Clear Sky Science · en
The co-delivery of Programmed Death 1 ligands enhances and prolongs rAAV-mediated gene expression in pre-immunized mice
Making Gene Therapy Work for More People
Gene therapies that use harmless viruses to deliver helpful genes are starting to change how we treat inherited diseases. But many people’s immune systems already recognize these viruses, which can cause the treatment to fade or fail. This study explores a way to "calm" the immune response right where the gene therapy is working, so that the treatment can last longer and help patients who have been exposed to these viruses before.
Why the Immune System Fights Helpful Viruses
The gene therapy tools in this study are based on adeno-associated virus (AAV), a tiny virus widely used to shuttle new genes into cells. AAV is generally safe and can keep working for years, but there is a catch: many people have already met natural AAV in the wild. Their immune systems remember the viral shell and can quickly attack treated cells that display pieces of that shell, cutting off gene expression. This is a particular concern for muscle treatments, which often require high doses and are not naturally shielded from immune attack. Current workarounds—such as excluding patients with pre-existing antibodies or giving broad immune-suppressing drugs—can leave patients unprotected against infections and are not ideal long-term solutions.
Borrowing a Natural Brake on Immunity
The researchers turned to a natural "brake" on the immune system called the PD-1 pathway. In healthy tissues, PD-1 and its partners, PD-L1 and PD-L2, help prevent overactive immune attacks that could damage the body itself. Tumors sometimes exploit this pathway to hide from immune cells, but the same mechanism can, in principle, be used to protect healthy cells that have been modified by gene therapy. The