Clear Sky Science · en
Sublethal DNA damage switches off B cell effector programs in an RA-FLS-PBMC co-culture
Turning Down an Overactive Immune Response
Rheumatoid arthritis is driven in part by immune cells that attack the body’s own joints, causing pain, swelling, and long-term damage. Many current drugs work by wiping out whole groups of immune cells, which can leave patients vulnerable to infections. This study explores a more subtle idea: whether a tiny, carefully controlled dose of DNA damage could temporarily switch off the harmful activity of certain immune cells—especially B cells—without actually killing most of them. If successful, this approach could point toward gentler treatments that calm the disease while keeping much of the immune system intact.
Why B Cells Are a Special Weak Spot
B cells are best known for making antibodies, but in rheumatoid arthritis they also help sustain inflammation and organize immune “hot spots” inside the joint. To fine-tune the antibodies they produce, activated B cells deliberately cut and re-stitch their own DNA. This unusual behavior makes them more sensitive to additional damage than many other cells. The researchers reasoned that a small extra “nudge” of DNA damage, below the level that causes mass cell death, might exploit this vulnerability and selectively quiet B cell activity. At the same time, they wanted to avoid broadly poisoning immune cells or harming the structural cells that form the joint lining.
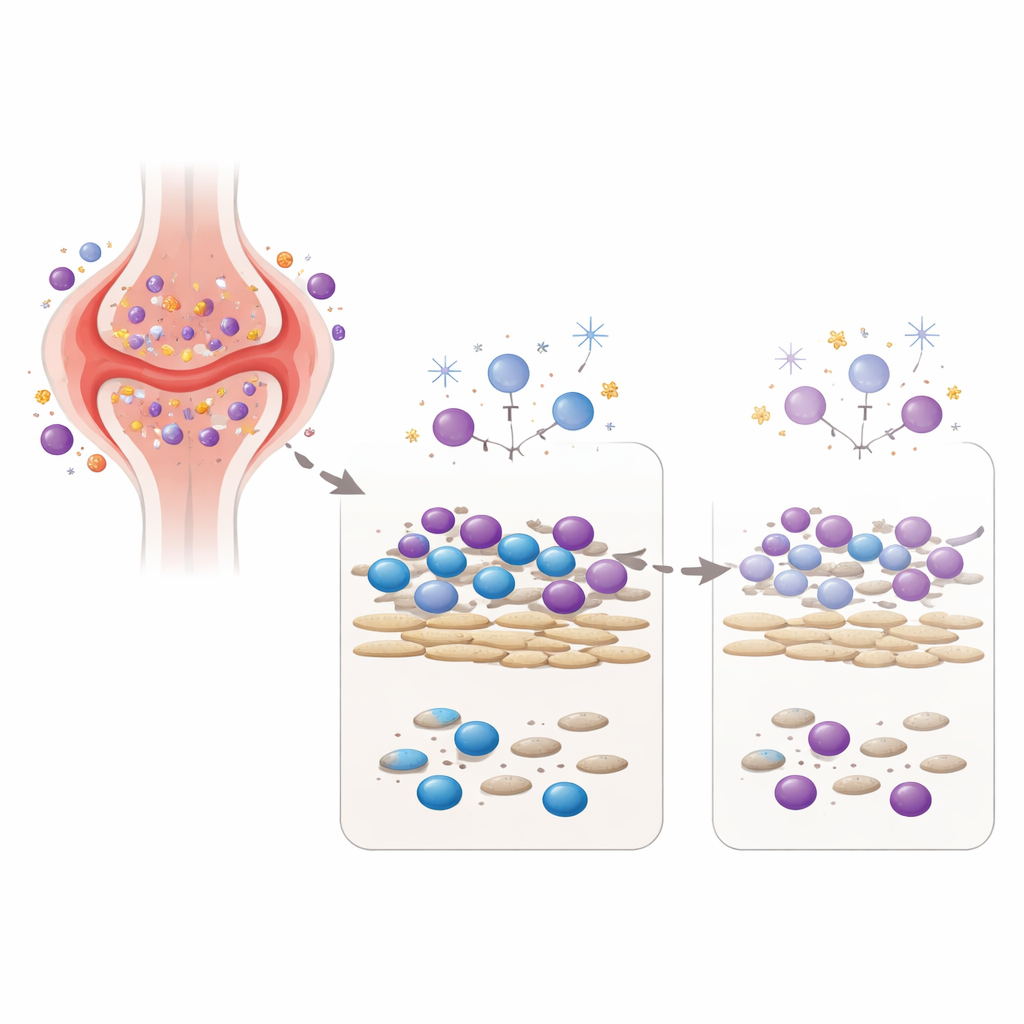
Recreating the Arthritic Joint in a Dish
To test this concept, the team set up a laboratory model that brings together two key players from the joint: fibroblast-like synoviocytes, which are connective tissue cells from patients with rheumatoid arthritis, and blood immune cells from healthy volunteers. In this co-culture, the fibroblasts provide signals that keep immune cells alive and active, somewhat like the environment inside an inflamed joint. The researchers then exposed this mixed cell community to a single, carefully chosen dose of three different DNA-damaging agents: low-dose gamma irradiation, hydrogen peroxide (a reactive oxygen species also found in inflamed tissue), and a drug metabolite related to cyclophosphamide, a chemotherapy used in severe autoimmune disease. They measured not only how many cells survived, but also how much antibody and cytokine—immune messenger molecules—the cells produced days later.
