Clear Sky Science · en
Glycolytic reprogramming mediated by the ADAM12/IGF1 axis promotes ossification of the posterior longitudinal ligament
When a Support Band in the Spine Turns to Bone
Deep inside the spine, a tough band of tissue called the posterior longitudinal ligament helps keep our vertebrae aligned. In some people, especially in East Asia, this flexible strap gradually turns into bone, squeezing the spinal cord and causing pain, weakness, or even paralysis. This condition, known as OPLL, usually requires risky surgery and currently has no drug treatment. The study summarized here asks a simple but powerful question: what if the way these cells use sugar is the hidden engine driving this unwanted bone growth?
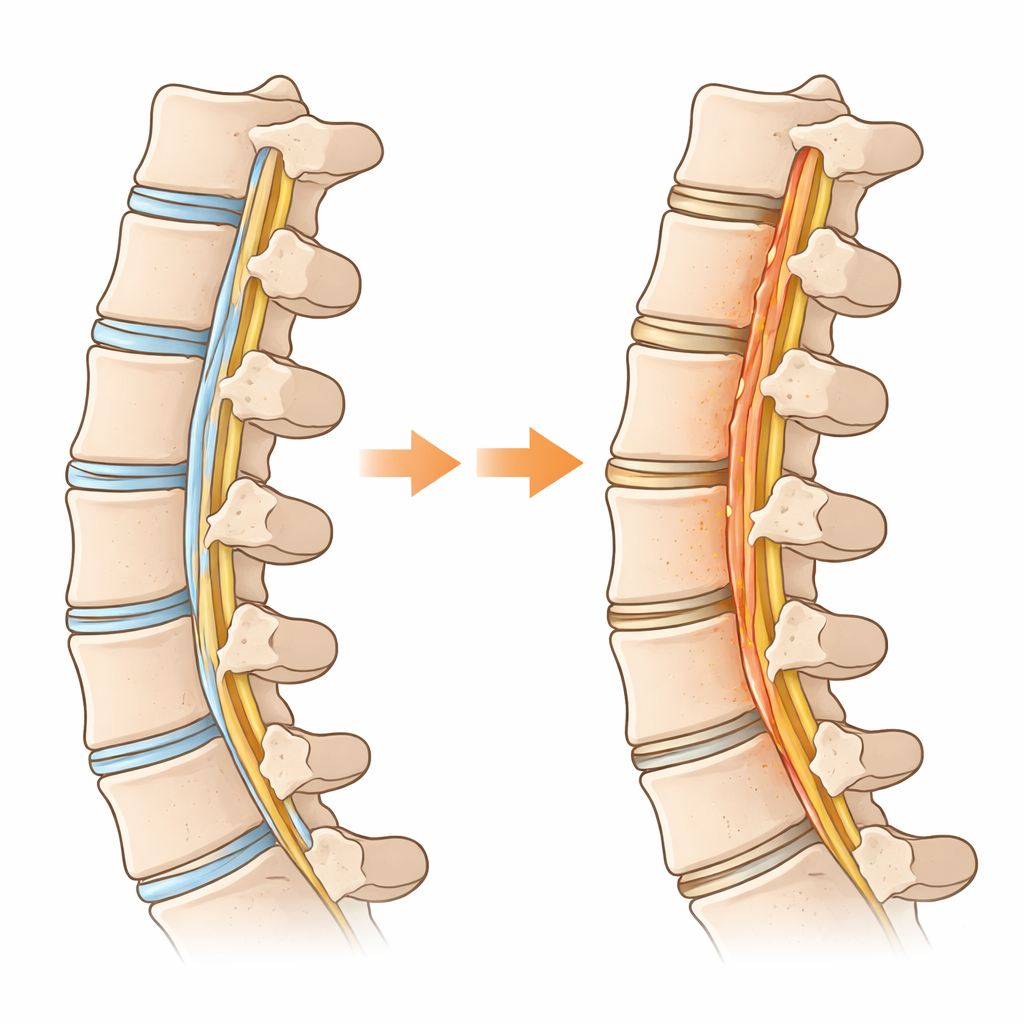
Why a Spinal Ligament Starts Behaving Like Bone
The researchers compared diseased ligament tissue from people with OPLL to normal ligament from patients with other neck problems. Using large-scale gene and single-cell analyses, they mapped out which cell types were present and what each cell was doing. They found that a subset of ligament cells, which normally act as repair-ready “progenitors,” were following a path toward becoming bone-forming cells. Along this path, the cells flipped their energy strategy: instead of mainly burning fuel in their mitochondria, they shifted toward fast sugar breakdown, a style of metabolism often seen in cancer and growing tissues. This switch to high-speed sugar use—called glycolysis—was especially strong in the cells that were actually forming bone inside the ligament.
