Clear Sky Science · en
Spatial transcriptomics uncovers vasculature-centered cellular interactions driving Japanese encephalitis progression in a mouse model
Why this brain infection study matters
Japanese encephalitis is a mosquito-borne brain infection that can leave survivors with lifelong movement and memory problems, especially children. Yet doctors still lack drugs that directly target the virus or the damaging inflammation it triggers in the brain. This study uses a powerful mapping technology to watch, almost cell by cell, how the virus and the body’s own immune cells move through the mouse brain. By doing so, it reveals a surprising partnership between blood vessels and roaming immune cells that helps explain how the disease becomes so destructive.
A virus that turns the brain’s defenses against itself
Japanese encephalitis virus (JEV) is carried by mosquitoes and threatens more than three billion people across Asia and the Western Pacific. Most infections are silent, but when the virus reaches the brain it can cause high fever, seizures, and coma. Vaccines reduce risk, but many people remain unprotected, and there is no specific antiviral treatment. Scientists have long suspected that the way immune cells enter the brain, and how they interact with brain blood vessels, is central to the illness. However, traditional methods could not easily show which exact cells were infected and how they were arranged in the delicate architecture of the brain.
A molecular map of infection in space and time
The researchers infected female mice with JEV and examined their brains on days 3, 5, and 7 after infection, spanning the rise in virus levels and the onset of severe symptoms. They used a technology called spatial transcriptomics, which reads out which genes are active at thousands of tiny points across a thin brain slice, while preserving where each point lies in the tissue. Custom probes allowed them to detect both mouse and viral RNA on the same slide. By combining these maps with brain atlases and cell-type references, they built a spatiotemporal atlas: which brain regions the virus favors, which cell types it infects, and how local immune responses unfold.
Blood vessels as gateways and amplifiers
The atlas showed that JEV first appears in a small fraction of brain tissue, then expands to heavily involve the thalamus and cerebral cortex. Even more striking was where the strongest immune signals appeared: along blood vessels, especially in the thin membranes (meninges) that wrap the brain and in the choroid plexus, a tissue that helps make cerebrospinal fluid. Here, a special subtype of endothelial cells—cells that line blood vessels—marked by a receptor called Ackr1 became highly activated. These cells produced many chemokines, molecules that attract immune cells, and turned on genes linked to inflammation while turning down genes that maintain tight junctions, the seals that keep the blood–brain barrier intact. At the same time, tests with a dye confirmed that the barrier had indeed become leaky.
Monocytes: Trojan horses and signal boosters
Among all immune cells entering the brain, one stood out: a population of Ly6c2-positive monocytes. Spatial maps and flow cytometry showed these cells were often infected with JEV, making them likely carriers that ferry the virus across damaged vessels into the brain—much like a Trojan horse. They did more than transport the virus. These monocytes produced large amounts of interferon-gamma, a potent immune messenger usually linked to T cells and natural killer cells. Interferon-gamma is known to help clear viruses but also to worsen blood–brain barrier damage. The study found that monocytes clustered near Ackr1-positive endothelial cells and communicated with them through multiple chemokines, forming a feedback loop that drew in even more immune cells. Around these vessels, many cells—including monocytes themselves—showed molecular signatures of explosive forms of cell death (pyroptosis and necroptosis) that spill inflammatory contents into surrounding tissue.