Shutting Off Function Without Massive Cell Loss
Across all three agents, a clear pattern emerged: B cell functions were strongly reduced at doses that still left most cells alive. For example, after a modest dose of gamma irradiation, overall cell survival remained above 80 percent, yet key signals such as the anti-inflammatory cytokine IL-10 and several antibody types dropped by roughly half or more. Hydrogen peroxide and the chemotherapy metabolite showed similar or even stronger dampening of antibody and cytokine production, sometimes while more than two-thirds of the cells were still viable. In other words, the genotoxic “hit” uncoupled immune activity from cell survival—cells were present, but their ability to fuel inflammation was sharply dialed down.
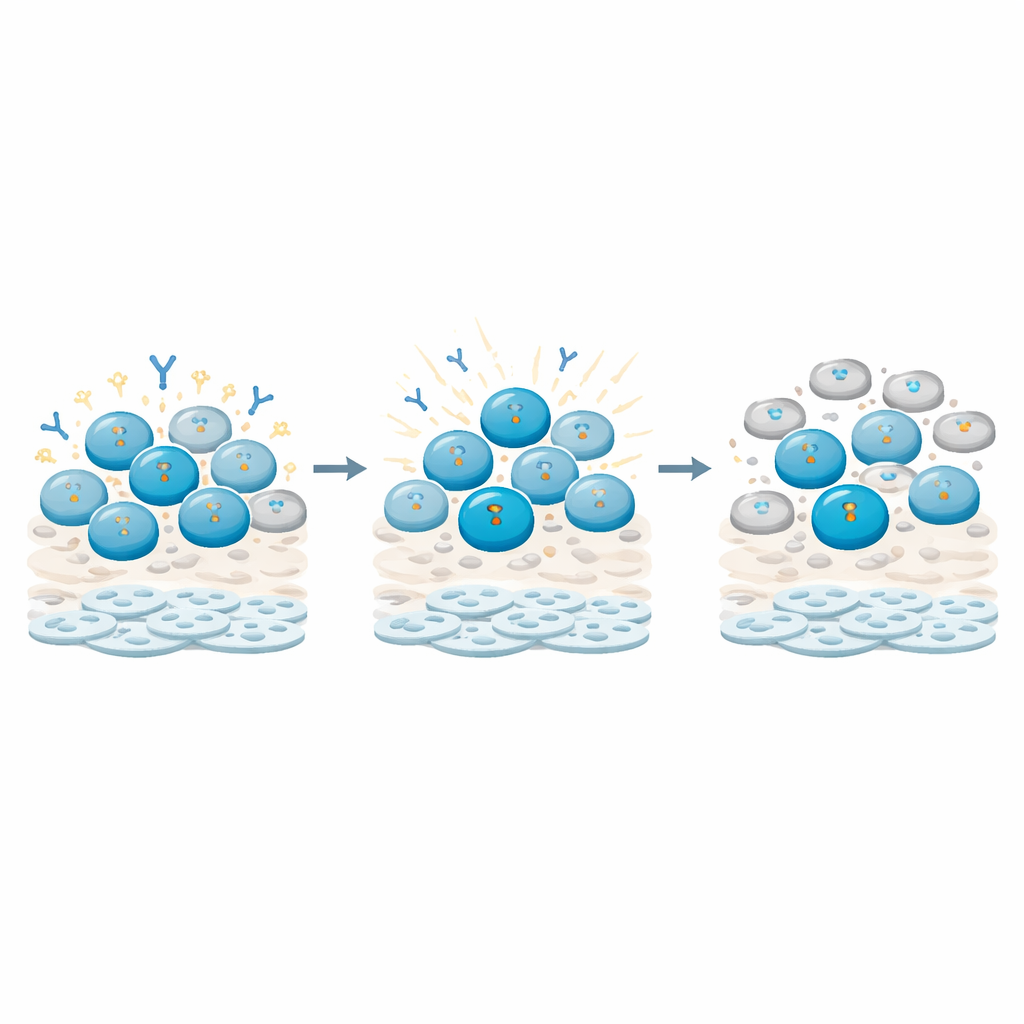
How B Cells Sense Damage and Hit the Brakes
Digging deeper, the investigators tracked molecular signs of DNA damage inside different immune cell types. They found that B cells, especially memory B cells (those primed by past encounters), accumulated particularly strong and lasting signals of DNA injury compared with T cells. These signals fed into checkpoints that halted the B cells’ cell cycle, keeping them stuck in a resting phase rather than actively dividing and differentiating. At the same time, gene activity linked to antibody production and B cell maturation was reshaped: some master regulators and antibody-chain transcripts rose or fell in patterns suggesting that the cells were being held in a “repair first, function later” state. T cells, by contrast, showed more transient responses and largely recovered their cycling behavior.
A New Angle on Softer Autoimmune Therapies
Overall, the study shows that a single, low-level burst of DNA damage can selectively quiet B cell programs in an arthritis-like environment while leaving most T cells and joint-lining cells intact. Rather than obliterating B cells, this approach nudges them into a checkpoint state where they stop pumping out inflammatory molecules and antibodies. For patients, the idea remains theoretical, but it suggests that precisely tuned, short-lived exposures to radiation or genotoxic drugs might someday be used not just to kill cells, but to temporarily silence the most troublesome ones. Such strategies could complement existing therapies and, in principle, offer a way to tame chronic autoimmune inflammation with less collateral damage to the immune system.
Citation: Bruci, D., Lowin, T., Fritz, G. et al. Sublethal DNA damage switches off B cell effector programs in an RA-FLS-PBMC co-culture. Cell Death Discov. 12, 161 (2026). https://doi.org/10.1038/s41420-026-03021-1
Keywords: rheumatoid arthritis, B cells, DNA damage, immune modulation, autoimmune disease