Clear Sky Science · en
R-loop editing by DNA cytosine deaminase APOBEC3B modulates the activity of oestrogen receptor enhancers
Why this research matters
Many breast cancers depend on the hormone oestrogen to grow. The same signals that switch on vital genes can also damage DNA, helping tumours evolve and resist treatment. This study uncovers how a naturally occurring DNA‑editing enzyme, APOBEC3B, works together with oestrogen receptors at regulatory DNA switches called enhancers. By showing exactly where and how this enzyme acts, the work links normal hormone‑driven gene control to the mutations and breaks that destabilise the cancer genome and suggests new ways to target hormone‑dependent tumours.
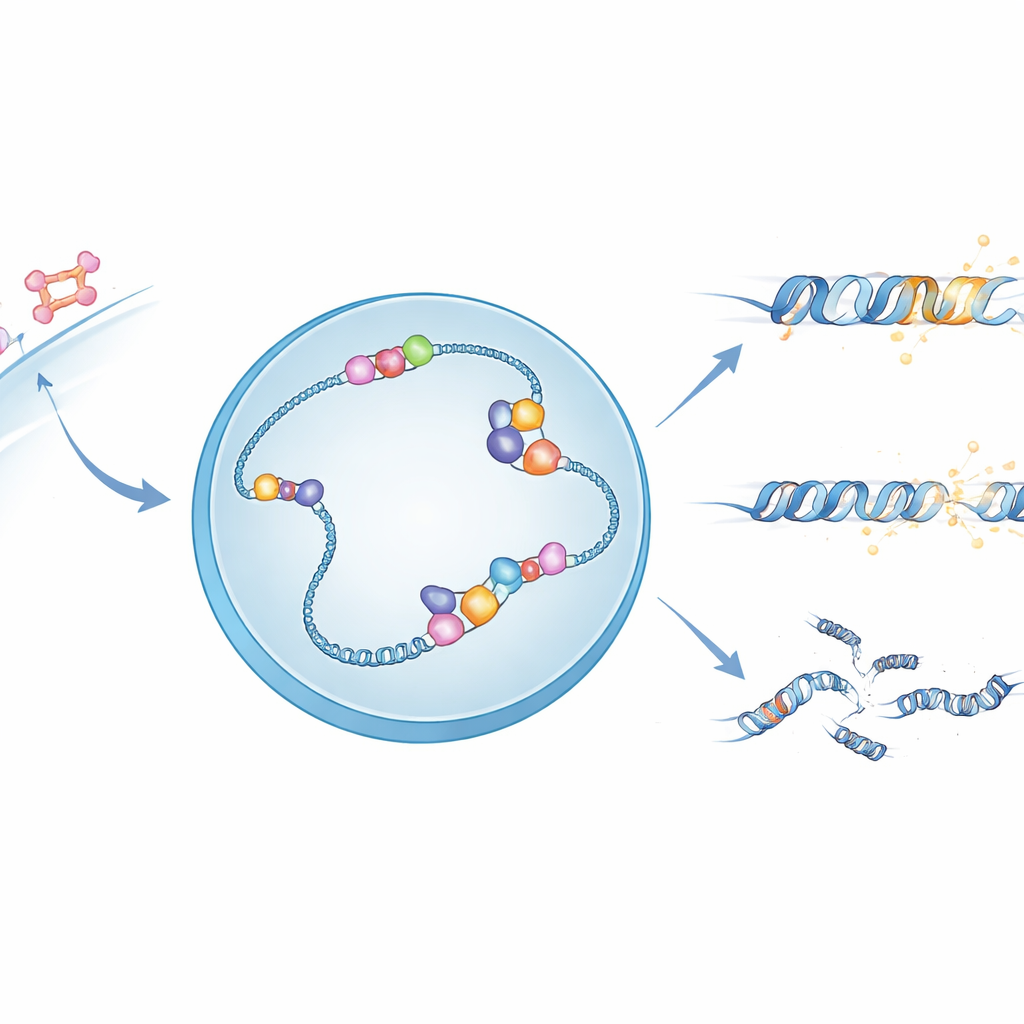
Hormone signals and fragile DNA structures
When oestrogen enters a cell, it activates the oestrogen receptor, a protein that travels to the nucleus and binds specific DNA sites to control nearby genes. At many of these sites, the act of reading DNA into RNA creates unusual three‑stranded structures called R‑loops, where the new RNA sticks to one DNA strand and leaves the other strand exposed. These R‑loops can help regulate genes, but they also make the DNA more fragile and prone to breakage. The authors suspected that APOBEC3B, an enzyme already known to chemically tweak single‑stranded DNA, might be using the exposed strand in R‑loops as its preferred working surface.
Mapping where the DNA editor strikes
To see APOBEC3B’s footprint across the genome, the team engineered breast cancer cells in which a key DNA repair pathway, base excision repair, was blocked. This meant that chemical edits made by APOBEC3B would remain visible long enough to be captured by whole‑genome sequencing. They found that the characteristic mutations made by this enzyme clustered not in protein‑coding regions, but in regulatory DNA near gene start sites and enhancers, especially in zones that replicate early and are highly active. These edited spots tended to lie in sequences biased toward the building blocks that favour R‑loop formation, hinting that APOBEC3B is intimately linked to these transient DNA:RNA structures.
