Clear Sky Science · en
SMARCA4 promotes lineage plasticity and enzalutamide resistance in prostate cancer by regulating PROX1 via H3K27 acetylation
Why this research matters for prostate cancer patients
Men with advanced prostate cancer are often treated with drugs that shut down male hormones, but many tumors eventually outsmart these therapies and return in a far more aggressive form. This study asks a crucial question: what lets prostate cancer cells change their identity and become both harder to kill and more deadly after treatment with the drug enzalutamide? By uncovering a key molecular switch that drives this shape‑shifting, the work points to new strategies that could delay or prevent this dangerous evolution.
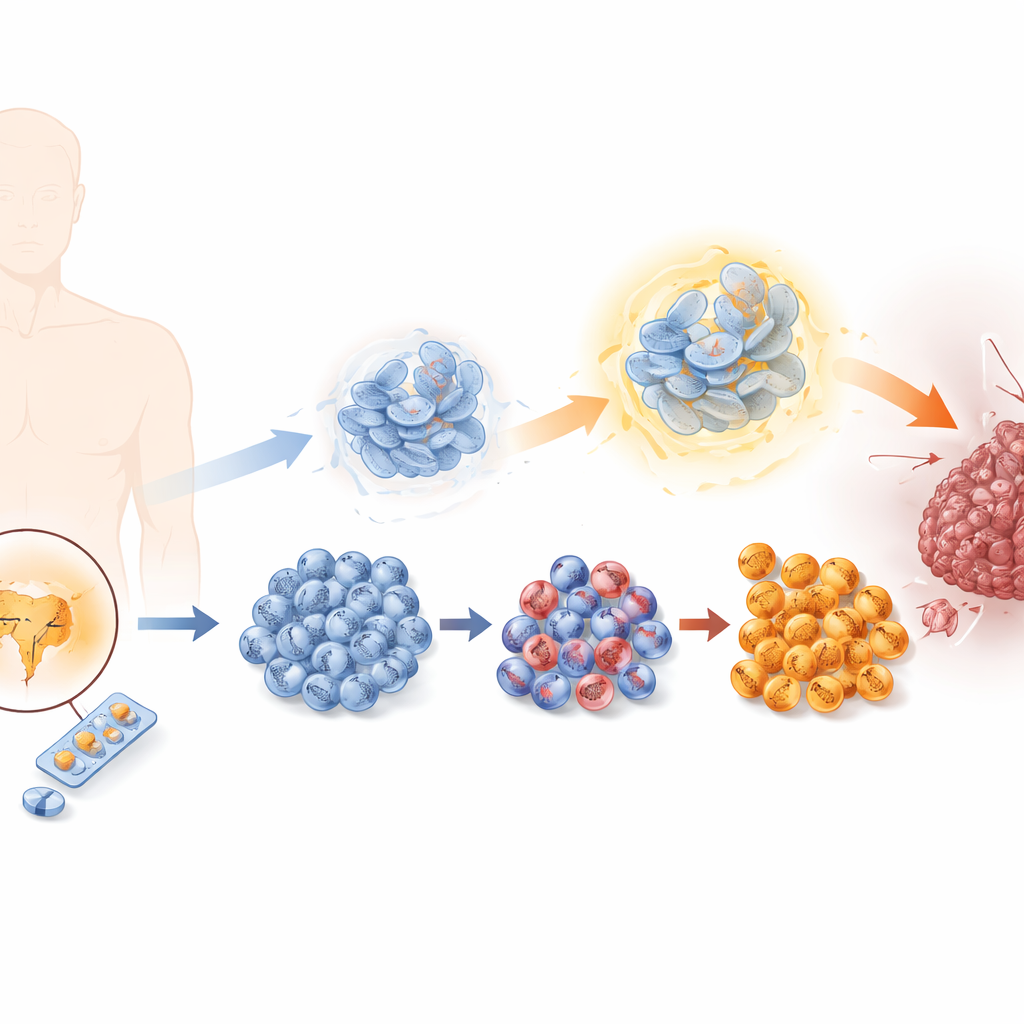
How tumors learn to dodge hormone‑blocking therapy
Prostate cancer cells usually depend on male hormones acting through the androgen receptor. Enzalutamide works by blocking this signal, but over time some cells escape. The researchers recreated this process in the lab by exposing prostate cancer cells to gradually increasing doses of enzalutamide, generating a series of cell lines that ranged from sensitive to highly resistant. As resistance grew, the cells divided faster, migrated more easily, and invaded surrounding tissue more readily, all hallmarks of a more malignant tumor. At the same time, they lost features of typical prostate cells and gained traits of so‑called neuroendocrine cells, a rare but highly aggressive tumor type with few treatment options.
Cell identity changes are written into the DNA’s packaging
To understand how these dangerous traits emerge, the team looked beyond the DNA code itself to the way DNA is packed and unpacked inside the cell nucleus. Using genome‑wide methods that map which regions of DNA are open and which are tightly wound, they found that resistance developed hand‑in‑hand with sweeping changes in chromatin, the protein–DNA material that organizes the genome. Regions controlling androgen‑dependent genes became less accessible, while stretches linked to stem‑like and neuroendocrine programs opened up. These shifts tracked closely with changes in gene activity, revealing a coordinated re‑wiring of the cells’ regulatory circuitry rather than random mutations alone.
A chromatin engine called SMARCA4 pushes tumors toward a deadlier state
Among many altered regulators, one stood out: SMARCA4, a core component of a molecular machine that slides and restructures chromatin. SMARCA4 levels were much higher in the most resistant, neuroendocrine‑like cells and in aggressive patient tumors, and high levels were tied to poorer survival. When the scientists reduced SMARCA4 in resistant cells, growth slowed, migration and invasion dropped, and more cells underwent programmed cell death. Importantly, markers of neuroendocrine identity faded and features of typical prostate cells re‑emerged, both in dishes and in mouse tumors. These findings position SMARCA4 as a central engine that helps tumors both resist treatment and adopt a more dangerous persona.
