Clear Sky Science · en
A20 enhances the migration and metastasis of gastric cancer cells by promoting occludin degradation
Why this matters
Gastric cancer, or cancer of the stomach, is one of the world’s deadliest tumors largely because it so often spreads before it is detected. This study uncovers how a single inflammation‑linked protein, called A20, helps stomach cancer cells break away from their neighbors and travel to distant organs. By revealing this hidden escape route, the work points to new ways doctors might someday slow or block the spread of gastric cancer.
A protein at the crossroads of cancer and inflammation
Doctors have long known that chronic inflammation in the stomach, such as that caused by long‑term infection, raises the risk of gastric cancer. A20 is a protein that usually helps keep inflammatory signals in check, but in several cancers it behaves more like a promoter of tumor growth. The authors began by asking whether A20 levels in human stomach tumors are linked to how aggressive the cancer is. Analyzing tumor samples from 122 patients, they found that cancers with high A20 levels were more invasive, more likely to have spread to lymph nodes and distant sites, and were associated with poorer survival. These clinical data suggested that A20 is not just a bystander but may actively drive the worsening of gastric cancer.
How A20 helps cancer cells slip their bonds
For cancer cells to metastasize, they must loosen the tight bonds that normally hold neighboring cells together. A key component of these bonds in many tissues is a protein called occludin, which helps form tight junctions that act like molecular zipper teeth between cells. In gastric cancer cell lines grown in the lab, the researchers showed that boosting A20 levels made cells spread out and migrate faster, whereas reducing A20 had the opposite effect. They noticed that when A20 was high, occludin at the cell surface was lost and shifted into the cell interior. Silencing occludin alone was enough to increase cancer cell movement, indicating that loss of this “zipper” is a crucial step in allowing cells to wander.
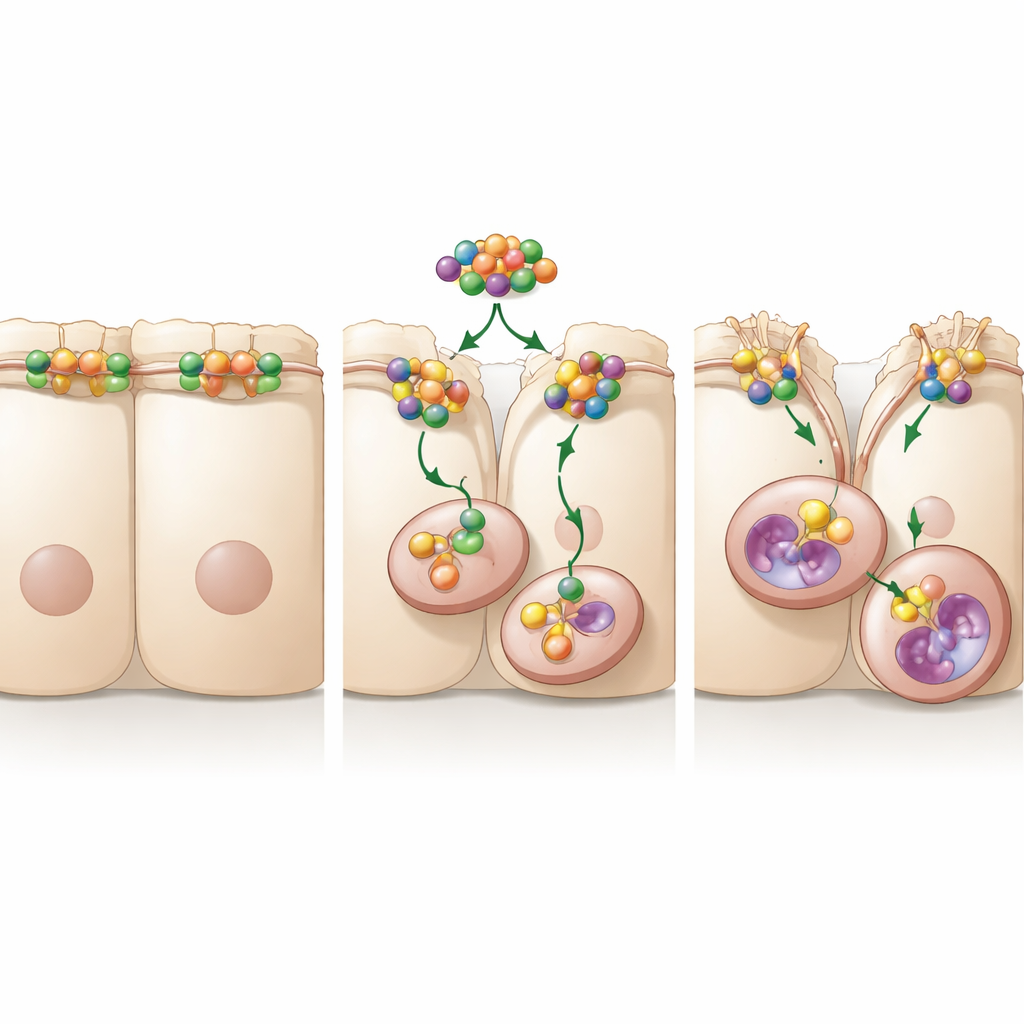
Turning down the cellular “zipper” through internal shredding
