Clear Sky Science · en
TIGAR regulates intestinal mucus barrier integrity by inhibiting lactylation of G6PD/6PGD in ulcerative colitis
Why the gut’s slimy shield matters
The lining of our intestines is covered by a slippery mucus layer that keeps trillions of microbes at arm’s length. In ulcerative colitis, a chronic disease that inflames the colon, this protective coat often thins or breaks, letting bacteria and irritants reach the wall of the gut. This study explores why that mucus barrier fails, tracing the story from sugar metabolism inside specialized mucus‑secreting cells all the way to damage in the intestine—and highlighting a molecule called TIGAR as a promising new target for therapy.
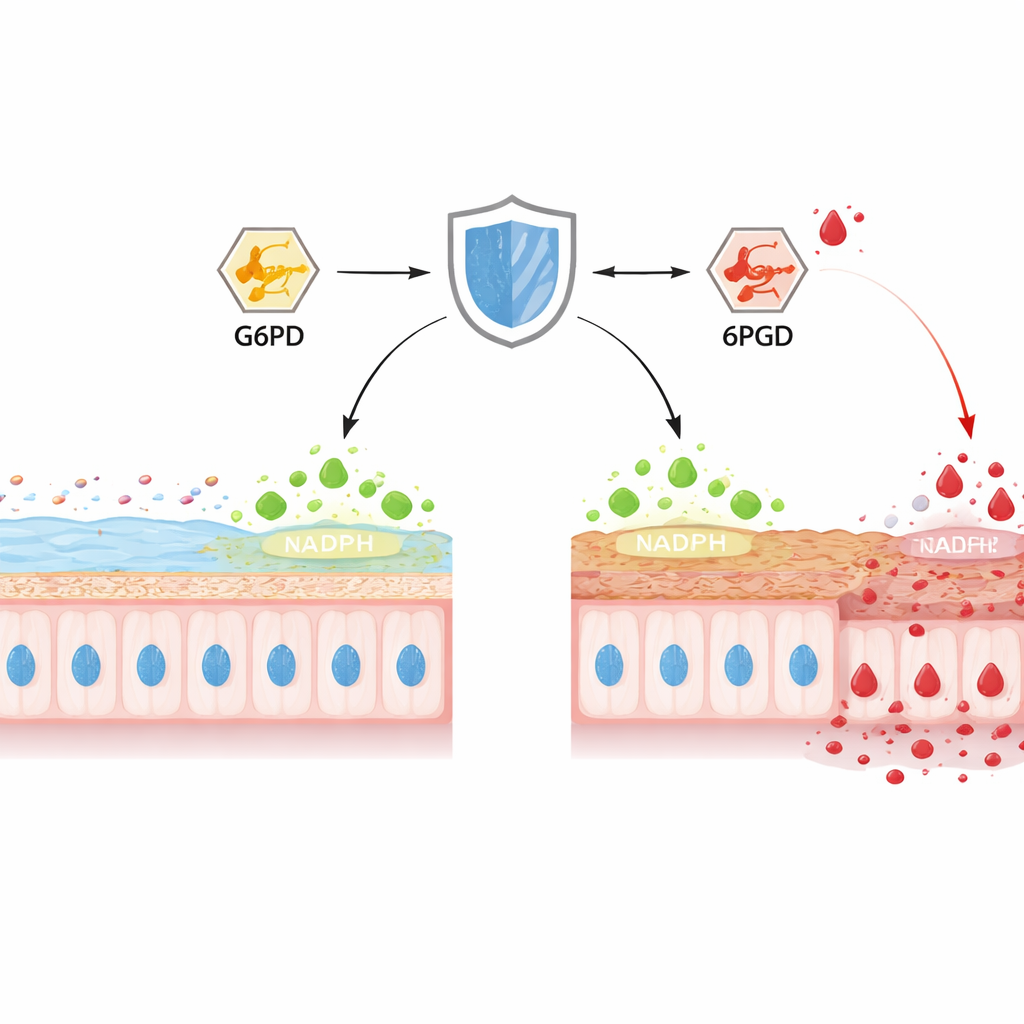
A hidden defender in gut cells
Inside the colon, goblet cells constantly manufacture MUC2, the main mucus protein that forms the inner protective gel. The authors focused on TIGAR, a protein previously known for steering glucose into a pathway that makes NADPH, a key “reducing power” that keeps harmful oxidants in check. In mice with chemically induced colitis, TIGAR levels in the colon dropped over several days, just as the mucus layer thinned, the epithelial surface eroded, and bacteria crept closer to the tissue. When the researchers selectively deleted TIGAR only in the intestinal lining of mice, colitis started earlier and became more severe, with distorted crypts, fewer goblet cells, and markedly less mature MUC2. Measures of cellular chemistry revealed depleted NADPH, weakened antioxidant systems, and surges in reactive oxygen and nitrogen species, linking the loss of TIGAR to intense oxidative stress and barrier failure.
