Clear Sky Science · en
GLP-1 activates KATP channels in coronary pericytes as the effector of brain-gut-heart signalling mediating cardioprotection
Why protecting tiny heart vessels matters
When a blocked heart artery is reopened after a heart attack, many people assume blood flow is fully restored. Yet in up to half of patients, the smallest blood vessels in the heart stay clamped shut, so parts of the heart muscle remain starved of blood. This hidden problem, called “no‑reflow,” greatly increases the risk of heart failure and death. The study summarized here uncovers a brain–gut–heart signalling pathway that can open these tiny vessels from within and suggests new ways to protect the heart using drugs already known from diabetes treatment.
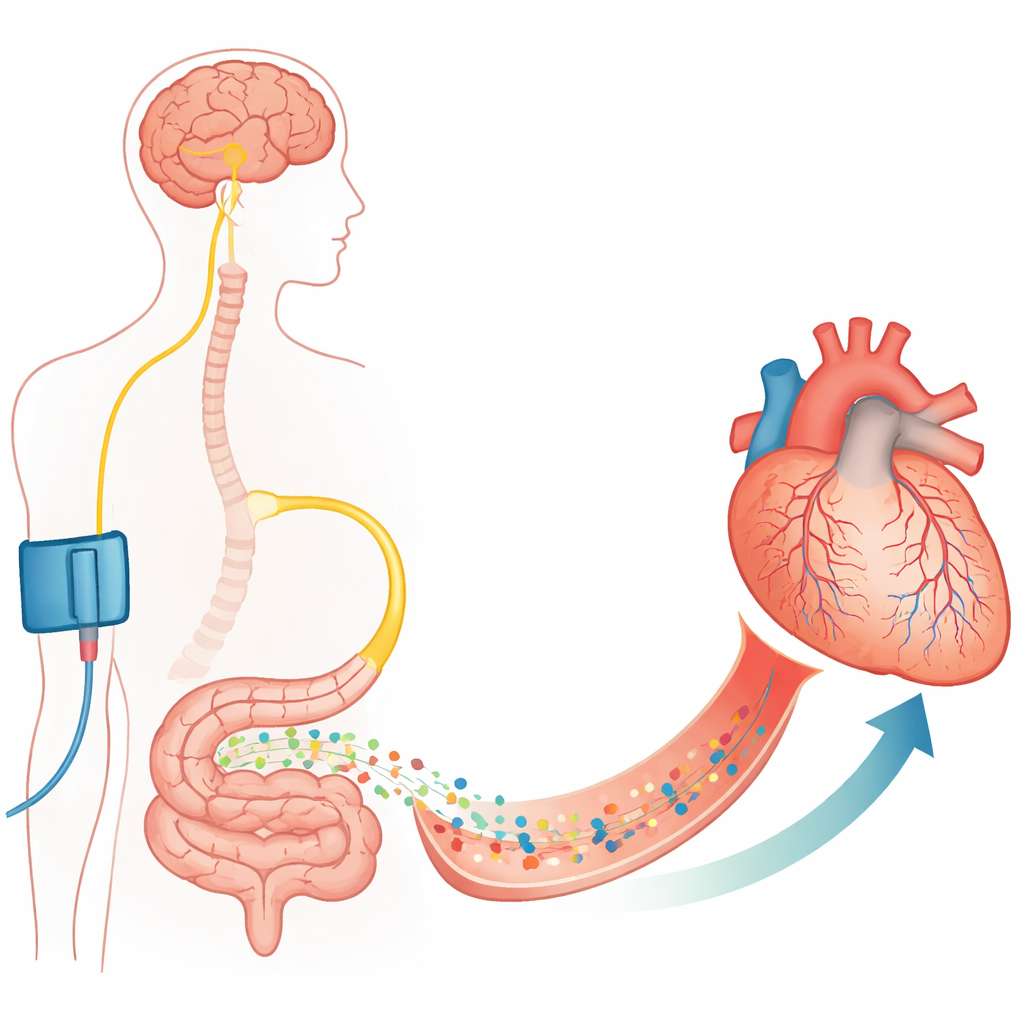
From large arteries to tiny gatekeeper cells
Opening a clogged coronary artery with a stent is lifesaving but not always sufficient. The authors focus on capillaries, the hair‑thin vessels that actually deliver oxygen to heart muscle. Wrapped around these capillaries are support cells called pericytes, which can squeeze or relax the vessel wall. Earlier work showed that during and after a period without blood, these pericytes tighten and sometimes die in a contracted state, permanently narrowing capillaries and causing no‑reflow. This study confirms in rat and mouse hearts that after a controlled blockage and reopening of a coronary artery, many capillaries remain constricted at pericyte sites and a large fraction no longer carry blood, even though the main artery is patent.
