Clear Sky Science · en
Impaired $$\alpha$$ -Synuclein aggregate clearance in neuronal cells drive their spread to microglia through tunneling nanotubes
Why tiny protein clumps matter for brain health
Parkinson’s disease and related disorders are marked by the slow build‑up of clumps of a protein called alpha‑synuclein inside brain cells. These clumps can damage cells, but the brain is not defenseless: it has its own clean‑up crews and support cells. This study explores how nerve cells and brain immune cells share the burden of clearing these clumps, and how a little‑known type of cell‑to‑cell bridge, called a tunneling nanotube, becomes a highway for moving toxic material out of struggling neurons and into hardier microglia.
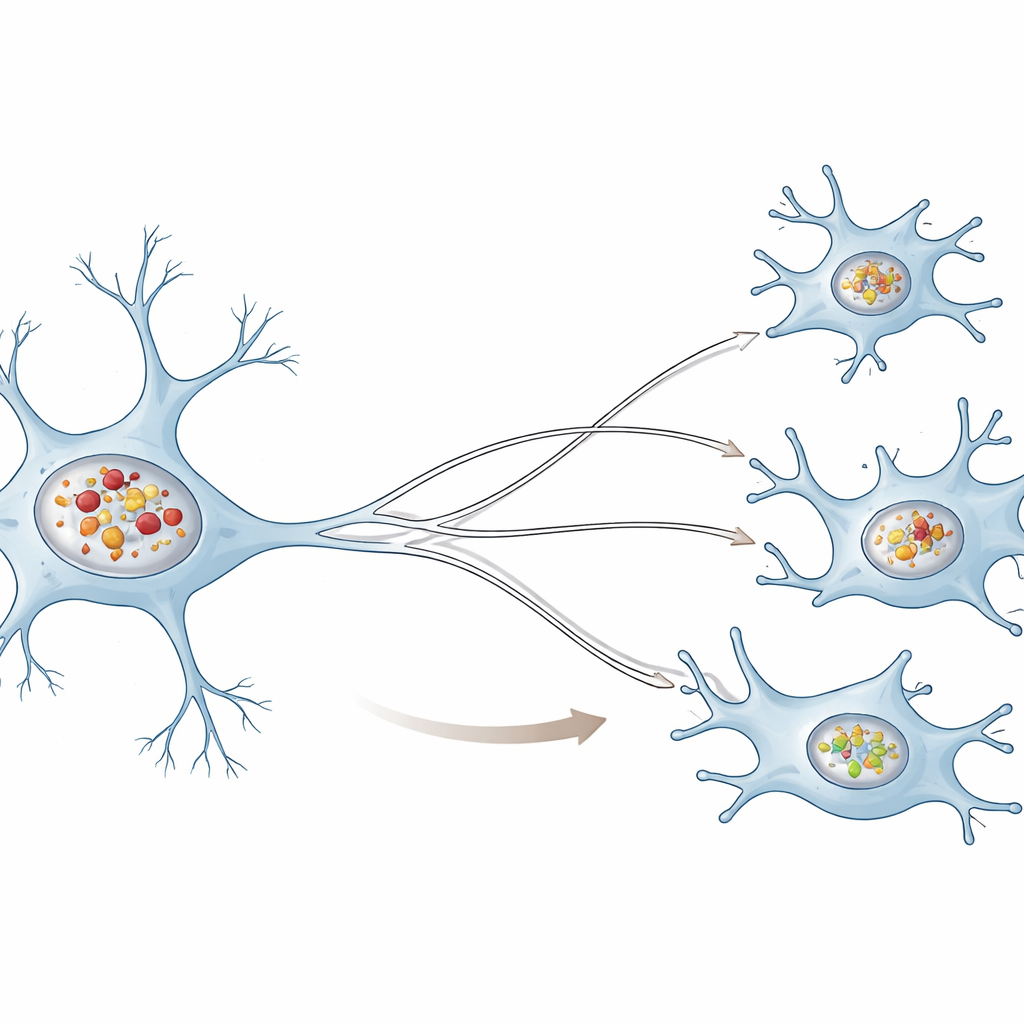
Two very different clean‑up crews
The authors compared human neuron‑like cells with human microglia, the brain’s resident immune cells, and exposed both to pre‑formed alpha‑synuclein fibrils, a laboratory version of the harmful clumps seen in disease. In both cell types, the incoming clumps were taken up and sent to small recycling compartments called lysosomes. However, neurons accumulated far more clumps inside their lysosomes than microglia did. Over time, neuronal lysosomes became less acidic and less able to break down a test protein, showing that their core recycling function was faltering. Microglial lysosomes, in contrast, largely preserved their acidity and breakdown capacity even when they contained alpha‑synuclein.
