Clear Sky Science · en
H pilin cyclisation and pilus biogenesis are promiscuous but electrostatic perturbations impair conjugation efficiency
Why tiny bacterial cables matter
Bacteria constantly swap DNA, sharing traits such as antibiotic resistance that can turn harmless microbes into difficult-to-treat threats. One major route for this exchange is conjugation, where a donor bacterium uses a long, hair‑thin surface cable called a pilus to connect to a neighbor and pass along plasmid DNA. This study asks a surprisingly subtle question with big implications: how do the exact atoms on the surface of that pilus, and the lipids in the recipient’s outer membrane, control whether DNA transfer succeeds or fails?
The secret handshake between bacterial cells
During conjugation, plasmid DNA travels from a donor cell to a recipient in a contact‑dependent process. Donor cells build a special pilus, a flexible helical tube made of repeating pilin protein units. The work focuses on the H‑pilus encoded by the IncHI1 plasmid R27, a plasmid family known for spreading antibiotic resistance. Earlier cryo‑electron microscopy revealed that its pilin, TrhA, is unusual: instead of having an open-ended chain, the protein is “tied into a ring” by joining its beginning (Gly1) and end (Asp69). This cyclisation is essential for forming a functional pilus capable of transferring DNA.
Highly conserved protein tips, surprisingly tolerant chemistry
By mining a large plasmid database, the authors examined 147 TrhA sequences from related plasmids. Although many positions varied, the two residues that close the ring—Gly1 and Asp69—were absolutely conserved, hinting at strong evolutionary pressure to keep them. The team then systematically changed these two positions in the lab, creating several mutant pilins. High‑resolution cryo‑EM showed that even quite different amino acids at Asp69 (such as Asn, Ala, Gly, or Arg) still allowed TrhA to cyclise and assemble into pili that were structurally almost indistinguishable from the wild type. In other words, the chemistry of ring‑formation and pilus building was surprisingly promiscuous.
When charge on the cable blocks the connection
Despite their normal appearance, the mutant pili behaved very differently in conjugation tests with a range of Gram‑negative recipients. When Asp69 was replaced by neutral amino acids (Asn, Ala, Gly), plasmids still transferred well into E. coli and Klebsiella pneumoniae. In some other species, subtle trends emerged: larger side chains generally supported slightly higher transfer efficiency than smaller ones. A very different picture emerged when Asp69 or Gly1 were replaced by positively charged amino acids such as arginine or lysine. These changes flipped the pilus exterior from negatively charged to strongly positive. Although the mutant pili still formed, conjugation into normal recipients dropped by up to a million‑fold compared with the wild‑type pilus.
