Clear Sky Science · en
Epigenetic profiling of hematopoietic stem cells from male mice identifies KDR and PU.1 as regulators of aging transcriptome and caloric restriction response
Why eating less might reshape our blood
Cutting calories without becoming malnourished has been shown to extend lifespan in many animals, and some people even adopt strict diets in the hope of living longer. But eating less can also weaken the immune system. This study asks a simple but important question: how does long-term calorie reduction change the stem cells in bone marrow that constantly rebuild our blood and immune system, and can those changes help keep these cells "young" while still protecting us?
How the study was set up in mice
The researchers worked with male mice that either ate freely or were kept on a lifelong diet with about 40% fewer calories, starting in early adulthood. Some calorie-restricted mice were later allowed to eat freely again, to see how quickly their blood system bounced back. The team measured blood counts, examined cell populations in the bone marrow, and profiled gene activity and epigenetic marks—chemical tags on DNA and proteins that help control which genes are turned on—in the rare hematopoietic stem cells that give rise to all blood cells.
Less total white blood, but survival cells are protected
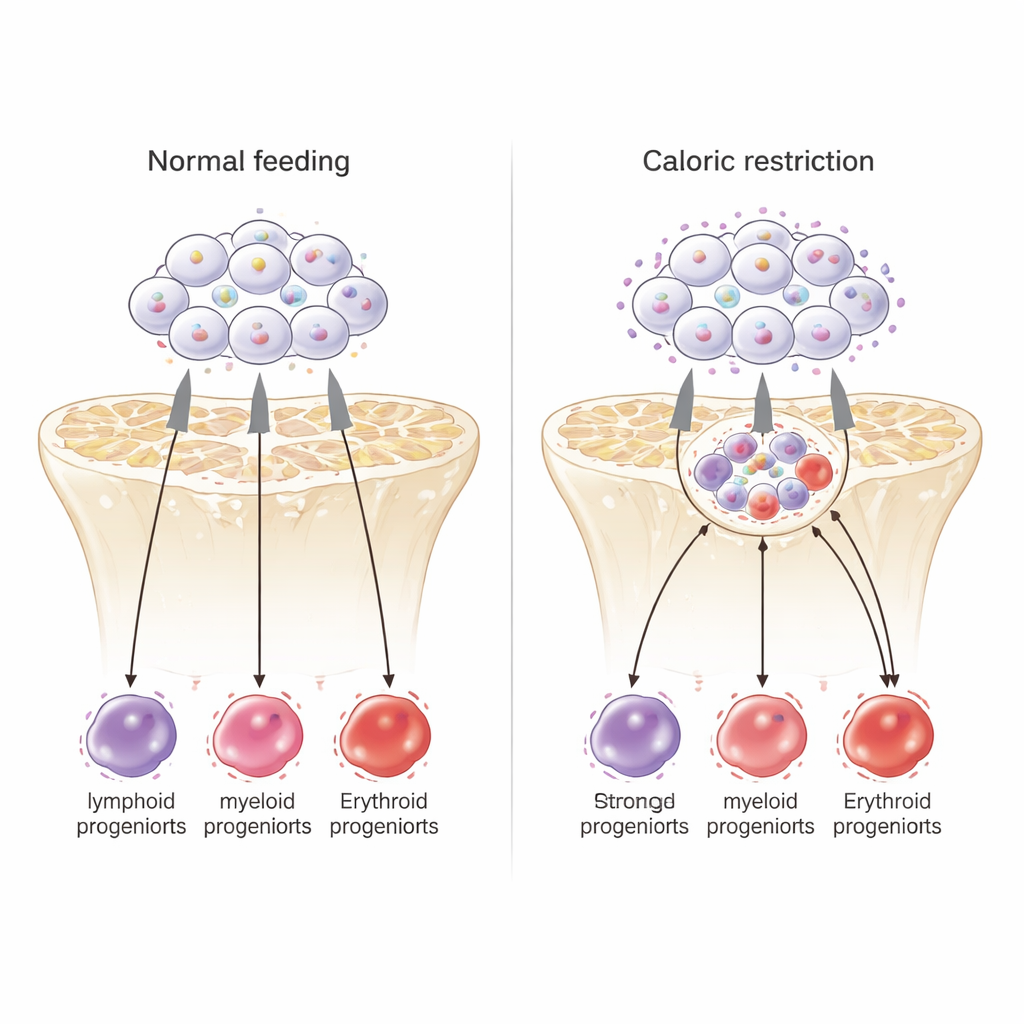
In older mice that had spent most of their lives on fewer calories, overall white blood cell numbers in the bloodstream fell. Both innate and adaptive immune cells dropped, but not equally. Cells important for immediate survival—red blood cells that carry oxygen, platelets that clot blood, and frontline myeloid defenders such as granulocytes—were relatively preserved. In contrast, lymphoid cells, especially B cells that support antibody responses, were strongly reduced from early progenitors in the bone marrow through to mature cells in the blood. When calorie-restricted animals were refed for only four weeks, white blood counts and spleen size largely rebounded, and lymphoid production surged, showing that many of these changes are rapidly reversible.