When recycling fails, damage spreads
Damaged lysosomes can leak and rupture. Using markers of membrane breaks and repair proteins, the researchers found that neurons experienced much more lysosomal injury than microglia after alpha‑synuclein exposure. The affected lysosomes in neurons lost mobility and clustered, another sign of dysfunction. Microglia responded differently: they turned on a process called lysophagy, a targeted removal of damaged lysosomes, and at the same time boosted the birth of new lysosomes. A master control protein that drives lysosome and clean‑up gene programs moved into the nucleus of both cell types, but only in microglia did the downstream genes switch on robustly. This asymmetry means neurons struggle to restore their recycling system, while microglia can renew theirs.
Autophagy: clogged in neurons, boosted in microglia
Protein clumps are normally handled by autophagy, a pathway that wraps unwanted material and delivers it to lysosomes. The team measured autophagy “flux” and found that alpha‑synuclein largely blocked this flow in neurons but not in microglia. Neurons exposed to clumps showed build‑up of a cargo‑adaptor protein (p62) without further increase when the pathway’s final step was chemically blocked, consistent with a stalled conveyor belt. Microglia, by contrast, made more p62 and showed a strong rise when the final step was blocked, indicating a still‑running, even upregulated, pathway. Microscopy revealed many more sites in microglia where alpha‑synuclein, autophagy markers, and lysosomes came together, suggesting that microglia are much better at recognizing and routing clumps for destruction.
Nanotube highways for toxic cargo
What happens when neurons cannot keep up with the trash? The study shows that they pass the problem to microglia. When autophagy in alpha‑synuclein‑laden neurons was chemically inhibited, more clumps were transferred to neighboring microglia, and when autophagy was stimulated, transfer dropped. These exchanges did not depend on material released into the culture medium, pointing instead to direct cell‑to‑cell connections. The authors focused on tunneling nanotubes—thin, actin‑based bridges between cells—and found that blocking autophagy in either neurons or microglia increased both the number of these connections and the amount of general cargo moving through them. Even when microglia were donors rather than recipients, impairing their autophagy increased transfer to neurons, suggesting that flawed self‑cleaning is a signal that encourages cells to connect and pass on aggregates.