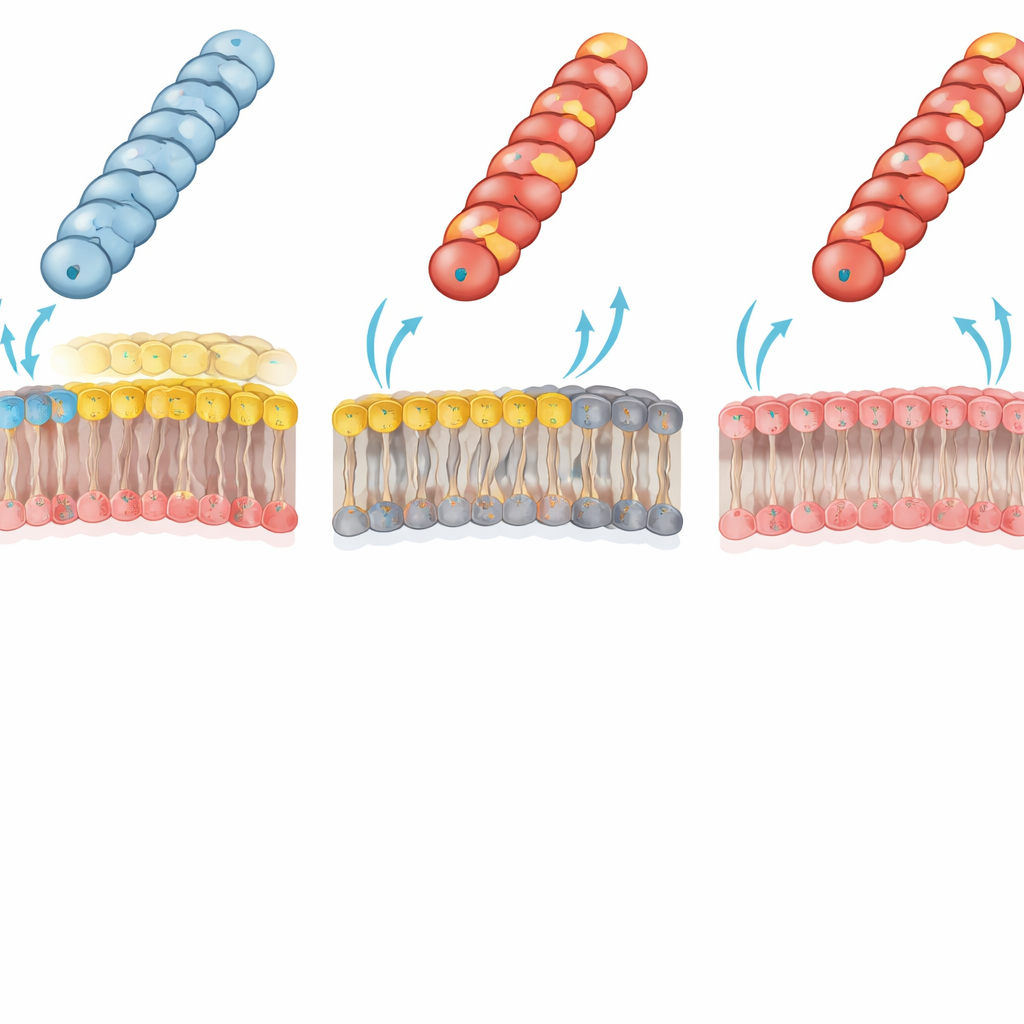
Membrane lipids decide who can receive DNA
The authors traced this dramatic failure to an electrostatic clash between the pilus and the recipient outer membrane. In typical Gram‑negative bacteria, the inner leaflet of the outer membrane contains a major “zwitterionic” phospholipid, phosphatidylethanolamine (PE), which carries both a positive and a negative group. The team used a specially engineered E. coli strain lacking PE; its outer membrane inner leaflet is instead built entirely from negatively charged lipids. In this PE‑deficient recipient, the previously crippled positively charged pili (with Gly1Lys, Asp69Lys or Asp69Arg) now transferred plasmids almost as efficiently as the wild type. This reversal strongly supports the idea that an unfavorable charge–charge interaction between a positively charged pilus surface and the PE‑rich membrane normally prevents the pilus tip from adopting the right contact geometry for efficient DNA transfer.
How electrostatic fine‑tuning shapes antibiotic resistance spread
Taken together, the results reveal a clear division of labor. The chemical machinery that cuts and rejoins the pilin chain to form a ring is quite flexible—many different end residues can still be stitched together and assembled into a normal‑looking pilus. But successful conjugation is exquisitely sensitive to the size and, especially, the charge of these exposed residues, because they shape the pilus surface potential that must match the lipid environment of the recipient membrane. This electrostatic “tuning” helps explain why Gly1 and Asp69 are so strictly conserved in nature. It also suggests that changes in membrane lipid composition—driven by environmental stress or host conditions—could modulate how readily bacteria exchange plasmids carrying antibiotic resistance genes. In effect, both partners in the bacterial “handshake” co‑evolve their surfaces to balance the benefits and risks of gene sharing.
Citation: He, S., Ishimoto, N., Wong, J.L.C. et al. H pilin cyclisation and pilus biogenesis are promiscuous but electrostatic perturbations impair conjugation efficiency. Nat Commun 17, 2888 (2026). https://doi.org/10.1038/s41467-026-69599-3
Keywords: bacterial conjugation, antibiotic resistance, pilus structure, membrane lipids, electrostatic interactions