Clear Sky Science · en
Dynamic bond-driven encapsulation of enzymes in metal–organic frameworks beyond pore size constraints
Why trapping fragile helpers matters
Enzymes are nature’s tiny workhorses, speeding up chemical reactions that power everything from living cells to industrial manufacturing. But outside their natural homes, these delicate proteins quickly fall apart, making them hard to reuse at scale. This article reports a way to tuck enzymes safely inside a porous crystalline material, so they stay active longer and can be recycled repeatedly. Crucially, the team shows how to slip in enzymes that are actually larger than the material’s built‑in openings, sidestepping a long‑standing size limit that has held back many promising applications in green chemistry and medicine.
Building a protective crystal house
The researchers work with metal–organic frameworks, or MOFs—solids made from metal clusters linked by organic “struts” into an orderly scaffold full of empty spaces. MOFs are appealing homes for enzymes because they offer large internal surface areas and protective cages that can shield proteins from heat, solvents, and harsh conditions. The catch is that many useful enzymes are simply too big to squeeze through the narrow windows of most MOFs. Earlier solutions either fastened enzymes to the outside surface, which limited how many could be loaded, or tried to grow the MOF around the enzymes under very gentle conditions, which only works for a small subset of MOF chemistries.
Adding tiny gates that open and heal
To escape this size trap, the authors designed a family of MOFs whose internal connections can briefly unhook and reattach like self‑healing stitches. They combined very stable metal–carboxylate clusters, which act as a rigid backbone, with more flexible metal–pyridyl links that behave as reversible “gates.” By swapping different metals and changing the length of the organic linkers, they could finely tune how easily these gates open in water without causing the whole structure to fall apart. Measurements of metal leaching, X‑ray diffraction, and computer simulations all pointed to the same picture: in water, only the softer connections at specific metal sites loosen, while the tougher backbone stays intact, preserving the overall crystal framework.
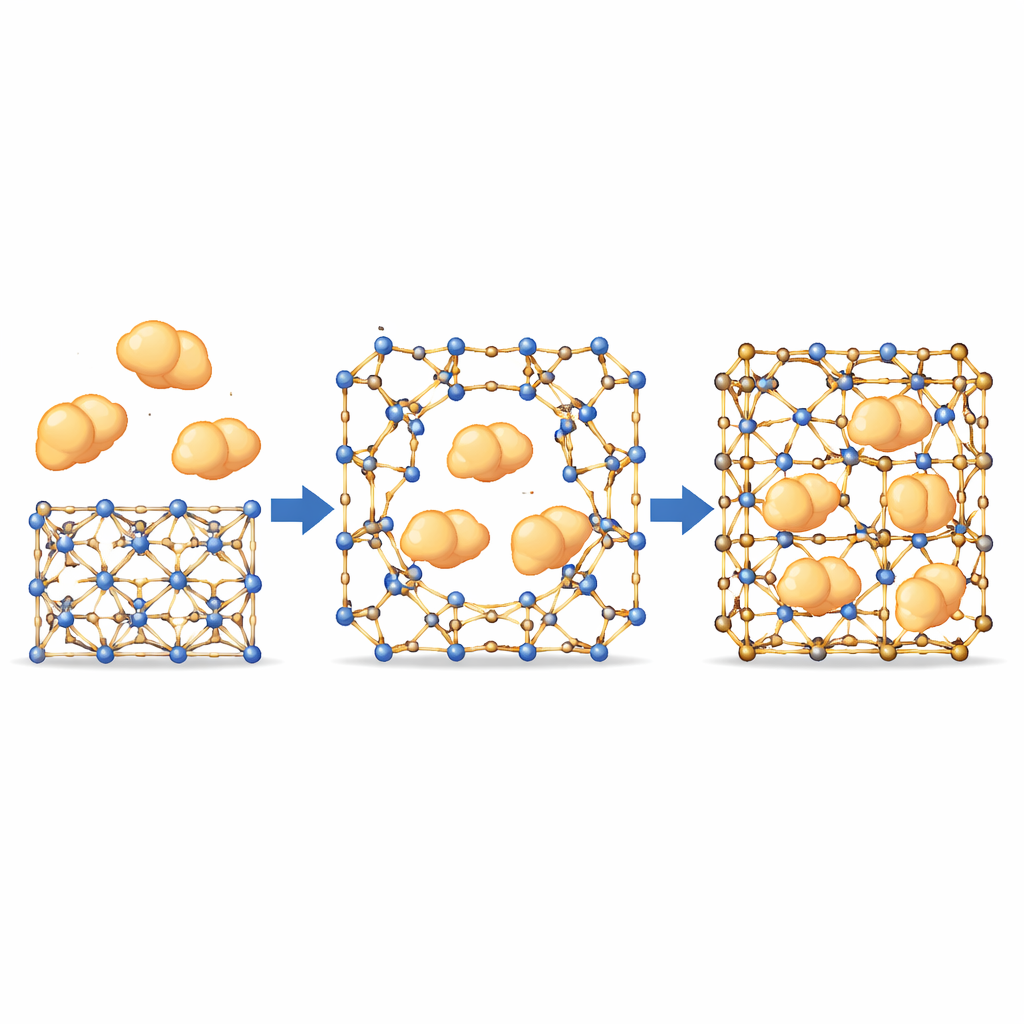
Letting big enzymes slip through small pores
With this gate‑like behavior in hand, the team tested whether large enzymes—bigger than the MOF’s nominal pores—could still be drawn inside. Using fluorescently labeled proteins and confocal microscopy, they watched the enzymes move uniformly from the crystal surface toward the interior, following paths that did not match the fixed internal channels. This suggests that temporary openings form where dynamic bonds briefly separate, allowing the enzymes to infiltrate before the framework “heals” around them. Importantly, tougher variants of the material, where these bonds barely move, showed almost no enzyme uptake, confirming that motion at these sites is essential. Across a panel of enzymes with different sizes and properties, the most finely balanced MOFs achieved high loading while still keeping their crystal order.