Sugar Burning as the Fuel for Rogue Bone Growth
To test whether this metabolic shift was merely a bystander or a driver of disease, the team isolated ligament cells from patients and watched them as they were nudged to become bone-forming cells in the lab. Cells from OPLL patients soaked up more glucose, produced more acid-forming byproducts such as lactate, and showed weaker mitochondrial activity than normal cells. When the scientists blocked glycolysis with a drug named 2-DG, the cells’ ability to turn into bone-forming cells dropped sharply, and the mineral deposits they normally laid down were greatly reduced. In mice, the same drug shrank artificially induced bone masses and decreased the number of early and mature bone cells, showing that cutting off this sugar-burning pathway can slow or soften the ossification process in living animals.
A Hidden Switch: The ADAM12–IGF1 Chain Reaction
Digging deeper, the researchers searched for master switches that might be flipping the cells into this high-glycolysis mode. One molecule, ADAM12, stood out. It was strongly elevated in OPLL tissue and was most active in ligament cells on the road to becoming bone. ADAM12 is a protein-cleaving enzyme that floats outside cells, and here it appears to slice open carrier proteins that hold onto a growth signal called IGF1. By trimming one of these carriers, IGFBP5, ADAM12 frees more IGF1 to activate receptors on ligament cells. That, in turn, triggers a well-known growth pathway inside the cell (PI3K–AKT–mTOR), which boosts the machinery for breaking down glucose and producing lactate. When ADAM12 was increased in cells, glycolysis and lactate production surged; when it was silenced or its cutting ability disabled, both dropped.
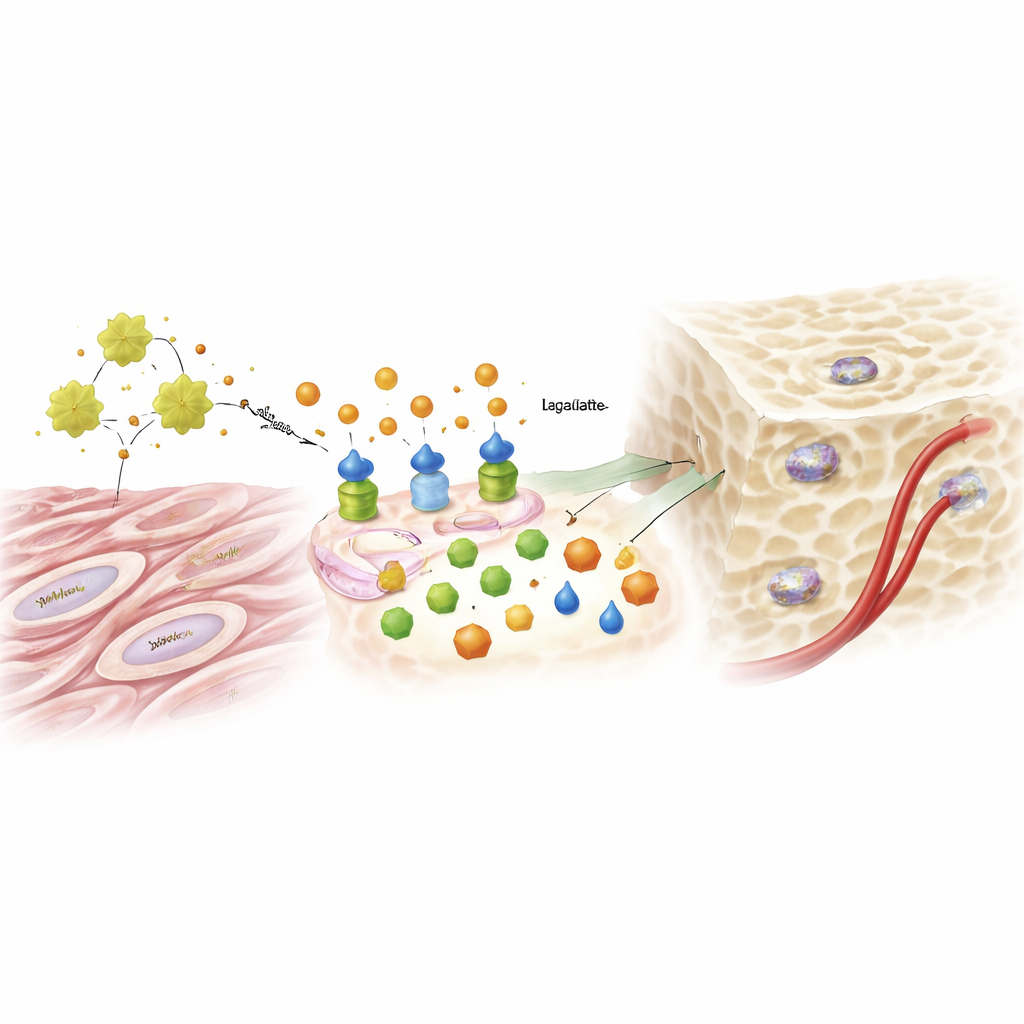
From Sugar Byproduct to Genetic Signal
