Clear Sky Science · en
Clustering and a conformational switch drive activation of the mammalian receptor tyrosine kinase ROS1
Why this matters for health and cancer
Many cancers hijack the same communication systems that healthy cells use to grow and develop. One such system centers on a surface protein called ROS1, which helps control tissue development but can also drive tumors when misused. Until now, scientists did not know what naturally switches ROS1 on, or how to shut it down with modern drugs. This study uncovers both the long-sought trigger for ROS1 and a detailed picture of how it works, opening new paths for treating cancers that depend on this molecule.
A long‑mysterious cell antenna
ROS1 belongs to a large family of cell “antennas” known as receptor tyrosine kinases. These proteins span the cell membrane: an outer portion senses signals, while an inner portion relays messages that tell the cell to grow, divide, or specialize. ROS1 is unusual within this family. It has one of the largest and most elaborate outer regions, and for decades no one knew what natural signal it recognized. Yet ROS1 shows up in many tumors, either as abnormal gene fusions that are already drug targets, or as full-length protein found at high levels in brain, lung, oral, and some breast cancers. Because existing drugs mainly hit the inner enzyme portion, researchers hoped that understanding the outer portion could inspire antibody therapies that are more selective and durable.
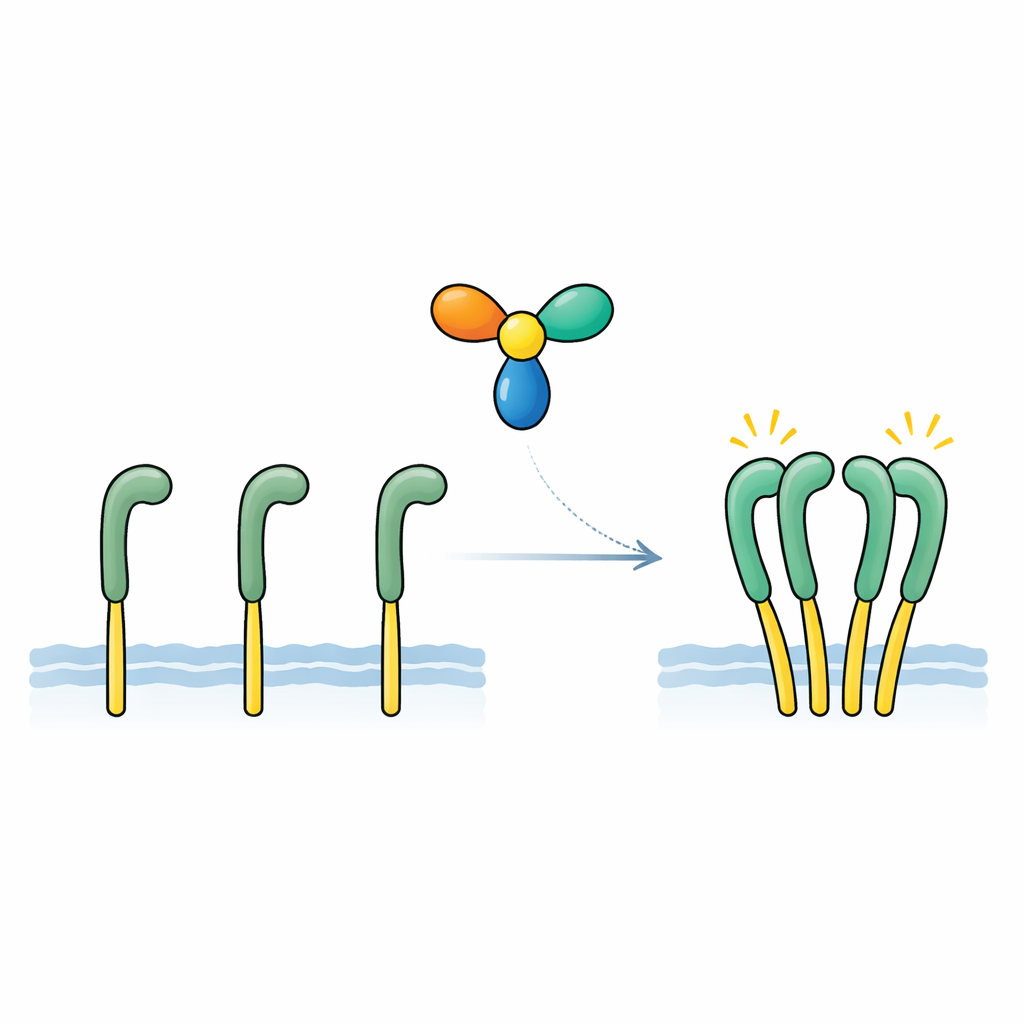
Finding and proving the missing signal
The team focused on a secreted protein called NELL2, previously linked to nerve development and to ROS1’s role in male fertility. Using sensitive binding measurements, they showed that NELL2 attaches directly and tightly to the outer portion of ROS1, with an affinity similar to classic growth factors and their receptors. In engineered cells carrying a ROS1-based test receptor, adding NELL2 strongly boosted the chemical “on” marks on the inner side of the receptor and activated downstream signaling pathways. A modified version of NELL2 that had been forced into a single-unit form could still bind ROS1 but no longer turned it on. This revealed two key points: NELL2 is a genuine activating signal for ROS1, and its ability to form a multi-unit complex is essential for switching the receptor to an active state.
