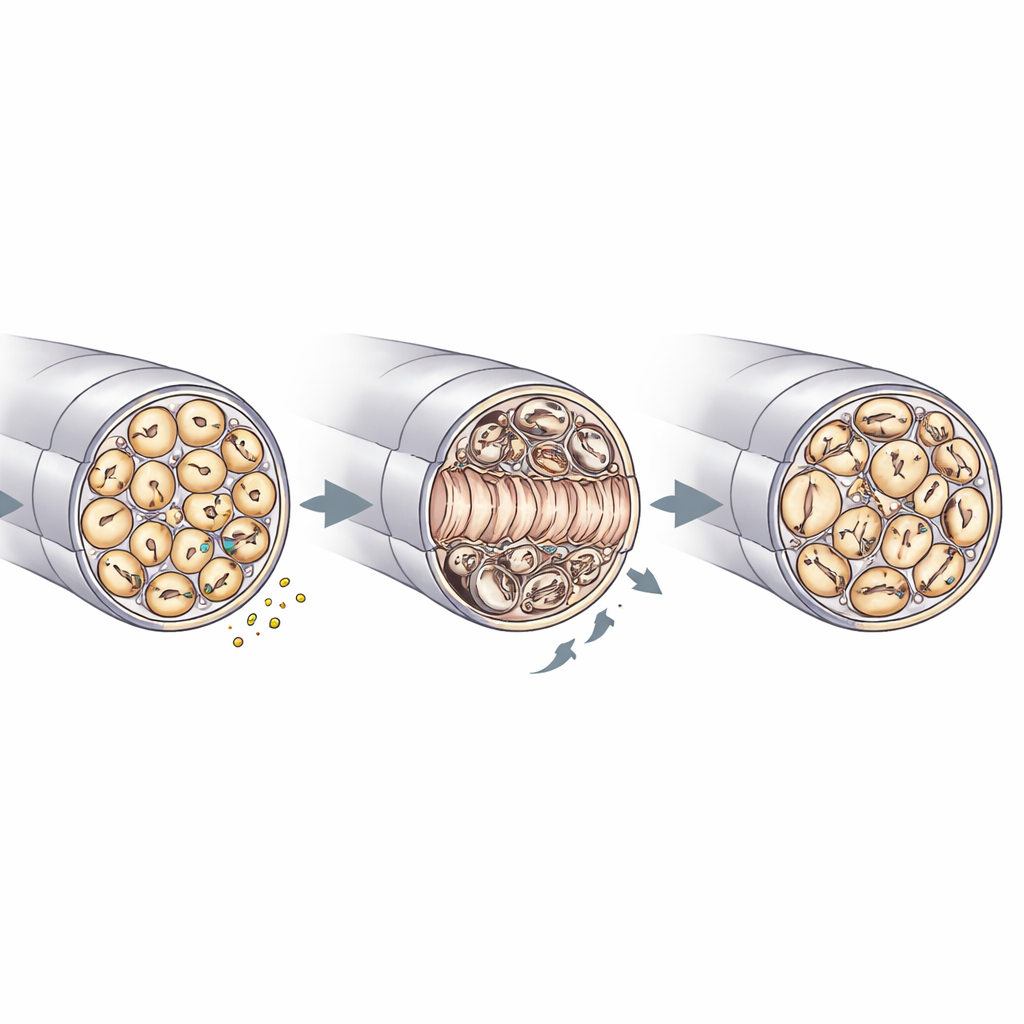
Repair, myelin quality, and nerve housekeeping
Electron microscopy of sciatic nerves offered a closer look at how RNase4 shapes nerve architecture. Before injury, knockout and control nerves looked similar, with comparable axon diameters, densities, and myelin thickness. After crush injury, however, nerves lacking RNase4 contained more regenerated axons in the damaged segment and showed faster re-filling of non-myelinated "Remak" bundles that house many thin fibers. At the same time, these knockout nerves displayed more myelin irregularities—such as folds, infolds, and focal thickening—and signs of stress in Schwann cells, the glial cells that wrap axons. There was also less myelin debris inside immune cells, consistent with more efficient clearance and quicker structural rebuilding. Together with the signaling studies, these findings support a model in which nociceptor-derived RNase4 tunes PI3K–AKT activity and, through non-local effects, coordinates how axons regrow, how myelin is remodeled, and how immune cells participate in cleanup.
What this means for future treatments
In plain terms, the study paints nociceptors not just as alarm bells but as local managers of nerve health. RNase4 acts as a molecular brake and guide: inside nociceptors, it helps maintain their normal firing properties and identity; outside, it limits overzealous regrowth and shapes how myelin and supporting cells respond after injury. Removing RNase4 can speed the return of touch after nerve damage but at the cost of more myelin defects and altered pain sensitivity. Any therapy aimed at blocking or boosting RNase4 will need to balance these trade-offs. Still, the work highlights RNase4 and its signaling network as promising levers for simultaneously modulating pain and promoting more orderly nerve repair.
Citation: Feng, X., Zhang, K., Techameena, P. et al. Ribonuclease 4 Functions in Nociceptor-Mediated Nerve Homeostasis. Nat Commun 17, 2862 (2026). https://doi.org/10.1038/s41467-026-70365-8
Keywords: nociceptors, nerve regeneration, myelin, neuropathic pain, RNase4