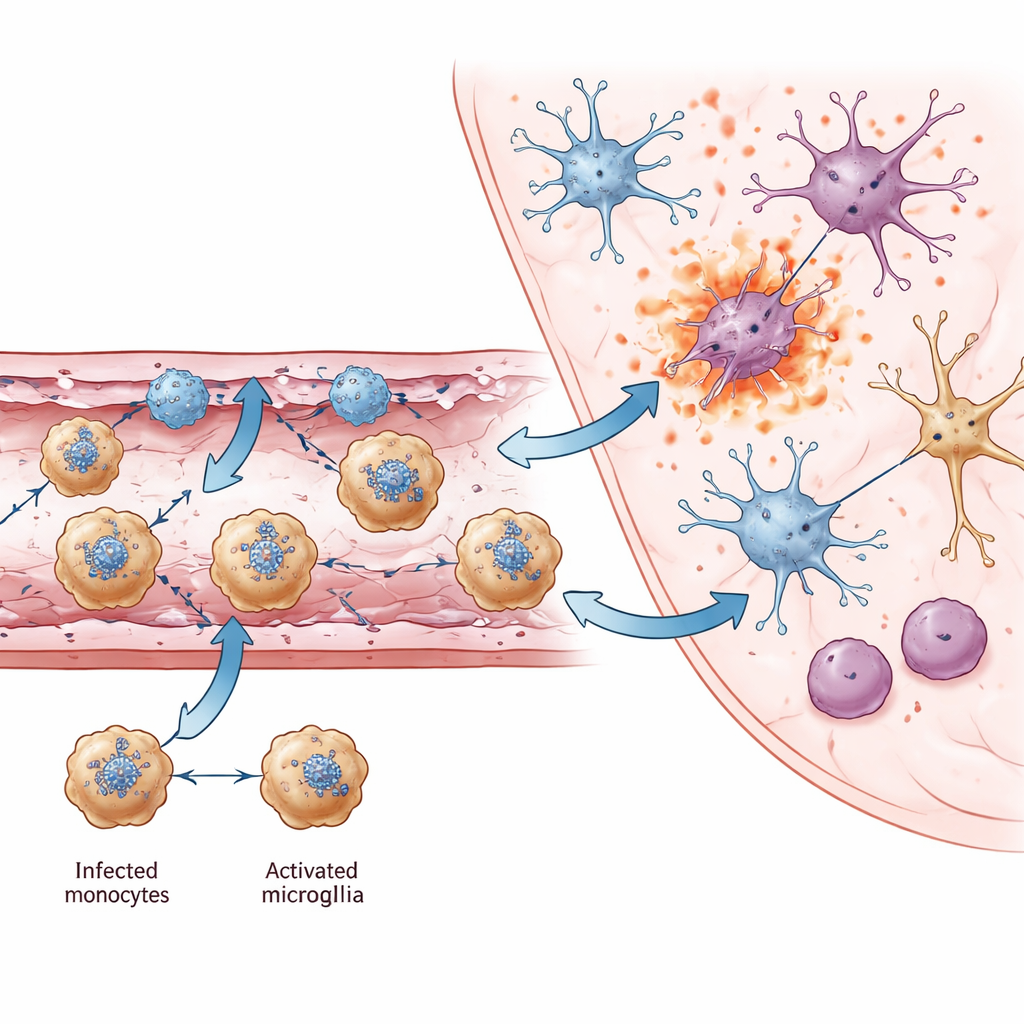
From local vascular damage to widespread brain injury
As infection progressed, resident brain cells also changed. Microglia, the brain’s innate immune sentinels, shifted into an activated state marked by a chemokine called Ccl12 and by strong antiviral gene activity. While these changes help fight the virus, they also coincided with loss of genes involved in synapses, learning, and memory. In heavily infected regions such as the thalamus, different neuron subtypes that tested positive for viral RNA showed elevated signals of pyroptosis and necroptosis, pointing to inflammatory cell death as a key cause of neuron loss. Across multiple brain regions, the authors identified common neuron genes whose reduced activity has also been linked to cognitive and motor problems in other brain diseases, suggesting a shared molecular basis for the long-term disabilities seen in survivors.
What this means for future treatments
To a lay reader, the core message is that Japanese encephalitis is not just a story of a virus attacking neurons. It is also a story of infected monocytes and specialized blood vessel cells forming a damaging alliance at the brain’s borders. Together they loosen the blood–brain barrier, draw in waves of immune cells, and trigger inflammatory forms of cell death that radiate from vessels into brain tissue. By pinpointing these key players—Ly6c2-positive monocytes and Ackr1-positive endothelial cells—the study suggests new therapeutic angles: drugs that reduce specific chemokines, block the Ackr1 receptor, or limit monocyte recruitment and pyroptosis could potentially blunt the worst brain damage while allowing antiviral defenses to work.
Citation: Ou, Z., Wang, Z., Chen, Q. et al. Spatial transcriptomics uncovers vasculature-centered cellular interactions driving Japanese encephalitis progression in a mouse model. Nat Commun 17, 4089 (2026). https://doi.org/10.1038/s41467-026-70047-5
Keywords: Japanese encephalitis, brain inflammation, blood–brain barrier, monocytes, spatial transcriptomics