A hormone from the gut that talks to heart capillaries
The researchers then asked whether the body has a built‑in way to relax these pericytes. They examined a phenomenon called remote ischaemic preconditioning, where briefly cutting off blood flow to a limb (for example with a cuff) somehow protects the heart from a later blockage. Previous work pointed to the vagus nerve and the gut hormone GLP‑1 as key messengers. Using fluorescent probes and antibodies, the team showed that GLP‑1 receptors are present on coronary pericytes in mice and in cultured human heart pericytes. In living rats, briefly making the hindlimbs ischaemic before a coronary blockage sharply reduced capillary blockages and restored perfused blood volume in the endangered heart region—but only if GLP‑1 receptors were active. Blocking these receptors erased the benefit and brought no‑reflow back.
How GLP‑1 makes capillaries widen again
To dissect the mechanism, the authors used an isolated, pressurized piece of mouse heart where they could watch single capillaries and pericytes under a microscope. When they removed oxygen and glucose from the fluid bathing the tissue, pericytes constricted and capillary diameters shrank. Adding a GLP‑1‑like drug (Exendin‑4) while low‑oxygen conditions continued caused the pericytes to relax and the capillaries to re‑expand almost to their original width. This effect vanished if they blocked a specific type of ion channel known as an ATP‑sensitive potassium (KATP) channel or genetically removed a key KATP component only in pericytes. Similarly, a direct KATP channel opener could reverse drug‑induced constriction in normal mice but not in pericyte KATP knockouts. Mice lacking pericyte KATP channels also lost the heart‑protective benefit of remote preconditioning in whole‑animal experiments, firmly tying these channels to the protective pathway.
Fine‑tuning the pathway with cellular energy and signaling
The story goes deeper into how these channels are controlled. The team showed that an enzyme sensing cellular energy status, AMP‑activated protein kinase (AMPK), is crucial. Blocking AMPK prevented GLP‑1 from reopening constricted capillaries and shifted KATP channels away from the cell surface, suggesting that AMPK helps move these channels into position on the pericyte membrane where they can function. They also found that blocking nitric oxide production or muscarinic (acetylcholine) receptors did not worsen constriction by itself, but specifically blocked the dilation triggered by GLP‑1. This fits a model where nitric oxide and acetylcholine act upstream by stimulating AMPK, which in turn promotes KATP channel trafficking and enables GLP‑1 to hyperpolarize and relax pericytes.