Stronger, longer‑lasting catalysts
Once inside, the enzymes behaved as robust, reusable catalysts. The authors examined several representative proteins, including ones that are notoriously sensitive to heat, alkaline conditions, and organic solvents. Compared with the same enzymes free in solution, the encapsulated versions kept much more of their activity when stressed, and could be cycled through multiple reaction runs with little loss of performance. The dynamic MOFs also enabled the co‑encapsulation of two cooperating enzymes that work in sequence to make a valuable sugar‑based building block. Housing both inside the same framework shortened the path between them, boosting reaction efficiency and allowing the system to run repeatedly with sustained output.
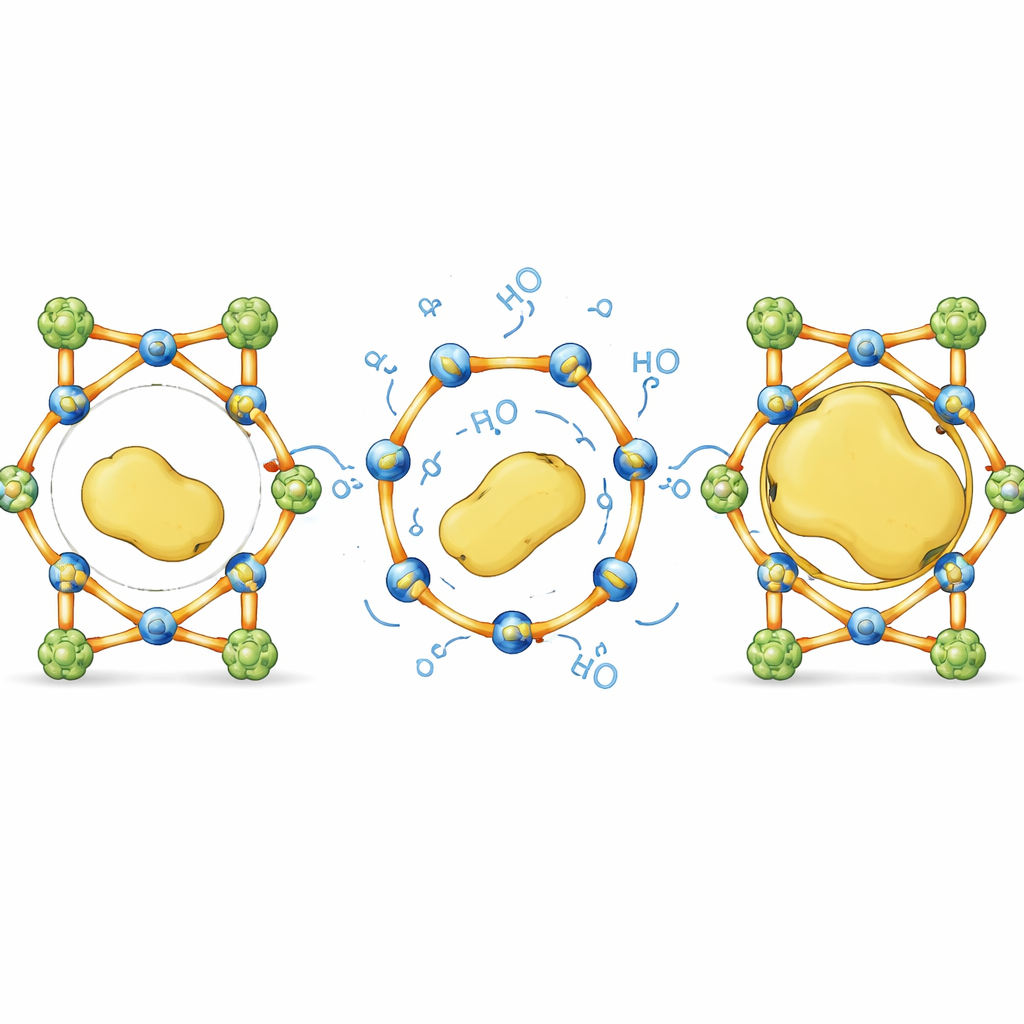
From size limits to size‑blind design
In everyday terms, this work turns a rigid sponge with fixed holes into a smart cage whose bars can flex just enough to admit large guests and then snap back into place. By carefully balancing unbreakable supports with selectively reversible links, the authors show that it is possible to pack bulky, fragile enzymes inside crystalline materials that would normally exclude them, without sacrificing structure or function. This dynamic bond‑driven approach offers a general recipe for designing size‑independent “enzyme hotels,” with potential to improve industrial biocatalysts, streamline multi‑step syntheses, and create more durable bio‑based technologies.
Citation: Li, Y., Qiao, M., Gao, L. et al. Dynamic bond-driven encapsulation of enzymes in metal–organic frameworks beyond pore size constraints. Nat Commun 17, 3642 (2026). https://doi.org/10.1038/s41467-026-70249-x
Keywords: enzyme immobilization, metal-organic frameworks, biocatalysis, dynamic coordination bonds, multi-enzyme cascades