Clear Sky Science · en
Spatiotemporal organization of membrane protein controls bacterial extracellular electron transfer
How Bacteria Move Electricity Outside Their Cells
Some bacteria can literally breathe minerals or electrodes instead of oxygen, moving tiny electrical charges in and out of their cells. This electrical trick fuels their survival in oxygen-poor environments and powers technologies like microbial fuel cells and bioreactors that clean up pollution. The study described here asks a deceptively simple question: how do the key proteins in a microbial cell arrange themselves in space and time so that this long-distance electron flow actually works?
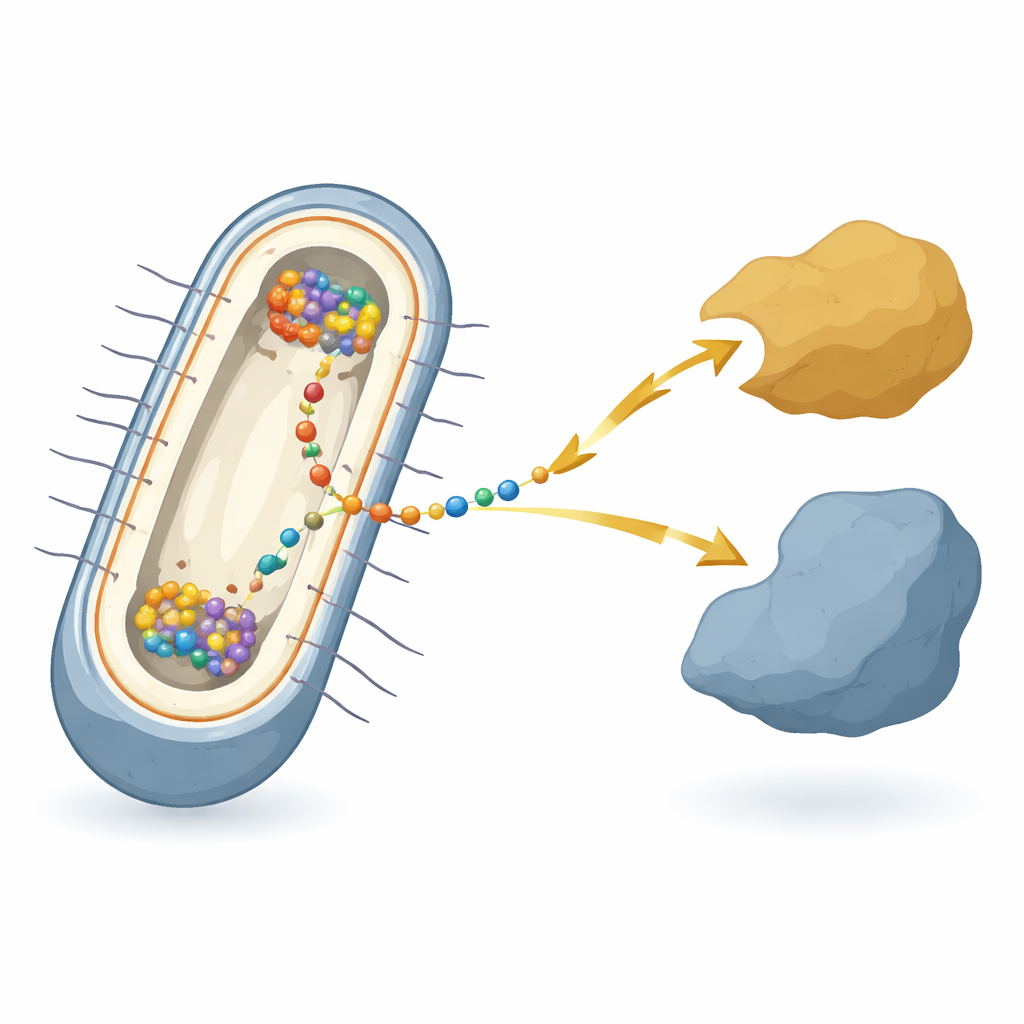
A Microbe That Lives on Rust and Wires
The work focuses on the bacterium Shewanella oneidensis, a model species famous for “extracellular electron transfer” — handing electrons to or from metals, minerals, or electrodes outside the cell. In these bacteria, electrons must cross two membranes and a gap between them, passing through a relay of many different proteins. At the heart of this relay, in the inner membrane, sits a hub protein called CymA. CymA talks to the cell’s internal metabolism on one side and to shuttle proteins in the space between the membranes on the other. Despite decades of work on the individual parts, it has not been clear how all of these components are coordinated inside each living cell when the bacterium actually performs this electrical respiration.