team designed AAV vectors that carry both a reporter gene (a harmless secreted enzyme called muSEAP, which can be easily measured in blood) and either PD-L1 or PD-L2, linked by a short self-cleaving peptide that lets cells produce both proteins separately from a single genetic cassette. They first confirmed in cell cultures that the "two-in-one" design worked: both the therapeutic stand-in and the PD-1 ligands were properly produced, biologically active, and able to bind to PD-1.
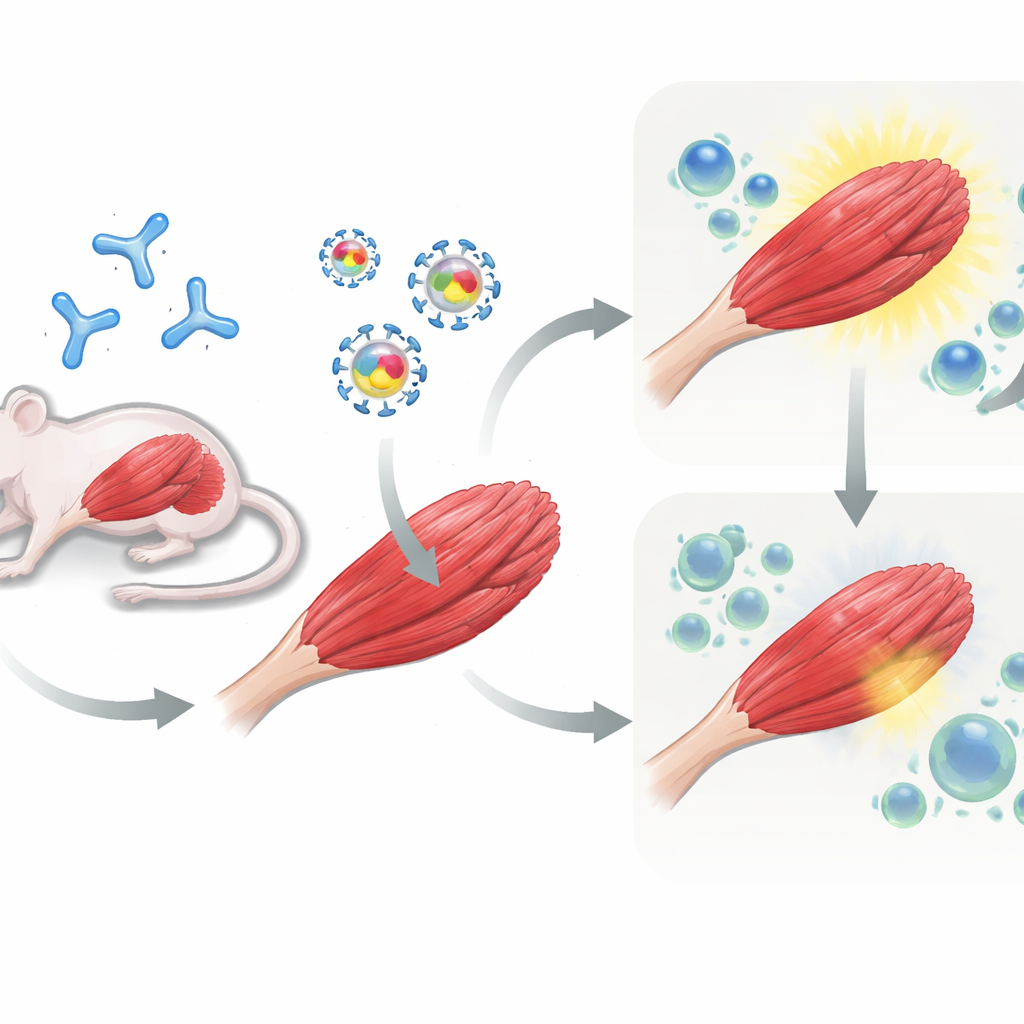
Testing the Strategy in Pre-Exposed Mice
Next, the team moved to mice. Some animals were first given an AAV dose to mimic patients who already harbor immunity to the vector, while others remained naïve. All mice then received gene therapy injected into their leg muscles: either AAV carrying muSEAP alone, or AAV carrying muSEAP plus PD-L1 or PD-L2. Over several weeks, the researchers measured muSEAP levels in the blood as a readout of how well the introduced gene was working. In mice that had been pre-immunized against AAV, co-delivery of PD-L1 was especially powerful: muSEAP levels were much higher and stayed elevated for up to 12 weeks compared with the control vector. PD-L2 also improved expression, though to a lesser and more variable extent. In contrast, in naïve mice that had never seen AAV before, adding PD-L1 or PD-L2 did not produce a lasting advantage and, in some cases, any early gain faded with time.
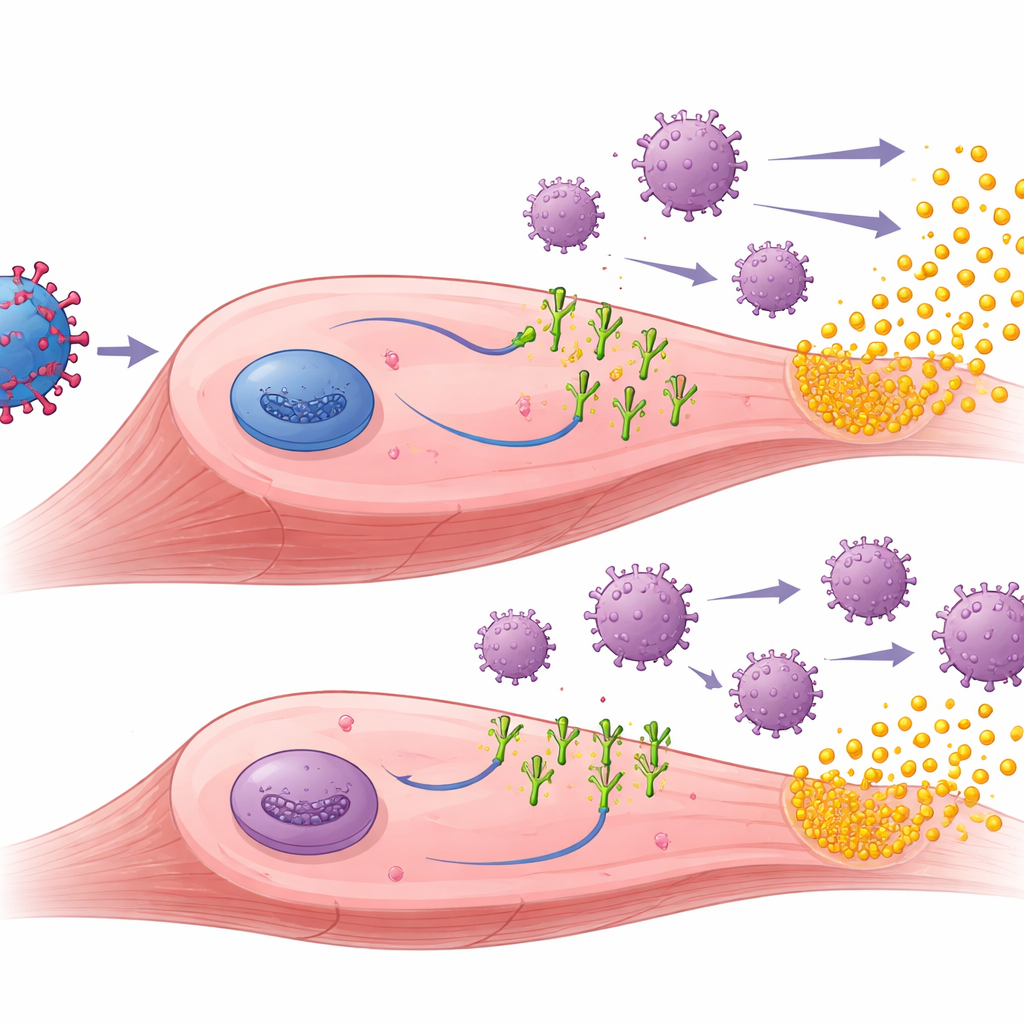
What Happens Inside the Treated Muscle
To understand why the PD-1 ligands helped, the researchers examined the treated muscles. They found that the gene therapy successfully drove production of both muSEAP and the PD-1 ligands in muscle tissue. In the pre-immunized animals, muscles that received the PD-L1 or PD-L2 vectors generally showed fewer invading T cells and other signs