Sugar use rewired toward wasteful burning
To understand how TIGAR shapes metabolism, the team profiled hundreds of small molecules in goblet‑like cell lines and in mouse colon tissue. Removing TIGAR did not change how much glucose cells imported or how quickly whole animals burned oxygen, but it profoundly altered the fate of that glucose. Using labeled sugar, they showed that less carbon flowed through the pentose phosphate pathway—the main source of NADPH—while more was pushed into classic glycolysis, ending in lactate. Two metabolites stood out: 6‑phosphogluconate, a hub in the pentose phosphate pathway, built up strongly, and lactate levels climbed in cells and tissues lacking TIGAR, especially during colitis. Gene‑expression data suggested that enzymes of glycolysis were cranked up, whereas the protein levels of two key pentose phosphate enzymes, G6PD and 6PGD, were unchanged, hinting that their activity—not their abundance—was being blocked.
A chemical tag that disarms antioxidant enzymes
The researchers then uncovered the blocking mechanism: lactylation, a chemical tag added to proteins using lactate as a donor. In colitis models and in inflammatory cell cultures, overall protein lactylation rose, and both G6PD and 6PGD carried high levels of this modification. Mass‑spectrometry pinpointed specific lysine positions on each enzyme—K432 on G6PD and K38 on 6PGD—as major lactylation sites. Structural modeling and mutational tests showed that modifying these spots interferes with the way G6PD forms its active dimer and how 6PGD binds its cofactor, sharply reducing their ability to generate NADPH. Blocking lactate production or preventing lactylation at these sites restored enzyme activity, increased NADPH, lowered oxidative stress, and improved MUC2 maturation. In TIGAR‑deficient mice, delivering mutant versions of G6PD and 6PGD that could not be lactylated preserved mucus thickness and reduced bacterial intrusion into the inner layer.
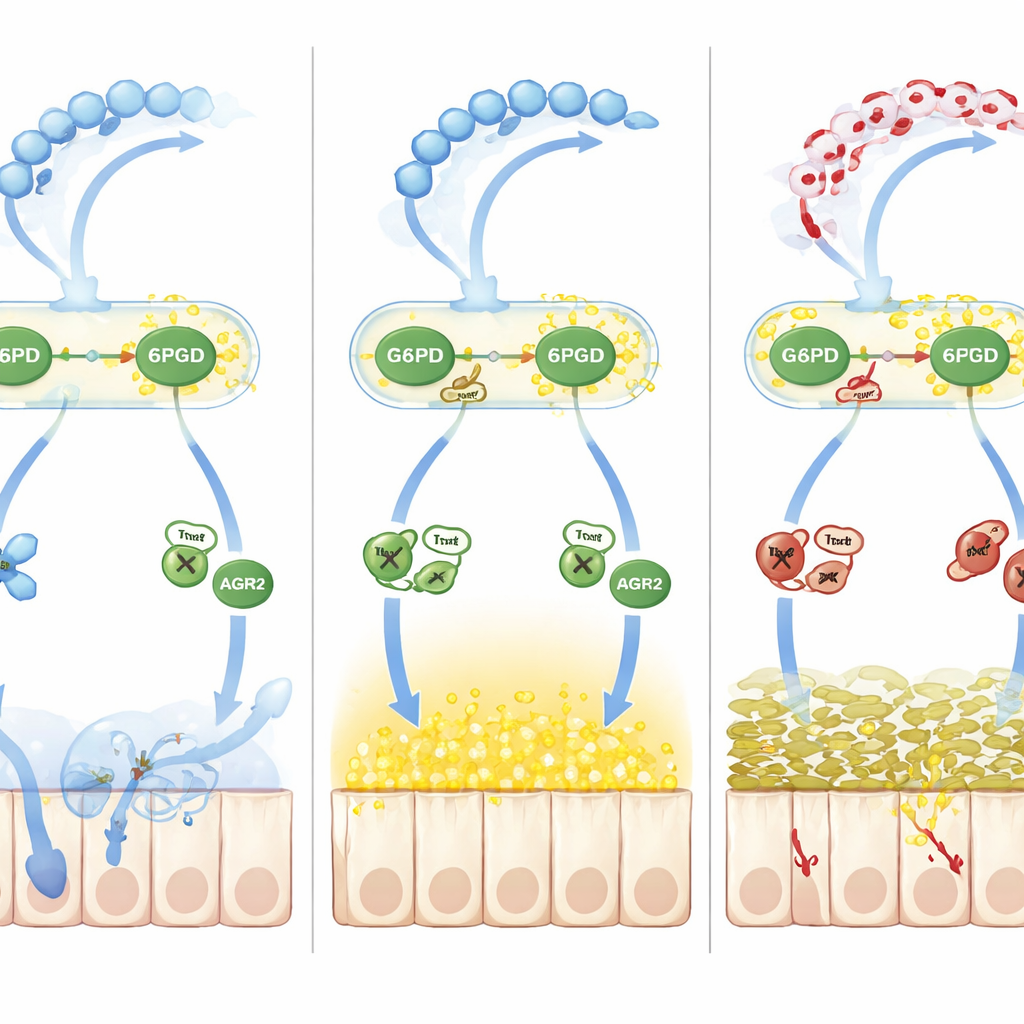
How redox imbalance derails mucus processing