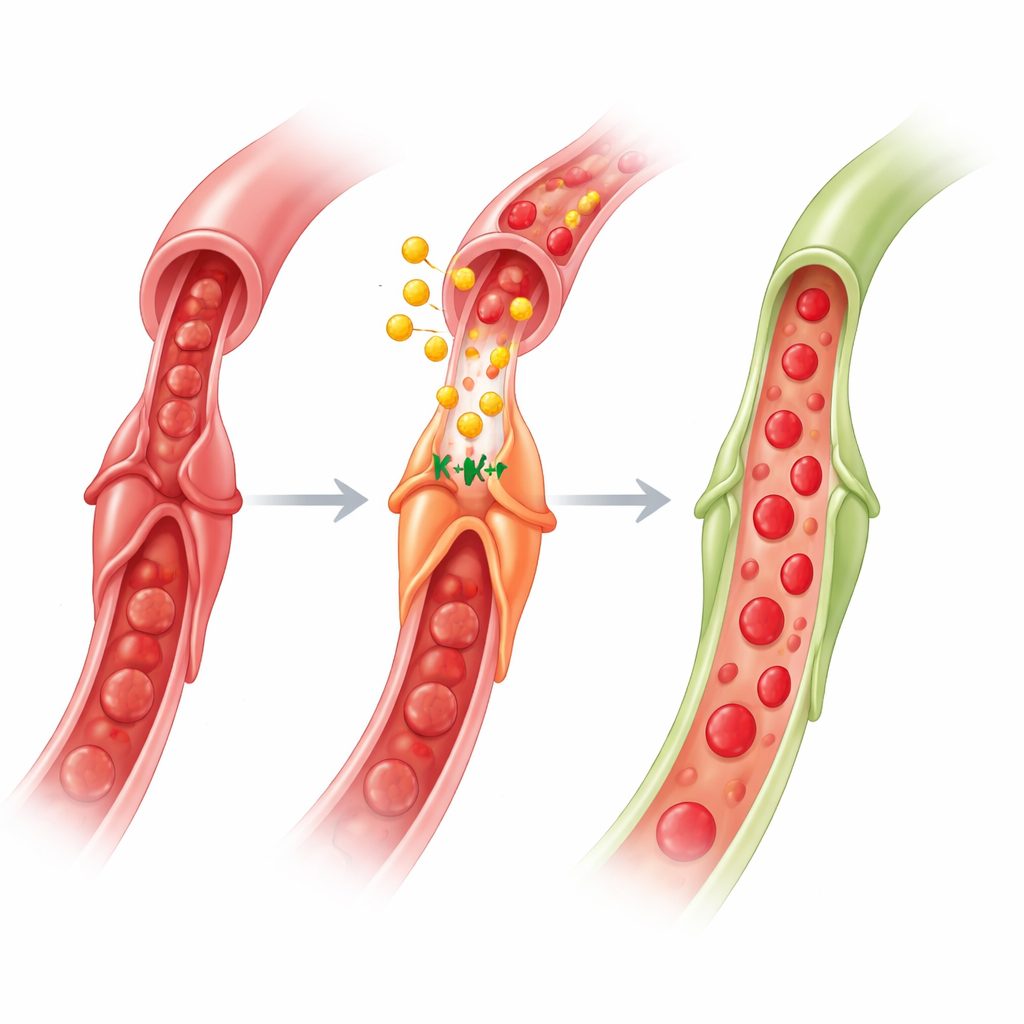
What this means for future heart treatments
Taken together, the work reveals a brain–gut–heart circuit: a brief ischaemic signal in a limb activates nerves to the gut, gut cells release GLP‑1 into the bloodstream, and GLP‑1 travels to the heart to act on pericytes. By engaging KATP channels—with help from AMPK, nitric oxide and acetylcholine—GLP‑1 makes pericytes let go of capillaries, restoring flow in the microcirculation after an artery is reopened. For non‑specialists, the key message is that protecting the heart is not only about unblocking big arteries; it is also about persuading tiny gatekeeper cells to open up. Because GLP‑1‑based drugs are already used widely for diabetes and obesity and have shown cardiovascular benefits, refining them to target pericyte function during and after heart attacks could offer a new way to reduce no‑reflow and improve recovery.
Citation: Mastitskaya, S., de Freitas, F.S.S., Evans, L.E. et al. GLP-1 activates KATP channels in coronary pericytes as the effector of brain-gut-heart signalling mediating cardioprotection. Nat Commun 17, 2773 (2026). https://doi.org/10.1038/s41467-026-69555-1
Keywords: heart attack, microcirculation, GLP-1, pericytes, cardioprotection