Clear Sky Science · en
The clinical missense variant E282K in PPP3CA/calcineurin shifts substrate dephosphorylation by altering active site recruitment
When a Tiny Change in a Brain Enzyme Causes Big Trouble
Our brain cells constantly turn signals on and off by adding and removing tiny chemical tags called phosphates. A key enzyme called calcineurin is responsible for removing many of these tags, and inherited or new mutations in its gene, PPP3CA, have been linked to a severe childhood disorder with epilepsy and developmental delay. This study asks a simple but important question: how can changing just one building block in calcineurin so dramatically disturb brain development?
How Cells Use Chemical Switches
Proteins inside cells act like workers on an assembly line, and phosphate tags are the on/off switches that control them. Kinases attach phosphate groups, while phosphatases remove them. Calcineurin is a phosphatase that responds to calcium signals and is especially active in the brain, where it helps control development, learning, and memory. Because calcineurin touches hundreds to thousands of different protein targets, even small errors in which proteins it acts on can ripple through many signaling pathways, contributing to disorders such as Alzheimer’s, Parkinson’s disease, and the rare developmental condition known as PPP3CA-related DEE91.
A Hidden Docking Pocket Near the Working Site
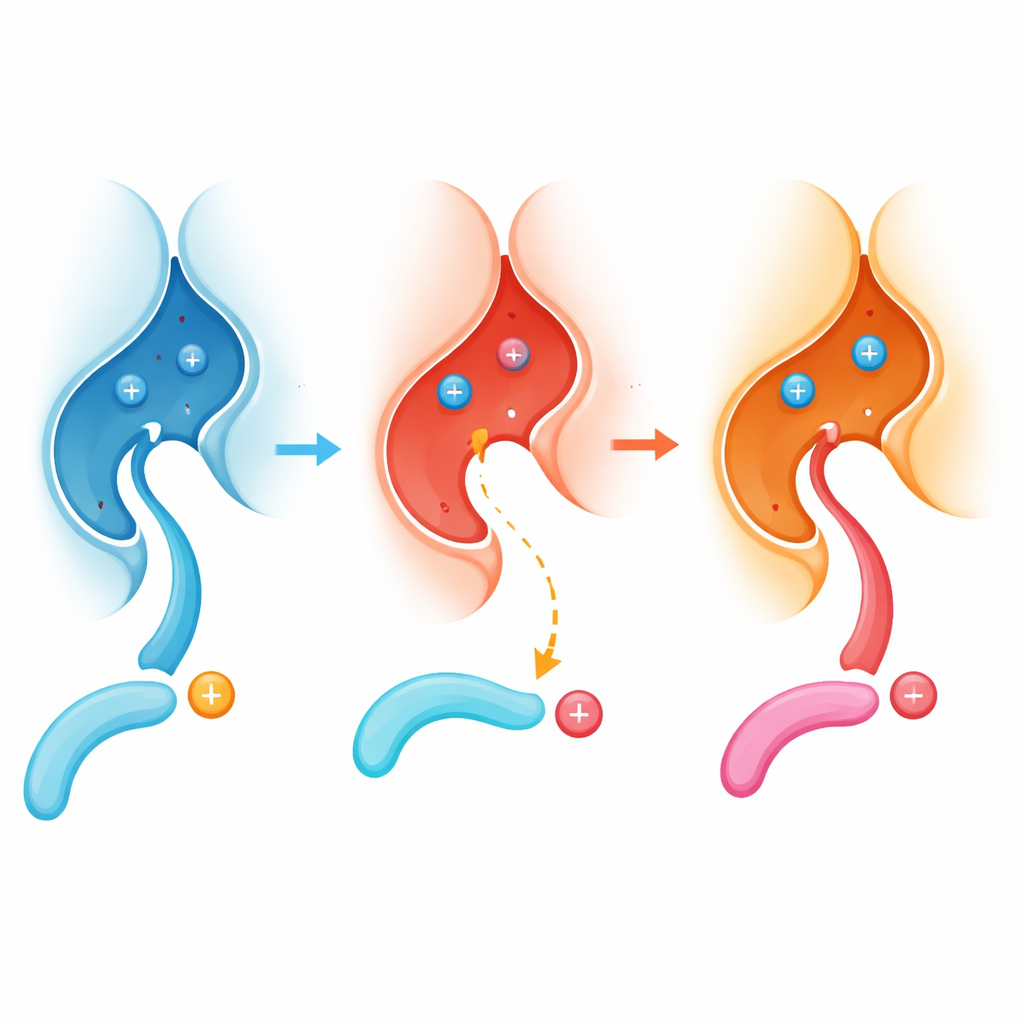
The researchers focused on how calcineurin recognizes specific spots on its protein targets. They discovered a previously unrecognized “recruitment pocket” right next to the enzyme’s working site. Many target proteins that are first modified by certain kinases carry a positively charged amino acid, arginine, immediately before the phosphate tag. Using structural biology, binding measurements, and test-tube reactions, the team showed that this arginine nestles into a negatively charged pocket built around a calcineurin residue called E282, helping pull the tagged site into place so the enzyme can efficiently remove the phosphate.
What the E282K Mutation Really Does
In children with PPP3CA-related DEE91, the most common change in calcineurin swaps E282 for a different amino acid, lysine, creating the E282K variant. Surprisingly, crystal structures revealed that the overall shape of the enzyme remains almost unchanged and its other docking sites still work. The crucial difference lies in the local chemistry: the once acidic, open pocket around E282 becomes more basic and partly blocked by a new internal salt bridge. As a result, peptides that rely on an arginine fitting into this pocket now bind poorly and are dephosphorylated much less efficiently.