Beyond simple oxidation, the team traced a second wave of damage driven by nitric oxide. Elevated nitric oxide in colitis increased S‑nitrosylation, another reversible chemical mark on cysteine residues. A folding helper protein called AGR2, essential for converting MUC2 precursors into their mature form in the endoplasmic reticulum, became heavily S‑nitrosylated at a critical cysteine (Cys81). This change weakened AGR2’s grip on MUC2 precursors, causing misfolded proteins to accumulate and endoplasmic‑reticulum stress to rise, further shrinking the pool of correctly formed mucus. The key go‑between was thioredoxin‑1 (Trx1), a redox‑sensitive enzyme that can either remove or transfer nitrosyl groups depending on its own oxidation state. When NADPH was scarce because G6PD and 6PGD were lactylated and sluggish, Trx1 shifted from a protective denitrosylating mode to a damaging transnitrosylating mode, shuttling nitrosyl groups onto AGR2 and deepening the mucus defect.
What this means for people with colitis
Taken together, the work outlines a chain reaction: chronic inflammation lowers TIGAR in gut epithelial cells; this diverts glucose away from NADPH‑producing routes, boosts lactate, and lactylates G6PD and 6PGD; NADPH production falls, Trx1 becomes over‑oxidized, AGR2 is excessively nitrosylated, and MUC2 fails to mature, leaving the mucus barrier thin and leaky. For a lay reader, the message is that a subtle shift in how gut cells burn sugar can translate into a very physical breach in the gut’s slimy shield. By protecting TIGAR function, preventing harmful lactylation of G6PD and 6PGD, or rebalancing Trx1 and AGR2 chemistry, future treatments may help reinforce the mucus barrier and tame inflammation in ulcerative colitis.
Citation: Wu, D., Su, S., Zhang, P. et al. TIGAR regulates intestinal mucus barrier integrity by inhibiting lactylation of G6PD/6PGD in ulcerative colitis. Nat Commun 17, 3382 (2026). https://doi.org/10.1038/s41467-026-70263-z
Keywords: ulcerative colitis, intestinal mucus barrier, TIGAR, oxidative stress, glucose metabolism