Clear Sky Science · en
Integration of immunogenic cell death in the treatment landscape of non-small cell lung cancer: harnessing the power of the immune system
Why Teaching Tumors to Alert the Immune System Matters
Lung cancer remains one of the deadliest cancers worldwide, in large part because tumors often return or resist standard treatments. This article explores a promising idea: instead of just killing cancer cells quietly, doctors may be able to make them die in a way that loudly alerts the body’s defenses. By turning tumor destruction into a kind of internal vaccination, a treatment called immunogenic cell death could help the immune system recognize, remember, and more effectively fight non‑small cell lung cancer.
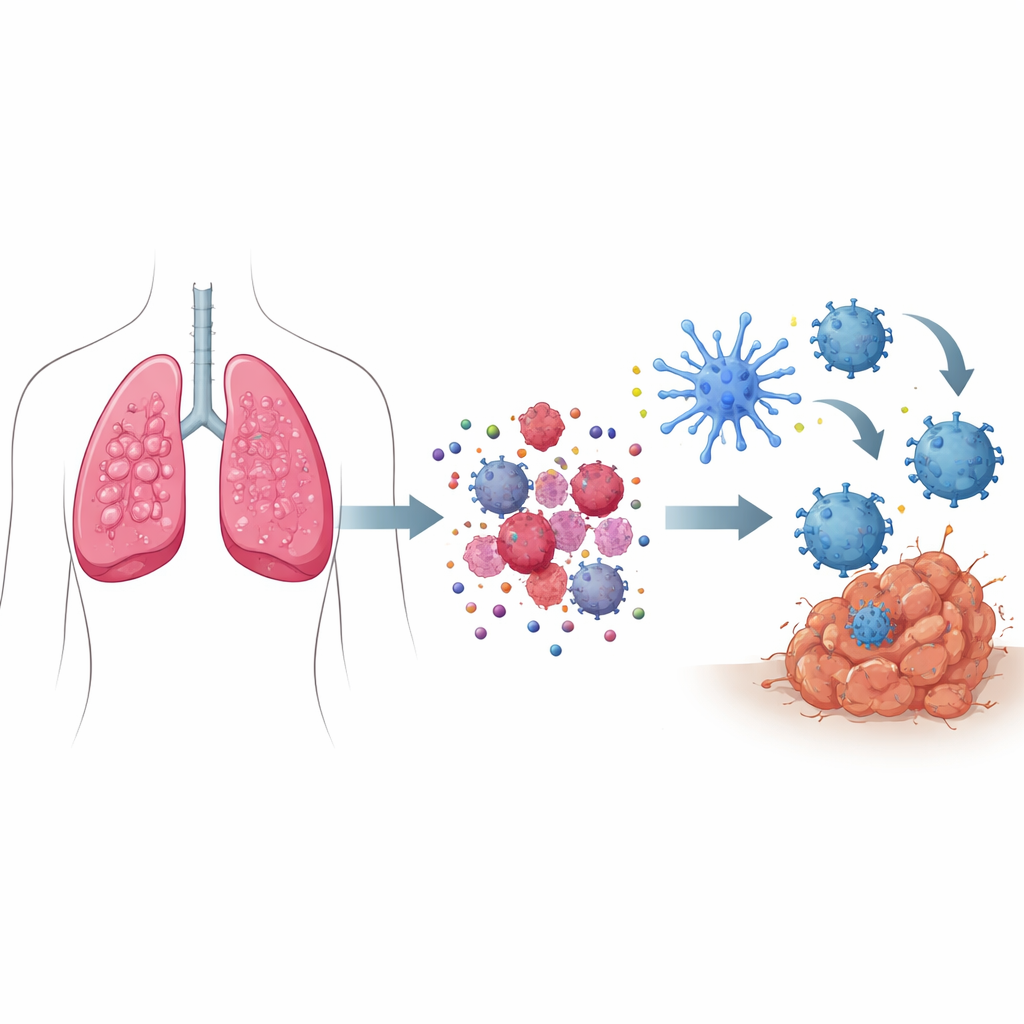
How “Danger Signals” Turn Cell Death into a Call for Help
When most cells die, they do so silently, without stirring the immune system. In immunogenic cell death, however, dying cancer cells send out a coordinated set of danger signals. Stressed components inside the cell push certain proteins to the cell surface, marking it as something to be “eaten” by immune sentinels. At the same time, small molecules such as ATP seep out, acting like flares that draw immune cells into the area, while other proteins are released that behave like alarms. Together, these signals attract and activate specialized scouts called dendritic cells, which swallow bits of the tumor, process them, and then present tumor fragments to T cells—training them to seek and destroy cancer cells throughout the body.
Turning Common Treatments into Immune Trainers
Many familiar lung cancer therapies can trigger this kind of alarm‑filled cell death if used in the right way. Certain chemotherapy drugs, including widely used taxanes and platinum compounds, can drive tumor cells into a stressed state that leads to the release of danger signals. These treatments not only shrink tumors directly but can also spur T cells and beneficial macrophages into action. Newer, more targeted forms of chemotherapy, called antibody–drug conjugates, deliver toxic payloads precisely to cancer cells. In early studies, these targeted drugs not only kill tumors more cleanly but also appear to spark stronger immune responses with fewer whole‑body side effects.
