Clear Sky Science · en
GID/CTLH E3 ligase complex control cell fate programs for sexual development of Plasmodium falciparum
Why this malaria study matters
Malaria still kills hundreds of thousands of people each year, and stopping the parasite from moving from humans into mosquitoes is a key goal for ending the disease. This paper uncovers a hidden control system inside the malaria parasite Plasmodium falciparum that decides whether its sexual forms, called gametocytes, mature properly and become infectious to mosquitoes. By explaining how a small group of proteins fine-tunes this decision, the study points to fresh targets for drugs or vaccines that could block transmission and help curb malaria worldwide.
The parasite’s long road to becoming infectious
To spread, P. falciparum must turn some of its blood-stage forms into specialised sexual cells, male and female gametocytes, that develop over 10–12 days. During this slow transformation, the parasite’s shape, metabolism, and interaction with the red blood cell are completely remodelled. Earlier work had revealed how a master switch in the parasite’s DNA turns on the decision to enter the sexual pathway, but much less was known about how the parasite then coordinates the thousands of molecular steps needed to reach fully formed, mosquito-ready stage V gametocytes. The authors suspected that, beyond turning genes on and off, the parasite also relies on controlled protein destruction to keep development on track.
A molecular referee for sexual development
The study focuses on a protein machine called the GID/CTLH E3 ligase complex, here termed PfGID. E3 ligases act like tagging devices, attaching small ubiquitin molecules to chosen proteins to mark them for degradation or adjustment. By scanning the malaria genome and comparing it with related organisms, the researchers identified six components that assemble into a PfGID complex. They showed that these proteins form a conserved scaffold within the parasite and are active throughout both asexual growth and gametocyte development. Strikingly, when any of the PfGID subunits were deleted, the parasite could still multiply in red blood cells, but sexual development stalled around the halfway point. The resulting gametocytes looked stuck at an intermediate shape and completely failed to infect mosquitoes, revealing PfGID as essential for transmission.
How PfGID steers male and female fates