Proteins Self-Organize Into Bright Spots
Using advanced fluorescence microscopy that can track single proteins in single cells, the researchers tagged CymA with glowing markers and watched its behavior under different growth conditions. When the bacteria grew with oxygen, a state where CymA is idle, the protein spread evenly around the inner membrane. But when the cells were forced to rely on extracellular electron transfer under oxygen-free conditions, CymA unexpectedly gathered into distinct bright spots, or “puncta,” often near the cell poles. This reorganization happened whether electrons were flowing out of the cell to minerals or into the cell from metal iron or electrodes. Importantly, the total amount of CymA did not change much; what changed was its spatial arrangement.
Dynamic Clusters Enable Stronger Electron Flow
Looking at many cells over time, the team found that only a fraction of cells formed CymA clusters at any given moment, and that the timing varied from cell to cell. In individual cells, however, the transition from a uniform distribution to puncta was surprisingly fast once it began, suggesting a sudden internal reorganization after a variable delay. To test whether these puncta actually matter for electrical performance, the researchers combined light-activated electrodes with microscopy and measured tiny photocurrents from single cells. Cells that had formed CymA puncta showed significantly stronger electron uptake from the electrode than those without puncta, even though both types contained similar amounts of CymA. This indicates that clustering, not mere protein abundance, is what enables efficient extracellular electron transfer.
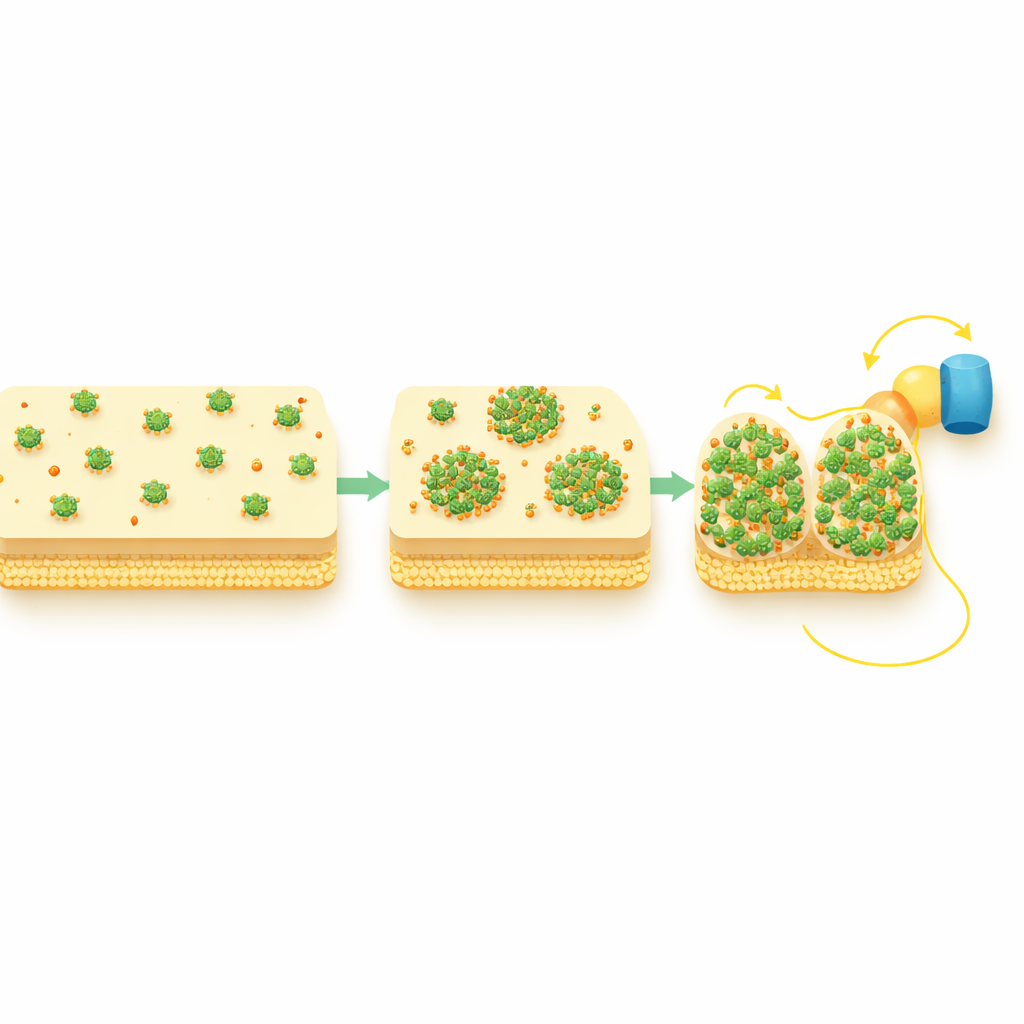
Building Shorter Pathways Across the Cell Envelope
The study also followed two periplasmic shuttle proteins, STC and FccA, which directly exchange electrons with CymA and pass them on toward the outer membrane. Under oxygen-rich conditions these shuttles were spread evenly, but during active extracellular electron transfer they also adopted punctate patterns and spatially overlapped with CymA clusters. Genetic tests showed that CymA reorganizes first and then draws in these partners, not the other way around. In other words, the CymA puncta act as hubs that gather the downstream shuttles into compact, high-traffic junctions, effectively shortening and strengthening the electron pathway from the cell’s interior to its surface.