Stem cells work harder and choose different fates
Surprisingly, the stem cells at the top of the blood hierarchy did not become more dormant under food shortage; instead, they cycled more actively. Detailed gene-expression analyses revealed that, under calorie restriction, these stem cells dialed down programs linked to long-term self-renewal and turned up genes associated with short-term stem cells and progenitors that make myeloid and red blood lineages. In other words, the stem cells were pushed to work harder in the short term to maintain key survival cell types, at the expense of slowly renewing themselves or generating lymphoid cells. Yet, despite this extra work, their overall pattern of gene activity looked more like that of young stem cells than old ones.
Resetting aging programs through epigenetic switches
The team next asked how eating less could make old stem cells look molecularly younger. They mapped two common histone marks that generally promote or repress gene activity. Although both marks were globally reduced in calorie-restricted stem cells, a small set of aging-associated genes gained a repressive mark and lost an activating one. Two stood out: Kdr, which encodes a receptor for blood vessel growth signals, and Bmpr1a, another cell-surface receptor. Under calorie restriction, both genes were more tightly shut down. When the researchers experimentally lowered Kdr and Bmpr1a in aged stem cells from normally fed mice and transplanted these cells into recipients, the cells adopted a gene-expression pattern that resembled that of calorie-restricted stem cells, suggesting these receptors help drive the aging program and can be epigenetically tuned.
An internal traffic cop tunes the response
To uncover internal control points, the scientists examined how accessible the DNA was across the stem-cell genome and where a key transcription factor, PU.1, was bound. Under calorie restriction, many regions of DNA became more open and showed stronger PU.1 binding. Genes near these PU.1 sites were enriched for cell-cycle control and for pathways steering cells toward red blood and other myeloid fates, while genes associated with deep stem-cell quiescence tended to be repressed. About half of the genes that responded when Kdr was knocked down showed PU.1 nearby, and PU.1 itself bound close to the Kdr gene, hinting at a feedback loop. Together, these data position PU.1 as an internal traffic cop that, in the context of lowered KDR signaling, redirects aging stem cells toward a calorie-restricted, youth-like state that favors survival-oriented blood cells.
What this means for healthy aging
This work paints a nuanced picture of how long-term calorie cutting affects our blood factory. In male mice, eating less pushes stem cells to prioritize making red cells, platelets, and fast-acting immune cells while holding back antibody-producing cells, which may explain weakened responses to infections under strict diets. At the same time, carefully targeted epigenetic changes at genes such as Kdr and shifts in PU.1 activity help preserve a more youthful gene program in these stem cells. Although the study was done in mice and only in males, it suggests that future therapies could aim to mimic the beneficial molecular effects of calorie restriction on blood-forming stem cells—without requiring extreme dieting or sacrificing immune protection.
Citation: Zong, L., Park, B., Tekin-Turhan, F. et al. Epigenetic profiling of hematopoietic stem cells from male mice identifies KDR and PU.1 as regulators of aging transcriptome and caloric restriction response. Nat Commun 17, 2978 (2026). https://doi.org/10.1038/s41467-026-69718-0
Keywords: caloric restriction, hematopoietic stem cells, immune aging, epigenetics, PU.1 and KDR