Lactate, long dismissed as metabolic waste, played a surprising starring role. As glycolysis ramped up under the influence of ADAM12 and IGF1, lactate levels rose and began to influence the way DNA is packaged inside the cell nucleus. The researchers found that lactate decorated specific histones—the proteins that organize DNA—with a chemical tag called lactylation. This tag loosened up regions of DNA linked to bone-forming genes, helping turn on key factors that push a cell to become an osteoblast. Supplying extra lactate could rescue bone-forming ability in cells where ADAM12 had been knocked down, while blocking glycolysis or the enzyme that makes lactate erased the boost from extra ADAM12. The same ADAM12-driven pathway also increased signals that encourage blood vessel growth, helping supply nutrients to the new bone.
New Paths Toward Non-Surgical Treatment
Taken together, the study paints a clear chain of events: a rise in the enzyme ADAM12 in spinal ligaments unlocks extra IGF1, which flips on a growth program that rewires how cells use sugar. This sugar rush produces lactate, which then rewrites the activity of bone-related genes and helps build both bone and blood vessels in places where they do not belong. By blocking IGF1 signaling or glycolysis, the researchers were able to blunt or reverse this process in cell cultures and mouse models. For patients, this work suggests that targeting the ADAM12–IGF1–glycolysis axis could one day offer a drug-based way to slow or prevent OPLL, reducing the need for high-risk spinal surgery and preserving mobility and nerve function.
Citation: Zhao, Q., Wu, H., Xie, D. et al. Glycolytic reprogramming mediated by the ADAM12/IGF1 axis promotes ossification of the posterior longitudinal ligament. Cell Death Discov. 12, 178 (2026). https://doi.org/10.1038/s41420-026-03044-8
Keywords: spinal ligament ossification, cell metabolism, glycolysis, growth factor signaling, bone formation