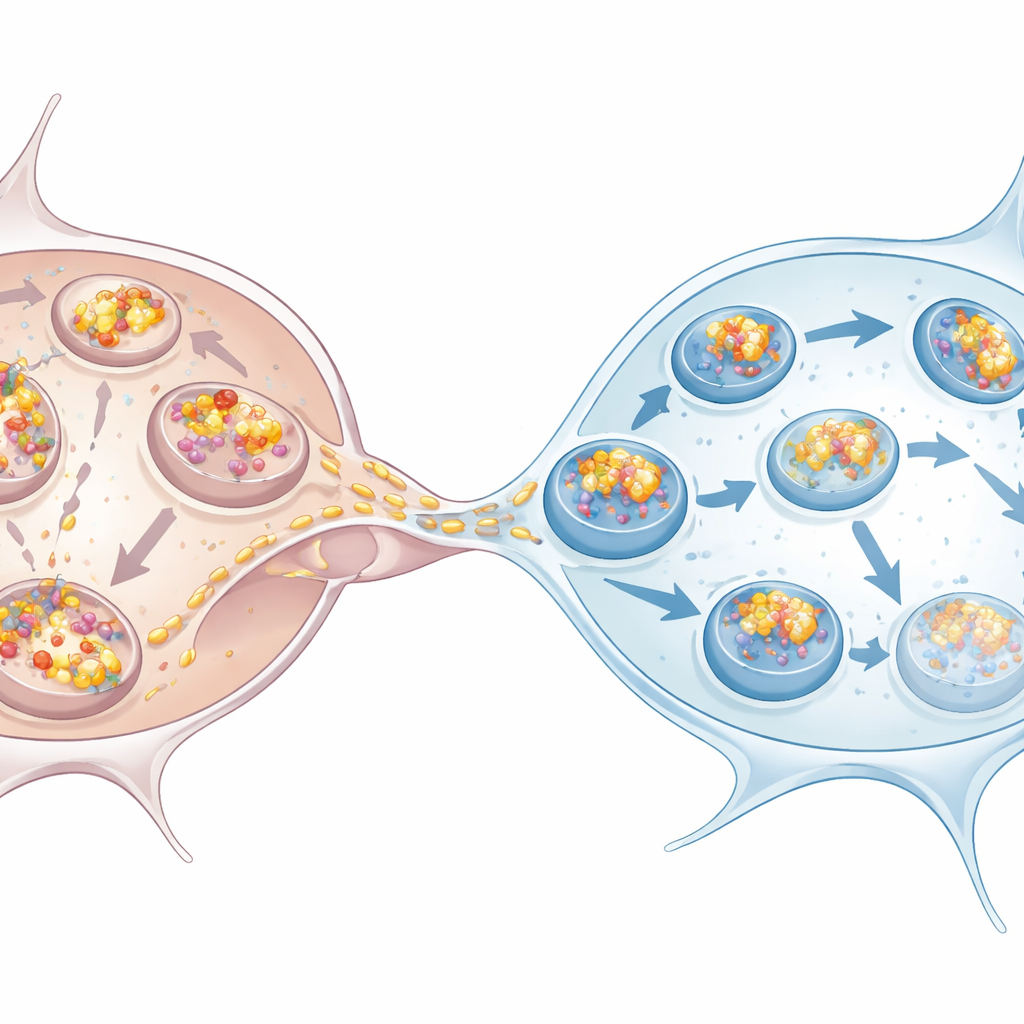
Microglia rise to the challenge
The work also examined human stem‑cell‑derived dopaminergic neurons and microglia, which better resemble cells affected in Parkinson’s disease. The same pattern emerged: neurons built up more alpha‑synuclein, showed signs of impaired autophagy, and relied on microglia to take over disposal. Microglia grown alongside overloaded neurons, or even just exposed to their conditioned medium, ramped up their own autophagy. Once they received alpha‑synuclein via nanotubes, a large fraction of the incoming clumps ended up in p62‑marked structures, consistent with a route toward degradation. Co‑culturing neurons with microglia reduced the number and overall load of clumps inside neurons, confirming that microglia act as effective sinks for toxic aggregates.
What this means for Parkinson’s and beyond
For a non‑specialist, the main message is that not all brain cells are equally equipped to deal with protein waste. Neurons, especially those vulnerable in Parkinson’s disease, have fragile recycling systems that can be overwhelmed by alpha‑synuclein clumps. Microglia, in contrast, are more robust cleaners and can help rescue neurons by taking in and breaking down their excess aggregates through tiny nanotube bridges. But this aid comes at a cost: the same process may also help disease‑related proteins spread through the brain. Therapies that restore neuronal autophagy or fine‑tune microglial clean‑up could not only lower the toxic burden inside individual cells but also slow the cell‑to‑cell propagation of damaging protein clumps.
Citation: Chakraborty, R., Palese, F., Samella, P. et al. Impaired \(\alpha\)-Synuclein aggregate clearance in neuronal cells drive their spread to microglia through tunneling nanotubes. Nat Commun 17, 3832 (2026). https://doi.org/10.1038/s41467-026-69930-y
Keywords: Parkinson’s disease, alpha-synuclein, microglia, autophagy, tunneling nanotubes