Clear Sky Science · en
Targeting notch signaling to restore neural development and behavior in mouse models of ASD
Why this research matters for families and society
Autism spectrum disorder (ASD) affects how people communicate, socialize, and respond to the world around them, yet its biological roots remain puzzling. This study in mice tackles a big question: do very different risk factors for autism—genes, infections during pregnancy, or certain medications—end up disturbing the same brain pathway? The researchers uncover a shared disturbance in a developmental signaling system called Notch and show that carefully dialing this pathway down before birth can restore more normal brain wiring and social behavior in several ASD-like mouse models.
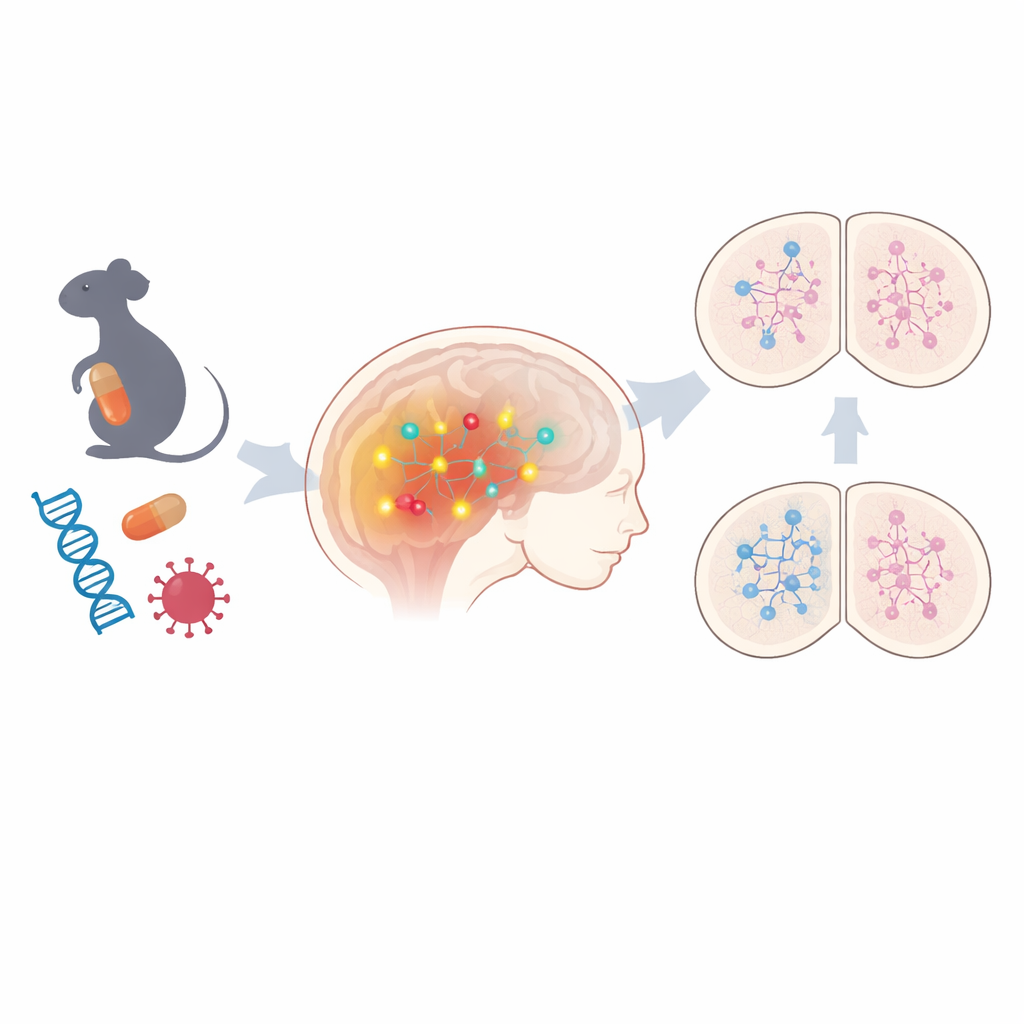
A common pathway behind many different risks
The team began by examining fetal mouse brains from two non-genetic ASD models: one in which pregnant mice were exposed to the anti-seizure drug valproic acid, and another mimicking maternal viral infection. Using broad gene-activity profiling, they found that hundreds of genes were altered in both models, with strong signatures of disrupted epigenetic regulation—chemical marks that turn genes on or off without changing DNA itself. Among the shared changes, one pathway stood out: Notch signaling, a key controller of how immature brain cells decide what type of neuron to become. Components of this pathway, including core receptors and downstream effectors, were consistently turned up. When the scientists looked at human stem-cell based neuron models carrying some autism-linked genetic variants, they again saw heightened Notch-related gene activity in several cases, hinting that this pathway may be a common thread across both environmental and hereditary forms of ASD.