Liquid-Like Condensates Driven by Membrane Chemistry
By tracking individual CymA molecules, the researchers discovered that proteins within puncta remained mobile and could move in and out of clusters, and that the clusters themselves could appear and disappear depending on growth conditions. These are hallmarks of “biomolecular condensates,” liquid-like droplets formed by collections of proteins and other molecules rather than rigid complexes or damaged aggregates. Measurements of a small membrane molecule called menaquinone, which ferries electrons inside the inner membrane, showed that its levels rise in step with the appearance of CymA puncta. The authors propose that regions of the membrane enriched in menaquinone act as scaffolds that recruit CymA into condensate-like domains, especially near the cell poles.
Why This Matters for Nature and Technology
Overall, the study shows that Shewanella does not rely only on which proteins it makes to control extracellular electron transfer; it also depends on when and where these proteins gather into dynamic clusters. CymA condensates, likely organized by local membrane chemistry, draw in partner proteins to form temporary “electrical hot spots” that boost the flow of electrons across the cell envelope. For a lay reader, this means that even bacteria use a kind of self-assembled circuitry, rearranging their internal parts on demand to plug into rocks, metals, or electrodes. Understanding and eventually steering this kind of protein self-organization could help scientists design better microbial fuel cells, more efficient bioelectrochemical factories, and new strategies for cleaning up metal and pollutant contamination.
Citation: Park, Y., Yan, T., Zhao, Z. et al. Spatiotemporal organization of membrane protein controls bacterial extracellular electron transfer. Nat Commun 17, 2855 (2026). https://doi.org/10.1038/s41467-026-69655-y
Keywords: extracellular electron transfer, Shewanella, bacterial bioelectricity, biomolecular condensates, microbial fuel cells