Direct proof that APOBEC3B works on R-loops
The researchers then used two genome‑wide assays to watch APOBEC3B and R‑loops in action. One method pulled down DNA:RNA hybrids, while another captured single DNA strands bound by tagged APOBEC3B. Together, they showed that the enzyme sits right inside R‑loops and chemically converts cytosine bases to uracil on the exposed DNA strand. When an enzyme that erases R‑loops (RNase H1) was overproduced, APOBEC3B binding dropped sharply, confirming that R‑loops are essential docking platforms. Upon short oestrogen stimulation, both R‑loops and APOBEC3B binding surged at promoters and, in particular, at actively transcribed enhancers used by the oestrogen receptor, tying hormone signalling to bursts of R‑loop‑dependent DNA editing.
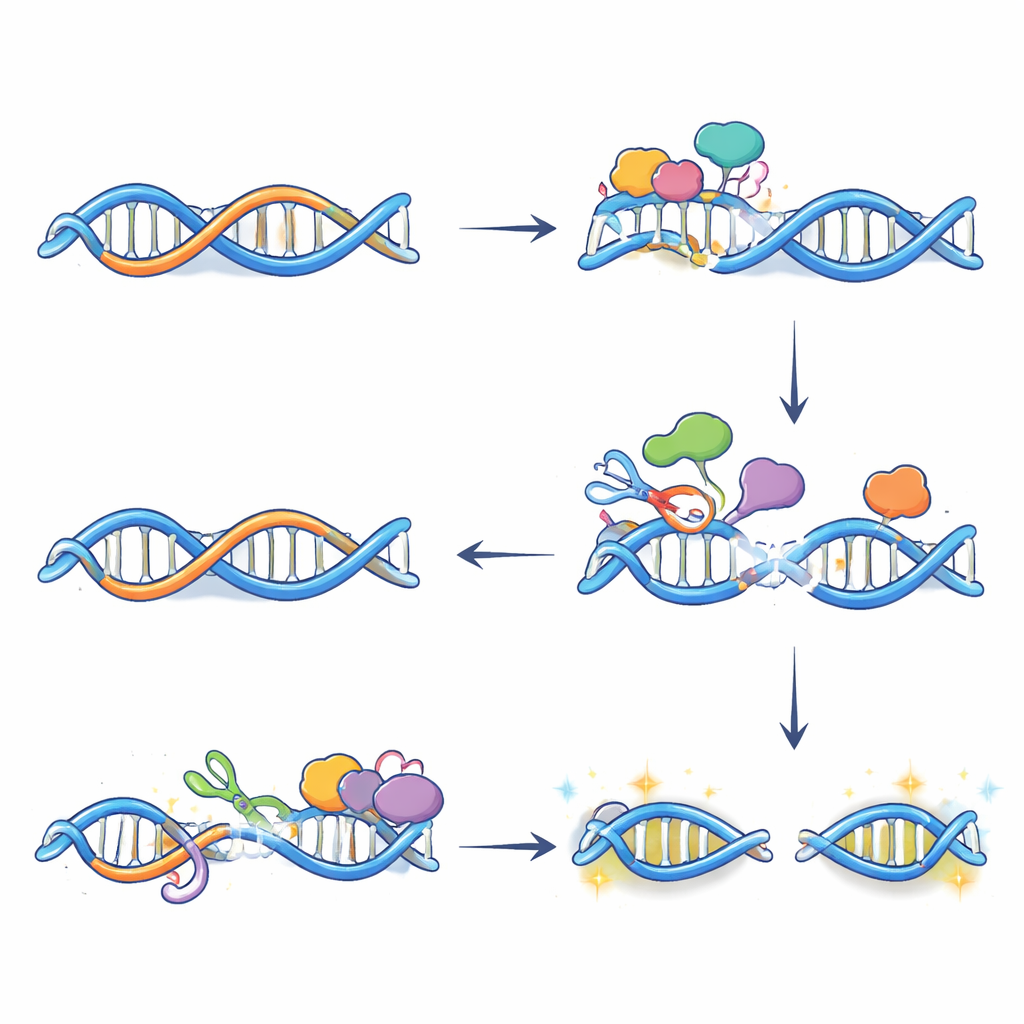
From chemical edits to DNA breaks and gene activation
Editing alone does not break DNA; the damage arises when repair systems process these altered bases. Using a technique that labels the ends of broken DNA, the authors showed that many double‑strand breaks appearing after oestrogen treatment depend on APOBEC3B and occur exactly where R‑loops and the enzyme coincide. Further experiments revealed that two repair pathways—base excision repair and transcription‑coupled nucleotide excision repair—work together at these sites. Each introduces cuts on opposite DNA strands near the R‑loop; when the cuts meet, a full break forms. Blocking either APOBEC3B’s activity or the repair steps reduced these breaks and blunted the normal surge in expression of many oestrogen‑responsive genes, indicating that carefully controlled DNA damage is actually helping establish a more open, active chromatin state at key enhancers.
What this means for cancer and therapy
This work paints APOBEC3B as a double‑edged sword in hormone‑driven breast cancer. By editing DNA at R‑loops near oestrogen receptor enhancers, it helps create temporary DNA breaks that remodel chromatin and boost the expression of growth‑promoting genes. At the same time, these events leave scars—mutations and structural changes—that fuel genomic instability and tumour evolution. Understanding this coupling between hormone signalling, R‑loop biology and DNA editing highlights APOBEC3B and its associated repair steps as potential drug targets. Inhibiting this circuit could dampen harmful gene activation and slow the genetic diversification that underlies resistance to treatments like tamoxifen.
Citation: Zhang, C., Lu, Yj., Chen, B. et al. R-loop editing by DNA cytosine deaminase APOBEC3B modulates the activity of oestrogen receptor enhancers. Nat Commun 17, 2887 (2026). https://doi.org/10.1038/s41467-026-69679-4
Keywords: oestrogen receptor, APOBEC3B, R-loops, breast cancer, DNA damage