The team next asked how A20 causes occludin to disappear. Cells constantly recycle their surface proteins by pulling them inward in small pockets and sending them either to be reused or to be destroyed. The study found that A20 accelerates the journey of occludin from the cell surface into these internal pockets and then on to digestive compartments similar to cellular recycling centers. Blocking the cell’s protein‑shredding machinery in the proteasome did not rescue occludin, but blocking endocytosis or the acidic sacs known as lysosomes did. Under these blocking conditions, cancer cells migrated less, even when A20 was abundant. Together, these experiments show that A20 encourages cancer spread by promoting the internalization and lysosomal breakdown of occludin, weakening the barrier between cells.
A signaling relay that stabilizes a motility switch
Digging deeper, the researchers looked for partners that might allow A20 to control cell movement. They discovered that A20 binds to RhoA, a small molecular switch known to reshape the cell’s inner scaffolding and to loosen junctions between cells. When A20 was present, RhoA levels and activity were higher, and downstream signals that stiffen cellular fibers were prolonged. When RhoA was reduced or chemically blocked, occludin levels went back up and cell migration slowed, even if A20 was high. The study pinpointed a specific region of A20, called the OTU domain, as essential: when this region was mutated, A20 could no longer stabilize RhoA, could not sustain the signaling needed to pull occludin inward, and failed to enhance cell movement. This indicates that A20 acts as a guardian of RhoA, protecting it from being tagged for disposal and thus keeping the motility machinery switched on.
What this could mean for future treatments
In simple terms, this work shows that A20 helps stomach cancer cells escape by protecting a motility switch (RhoA) and triggering the internal shredding of a key cellular zipper (occludin). Tumors with more A20 are more likely to invade deeply and spread, and patients with such tumors fare worse over time. By mapping out this A20–RhoA–occludin pathway, the study highlights several potential intervention points, such as drugs that disrupt A20’s OTU region, reduce RhoA activity, or prevent occludin from being routed to lysosomes. Although such treatments are not yet available and would need to be carefully balanced against A20’s normal roles in controlling inflammation, the findings provide a clearer target for efforts to slow or prevent the deadly spread of gastric cancer.
Citation: Kuo, YT., Wang, HC. & Shan, YS. A20 enhances the migration and metastasis of gastric cancer cells by promoting occludin degradation. Cell Death Discov. 12, 206 (2026). https://doi.org/10.1038/s41420-026-03082-2
Keywords: gastric cancer, metastasis, cell junctions, inflammation, RhoA signaling