To understand what goes wrong when PfGID is lost, the team examined global RNA and protein patterns in the mutant parasites. They found that key processes such as RNA processing, protein production, and protein recycling were misregulated, and that proteins normally enriched in male and female gametocytes were strongly disturbed. In particular, the abundance of many male-specific proteins dropped, while markers of mature gametocytes were reduced in both sexes. 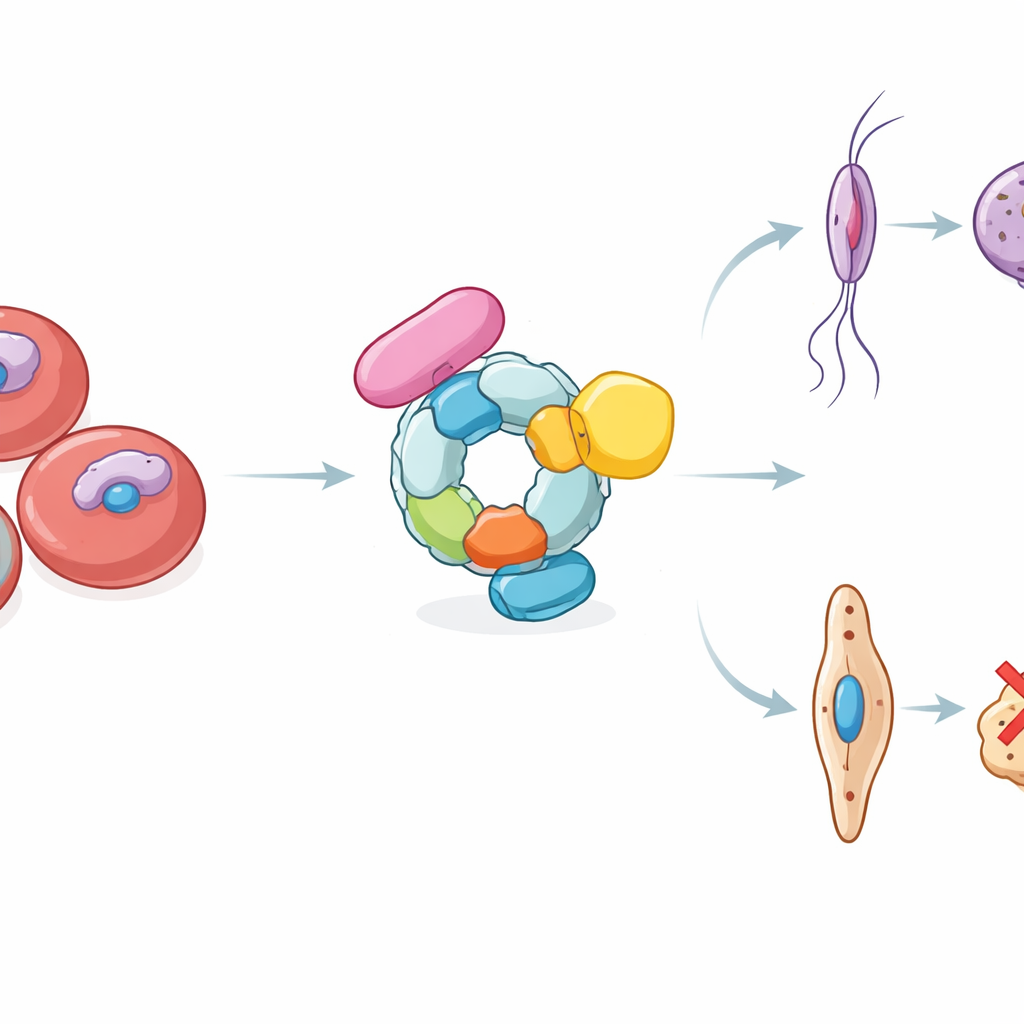
Two intertwined control arms: DNA organisation and message silencing
PfDPL appears to work mainly in shaping the male lineage. It localises to the nucleus of developing gametocytes and associates with histones, chromatin remodellers, nuclear pores and other factors that govern how DNA is packaged and read. When PfDPL is removed, proteins needed for DNA replication, cell division, flagellar assembly and fertilisation in male gametes are sharply reduced, and male exflagellation—the explosive release of sperm-like forms—almost disappears. In contrast, GD1 operates in the cytoplasm, where it clusters into tiny granules known as P-bodies. These structures act as storage and silencing hubs for messenger RNAs. The authors show that GD1 partners with several classic translational repressors and binds hundreds of RNA messages, including many that encode key gametocyte factors. When GD1 levels are either too low or too high—because it is deleted, overexpressed, or insufficiently degraded by PfGID—P-body components become unbalanced, and gametocytes arrest early in development. 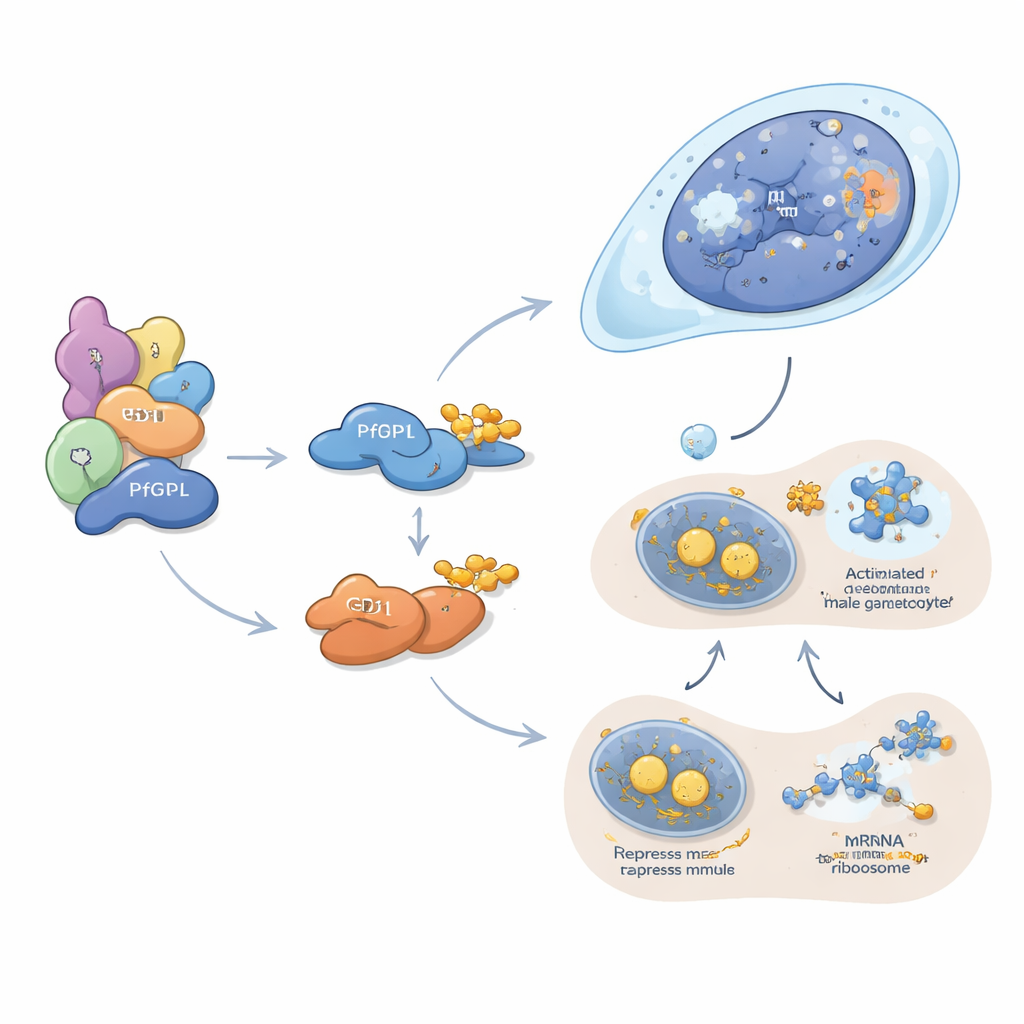
A finely tuned timer for parasite transmission
Together, the findings support a model in which the PfGID complex acts as a molecular referee, tuning the amount and activity of PfDPL and GD1 at precise times. By adding or removing ubiquitin tags on intrinsically flexible regions of these proteins, PfGID can rapidly adjust how strongly PfDPL organises male-specific genes in the nucleus and how tightly GD1 holds key RNA messages in a silent state. If this timing is disturbed, male and female gametocytes fail to mature, and the parasite cannot move to the mosquito. For a lay reader, the takeaway is that malaria parasites rely on a sophisticated protein “quality control” and message-silencing network to become infectious. Targeting PfGID, PfDPL, GD1 or their interactions could therefore provide new ways to freeze the parasite before it ever reaches the mosquito, cutting the chain of transmission.
Citation: Marapana, D.S., Lopaticki, S., Balan, B. et al. GID/CTLH E3 ligase complex control cell fate programs for sexual development of Plasmodium falciparum. Nat Commun 17, 3497 (2026). https://doi.org/10.1038/s41467-026-69183-9
Keywords: malaria transmission, Plasmodium falciparum gametocytes, ubiquitin ligase, RNA translational repression, parasite cell fate