of inflammation than muscles treated with muSEAP alone. In these pre-exposed mice, higher muSEAP levels tended to go hand in hand with lower numbers of immune cells, suggesting that local immune dampening allowed the treated cells to survive and keep making the therapeutic protein longer. The team also measured antibodies against AAV and against the human PD-1 ligands themselves. As expected, AAV antibodies rose after pre-immunization and stayed high, but PD-1 ligands did not reduce these anti-vector antibodies. Interestingly, only PD-L1 triggered detectable anti-ligand antibodies, and mainly in naïve mice, hinting at subtle differences in how foreign each ligand appears to the immune system.
What This Could Mean for Future Treatments
In simple terms, this study shows that packaging a local immune "peacekeeper" together with a therapeutic gene can help gene therapy work better in subjects whose immune systems are already primed against the delivery virus. By co-delivering PD-L1 or PD-L2 directly with the transgene in a single AAV vector, the authors achieved stronger and longer-lasting gene expression in pre-immunized mice, while also seeing fewer immune cells attack the treated muscle. Although many questions remain—such as how best to balance benefits with potential risks of dampening immunity, and how these findings will translate to humans—the work points toward a strategy that could open gene therapy to a broader group of patients who otherwise might be excluded because of prior exposure to AAV.
Citation: Käyhty, P., Nieminen, T., Eriksson, R.A.E. et al. The co-delivery of Programmed Death 1 ligands enhances and prolongs rAAV-mediated gene expression in pre-immunized mice. Gene Ther 33, 127–137 (2026). https://doi.org/10.1038/s41434-025-00588-9
Keywords: gene therapy, AAV vectors, immune tolerance, PD-1 checkpoint, skeletal muscle