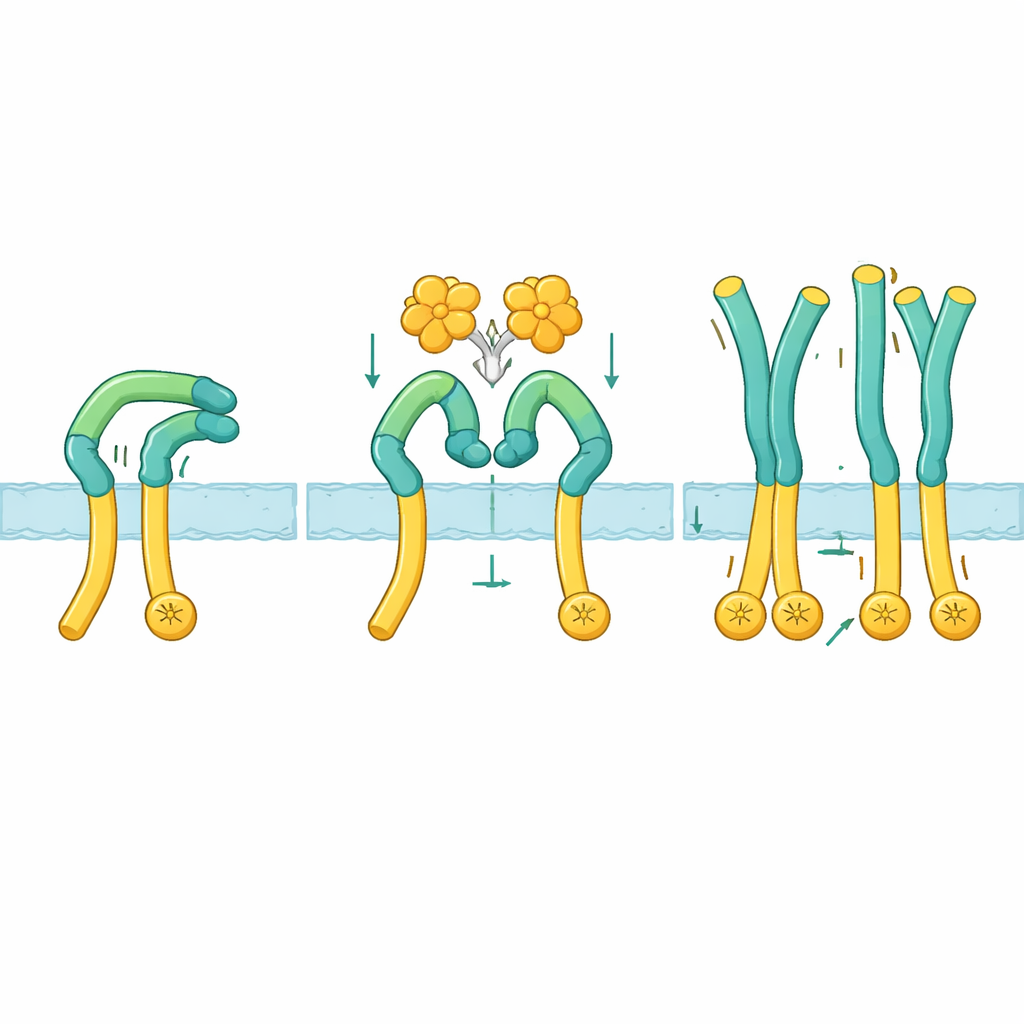
A shape shift that unlocks activity
To see how this activation actually works, the researchers used cryo–electron microscopy to visualize ROS1’s outer region alone and in complex with NELL2. In the resting state, ROS1 folds back on itself: an upper “arm” portion bends down and clamps against a long “leg” that leads toward the cell membrane. This bent pose holds the lower part rigid and appears to keep the inner enzyme ends too far apart to communicate. When NELL2 binds, it grips three different spots across several domains on the ROS1 arm. This binding not only clusters multiple ROS1 molecules together, it also pries the arm away from the leg and rotates it by about 130 degrees into a more upright pose. Once this clamp is released, the leg segment becomes flexible, allowing the membrane-proximal regions, and thus the inner enzyme parts, to move close enough to activate each other.
Turning structural insight into drug candidates
Armed with atomic-level maps of these shapes, the authors then used antibody-display libraries to find lab-made antibodies that stick to ROS1’s outer region. They uncovered two distinct types. One antibody binds precisely where NELL2 first docks, physically blocking the ligand and preventing activation. A second antibody wedges into the cleft between the arm and the central hub of ROS1, stabilizing the original bent, clamped conformation. Both antibodies potently shut down ROS1-driven signaling in cells, even though, in principle, their two arms could bring receptors together. This shows that simple clustering is not enough: activation absolutely requires the NELL2-induced shape change that frees the leg and lets the inner enzymes meet.
What this means for future cancer therapies
This work reveals that ROS1 is controlled by a two-step safety system: receptors must be pulled together by a multi-unit signal and must also undergo a major shape change that releases an internal brake. NELL2 provides both functions, while the newly discovered antibodies selectively interfere with either the initial grip or the unlocking motion. For non-expert readers, the takeaway is that scientists have finally identified a natural switch for ROS1 and have mapped, almost atom by atom, how it turns an off-state into an on-state. These insights lay the groundwork for next-generation biologic drugs that could specifically silence ROS1 in tumors, while leaving other cellular switches undisturbed.
Citation: Li, H., Zhang, J., Li, T. et al. Clustering and a conformational switch drive activation of the mammalian receptor tyrosine kinase ROS1. Nat Commun 17, 3657 (2026). https://doi.org/10.1038/s41467-026-69630-7
Keywords: ROS1 receptor, receptor tyrosine kinase, cancer signaling, NELL2 ligand, therapeutic antibodies