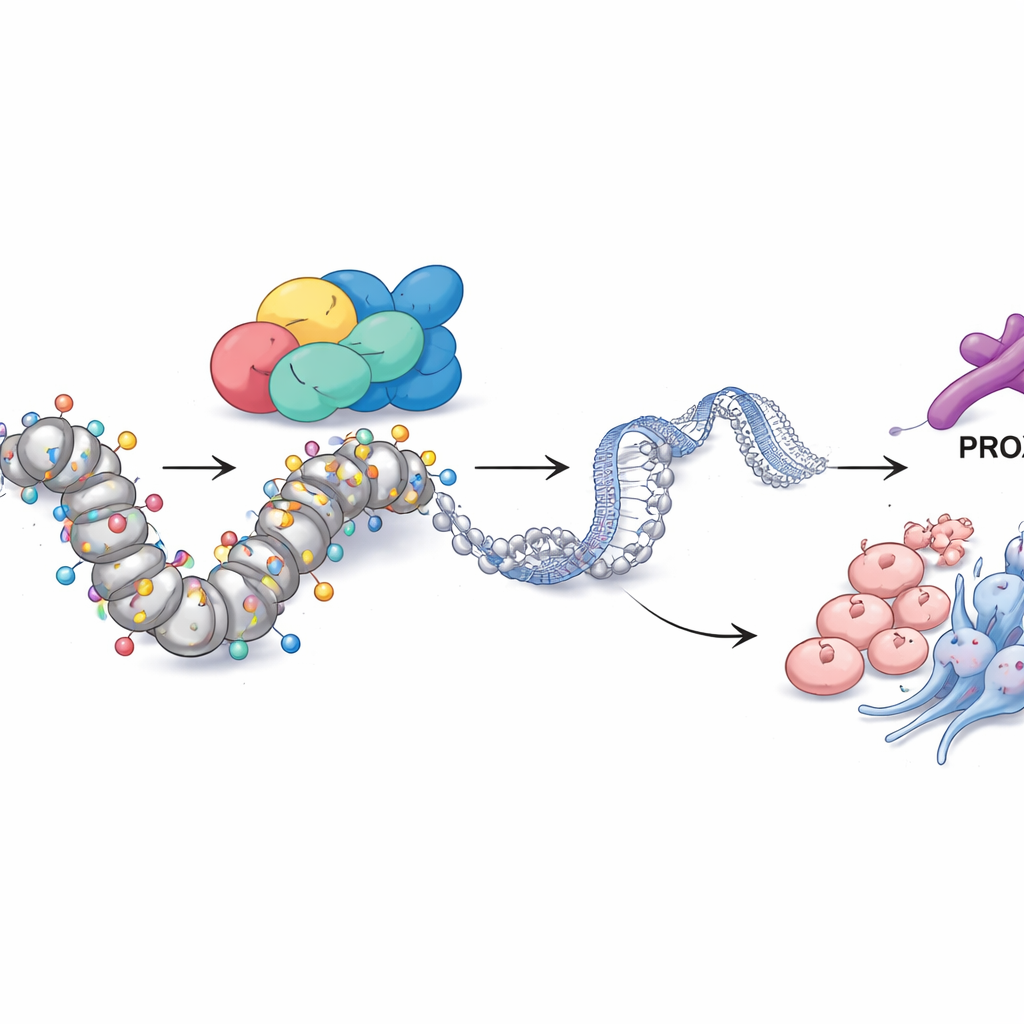
PROX1 and chemical tags connect SMARCA4 to tumor behavior
Digging deeper, the team asked how SMARCA4 exerts such wide‑ranging control. By integrating several layers of genomic data, they identified another protein, the transcription factor PROX1, as a key partner. In resistant cells, PROX1’s control region in the genome was unusually open and decorated with a chemical mark of active DNA, a type of histone acetylation known as H3K27ac. SMARCA4 sat directly at this site, and when SMARCA4 was reduced, both PROX1 levels and H3K27ac at this region fell. Turning down PROX1 alone was enough to cut cell growth, invasion, neuroendocrine markers, and stem‑like properties, showing that PROX1 acts as an important executor of SMARCA4’s program. Remarkably, blocking enzymes that remove acetyl groups (with a histone deacetylase inhibitor) or artificially boosting PROX1 could largely rescue the loss of SMARCA4, underscoring that this pathway runs through acetylation at the PROX1 locus.
What this means for future treatments
Put simply, the study reveals a chain of events: under the pressure of enzalutamide, prostate cancer cells flip on SMARCA4, which reshapes chromatin to boost acetylation and activation of PROX1, and this duo then helps the cells shed their original identity and adopt a resistant, neuroendocrine‑like state. For patients, this suggests that drugs aiming at SMARCA4, the enzymes that place or remove acetyl marks, or PROX1 itself could be combined with standard hormone‑blocking therapies to keep tumors from evolving into this high‑risk form. While these findings still need confirmation in human samples, they outline a concrete epigenetic pathway that clinicians and drug developers can now target to combat one of the most lethal escape routes in prostate cancer.
Citation: Wu, C., Luo, M., Wu, C. et al. SMARCA4 promotes lineage plasticity and enzalutamide resistance in prostate cancer by regulating PROX1 via H3K27 acetylation. Cell Death Discov. 12, 175 (2026). https://doi.org/10.1038/s41420-026-03068-0
Keywords: prostate cancer, drug resistance, epigenetics, neuroendocrine transformation, chromatin remodeling