Vulnerable brain cells that fine-tune activity
To understand the consequences of Notch overactivity, the researchers focused on the balance between excitatory and inhibitory signaling in the cortex—often disrupted in autism. They homed in on a small population of inhibitory nerve cells called VIP interneurons, which help regulate how strongly cortical circuits respond to input. In both valproic acid and maternal immune-activation mouse models, these VIP cells were dramatically reduced in the cortex, whereas most other interneuron types were largely spared. Tracing the origins of these cells back to a fetal brain region called the caudal ganglionic eminence, they found that progenitor cells there were proliferating excessively but failing to properly switch into post-mitotic VIP precursors and migrate into the cortex. The pattern suggested that overactive Notch signaling was trapping these progenitors in a “never grow up” state, leading to a lasting shortage of VIP interneurons and a tilt toward excessive excitation.
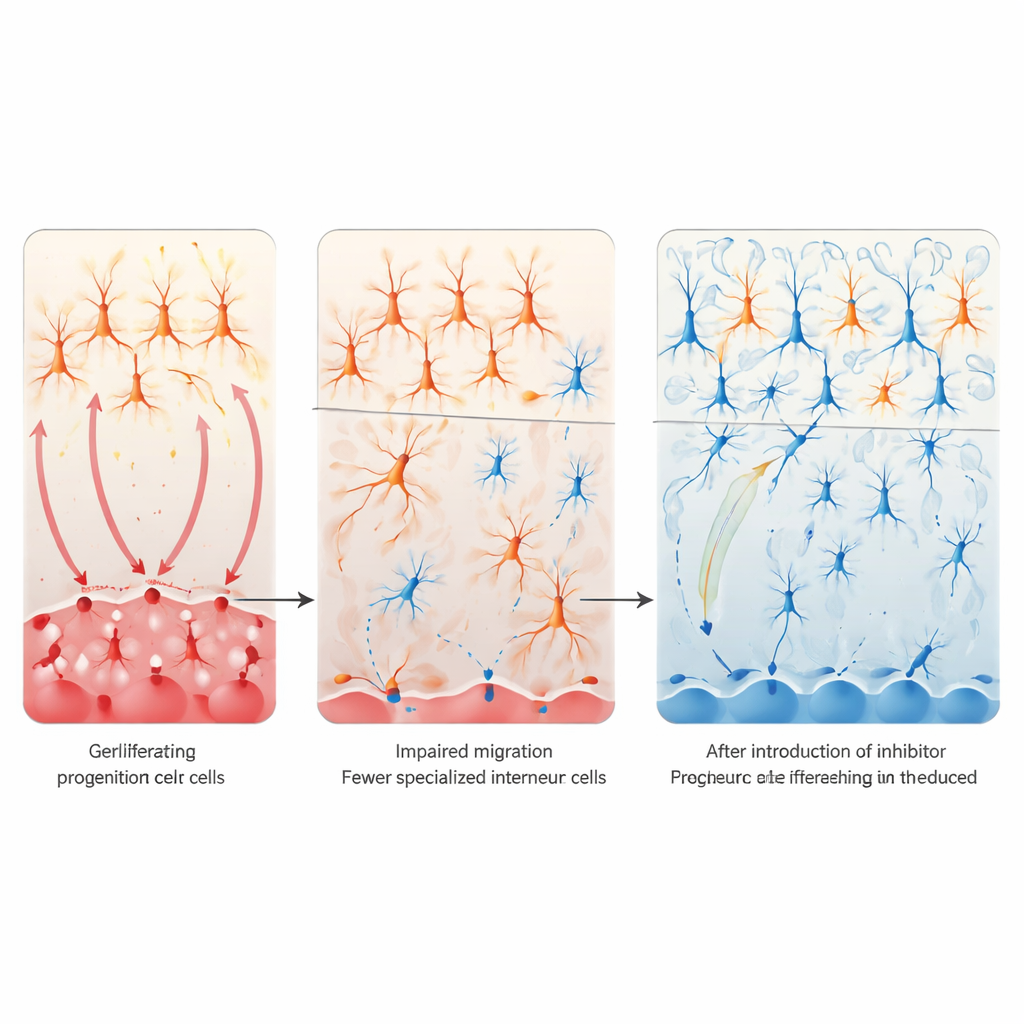
Turning down Notch to rescue brain circuits
The next question was whether calming Notch signaling could rescue these defects. In cell culture, forcing the active form of the Notch receptor into developing forebrain cells reduced markers of VIP interneurons, whereas blocking Notch genetically or with a γ-secretase inhibitor had the opposite effect and could restore VIP markers even after valproic acid exposure. In live mice, the team selectively deleted Notch1 and Notch2 in caudal ganglionic eminence–derived cells. This local genetic “brake” prevented the loss of cortical VIP interneurons and largely normalized social interaction behaviors in offspring exposed to valproic acid during gestation. Transcriptomic analysis of adult brains showed that this rescue also dampened an abnormal upregulation of genes linked to excitatory glutamate synapses, suggesting that restoring inhibitory VIP circuits helps re-balance broader cortical networks.
A one-time prenatal drug that reshapes development