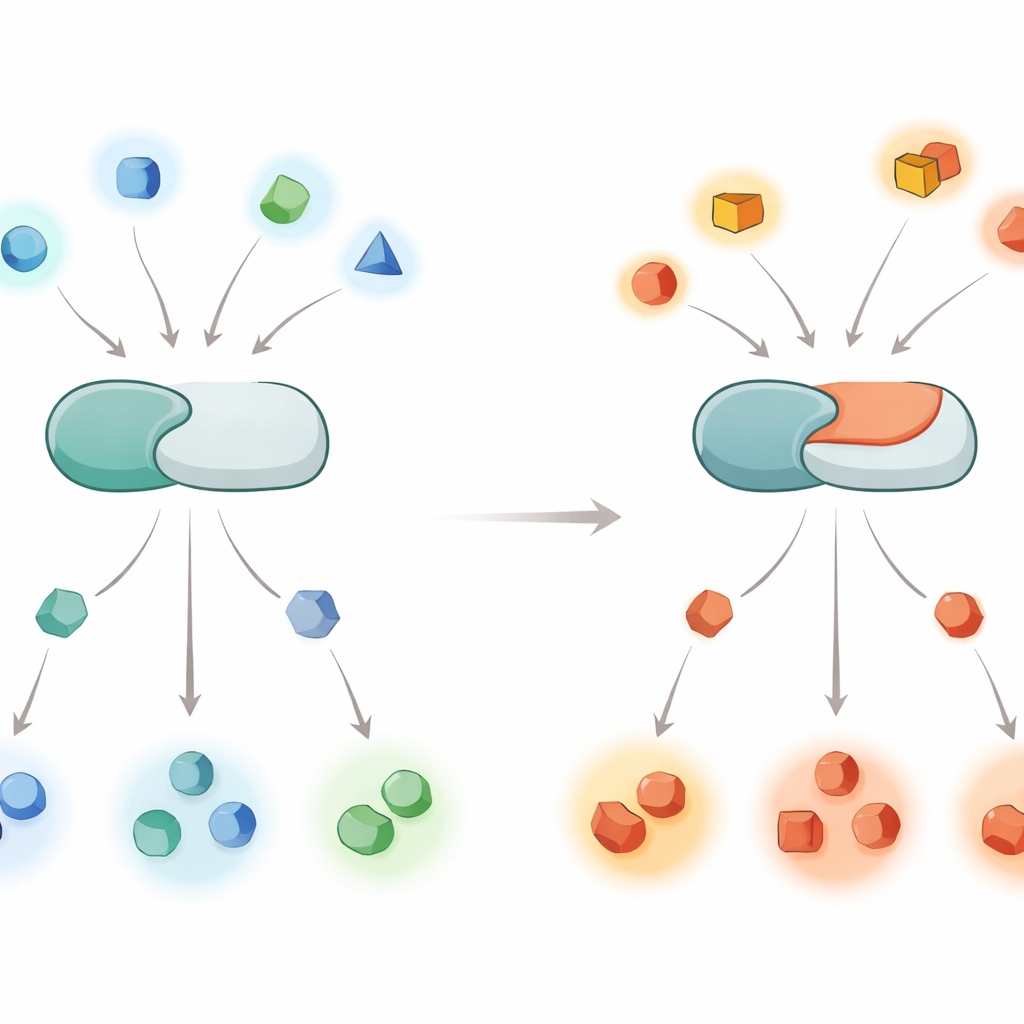
A Global Shift in Who Gets Switched Off
To see how far this effect reaches, the authors examined thousands of phosphopeptides derived from human cells and compared how normal and mutant calcineurin acted on them over time. Normal calcineurin strongly favored sites with positively charged residues (such as arginine or lysine) just before the phosphate. In contrast, the E282K version had lost much of this preference and instead more readily processed sites with negatively charged residues in that position. Similar patterns appeared when the team expressed the two enzyme forms in living cell lines, including a neuron-like line, and used mass spectrometry to catalog which protein sites were more or less dephosphorylated. The mutation effectively retunes the enzyme from liking “basic” sites to favoring “acidic” ones.
Why This Matters for Children with DEE91
The work shows that the E282K mutation does not simply weaken calcineurin or switch it off. Instead, it rewires which protein switches the enzyme can flip, shifting signaling networks throughout the cell. In growing and developing brain cells, this broad misdirection of signals—away from their normal arginine-rich targets and toward different sites—likely disturbs the finely balanced programs that control neuronal growth, connectivity, and electrical activity. By pinpointing this specific recruitment pocket and how E282K alters its charge and access, the study lays a molecular foundation for understanding the disease and suggests new ways to search for therapies that might restore proper substrate selection, even if the mutation itself cannot be reversed.
Citation: Shirakawa, K.T., Parikh, T., Machado, L.E. et al. The clinical missense variant E282K in PPP3CA/calcineurin shifts substrate dephosphorylation by altering active site recruitment. Nat Commun 17, 2837 (2026). https://doi.org/10.1038/s41467-026-69535-5
Keywords: calcineurin, PPP3CA mutation, protein phosphorylation, neurodevelopmental disorders, enzyme specificity