Because genetic interventions are not practical in humans, the researchers tested a drug-based strategy. They used Ro4929097, a potent γ-secretase inhibitor previously evaluated in cancer and Alzheimer’s trials, and administered a single dose to pregnant mice alongside valproic acid. This brief prenatal treatment reduced active Notch protein levels, largely restored VIP interneuron numbers in the offspring’s cortex, and improved multiple ASD-like behaviors, including repetitive grooming and social deficits. Single-cell RNA sequencing of newborn brains revealed that the drug did not only normalize the VIP lineage; it also corrected broader disruptions in neural progenitors, oligodendrocytes (myelin-forming cells), and other cell populations, nudging the overall cellular composition back toward that seen in control animals.
What this means for understanding and treating autism
To a layperson, the take-home message is that diverse autism-related influences may converge on a shared “switchboard” in the developing brain. When the Notch pathway is stuck in the “on” position during a critical fetal window, key inhibitory nerve cells fail to mature, and cortical circuits become imbalanced, contributing to social and behavioral problems. In mice, carefully timed dampening of this pathway—either in specific cell types or with a single prenatal drug dose—can largely restore normal development and behavior. While translating such an approach to humans will require great caution, these findings highlight Notch signaling, and especially the development of VIP interneurons, as promising targets for early-life interventions aimed at preventing at least some forms of ASD-related brain dysfunction.
Citation: Hanno, Y., Nakanishi, M., Takase, A. et al. Targeting notch signaling to restore neural development and behavior in mouse models of ASD. Nat Commun 17, 2587 (2026). https://doi.org/10.1038/s41467-026-70321-6
Keywords: autism spectrum disorder, Notch signaling, VIP interneurons, neurodevelopment, epigenetics