Radiation, Targeted Drugs, and Light-Based Therapy as Immune Partners
Radiation therapy does more than scorch tumors; by damaging DNA and generating reactive molecules inside cancer cells, it can push them toward immunogenic death. However, the effect depends heavily on dose and schedule: moderate, well‑timed radiation can be strongly immune‑stimulating, while overly high doses may actually dampen key sensing pathways. Researchers are also designing nanoparticles that amplify the helpful signals and counteract the suppressive ones. Meanwhile, precision drugs that block growth drivers such as EGFR, ALK, AXL, or blood‑vessel signals are now understood to do double duty: they slow tumor growth and, in many models, help expose tumor material and danger cues that wake up T cells. Light‑activated treatments, which use photosensitive compounds to generate bursts of toxic oxygen species inside tumors, can be tuned to hit cellular structures that most effectively trigger immune‑visible cell death.
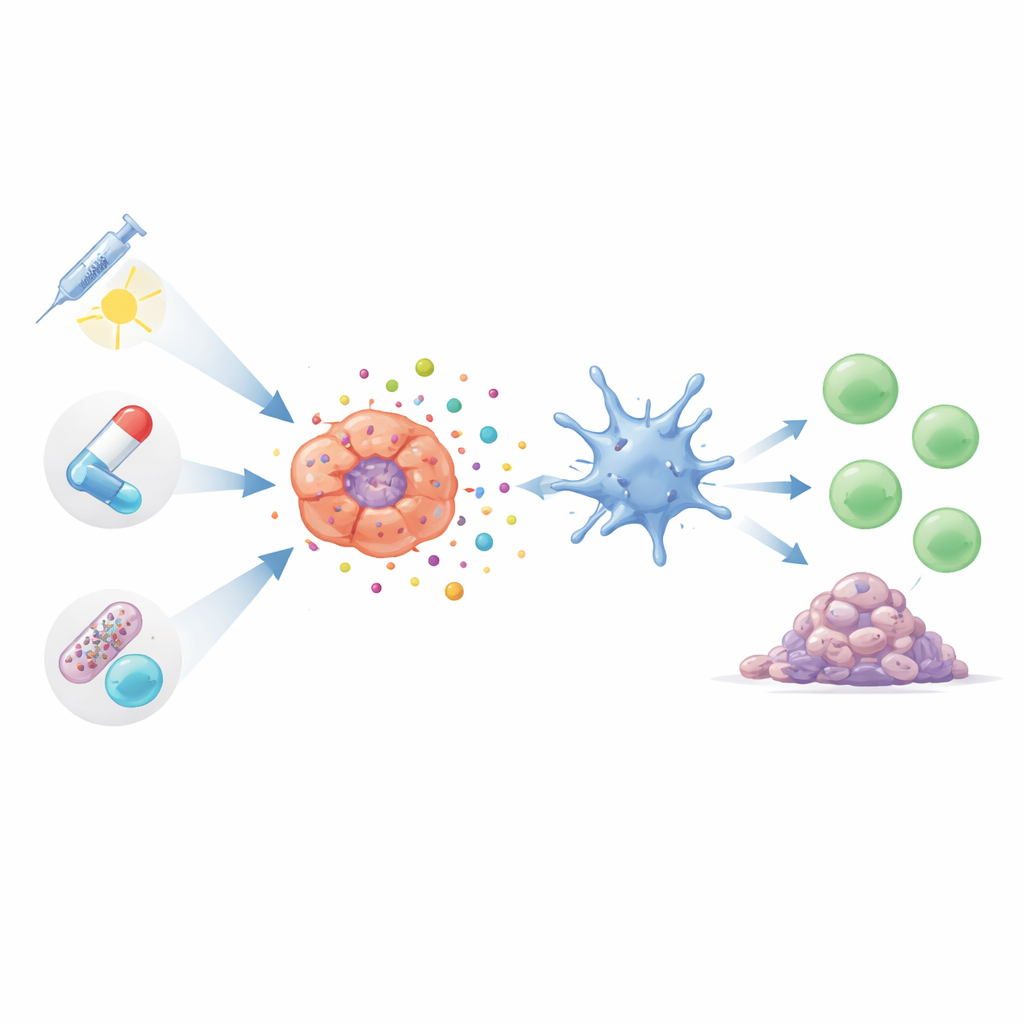
New Players: Viruses, Electric Fields, and the Gut Microbiome
Beyond the standard pillars of cancer care, several emerging tools also rely on immunogenic cell death. Engineered viruses that selectively infect lung cancer cells burst them open while at the same time drawing in immune cells, sometimes leading to regression of distant, untreated tumors in experimental models. Low‑intensity alternating electric fields, already used in other cancers, have been shown to stress lung cancer cells in ways that release danger signals and encourage T‑cell infiltration. Even the bacteria living in our intestines may influence how strong these ICD‑driven responses are. Certain gut microbes seem to promote a more active anti‑tumor environment, while others foster an immune‑dampening state that could blunt the benefits of ICD‑based treatments.
What This Means for Future Lung Cancer Care
The overall message of the article is that how a tumor cell dies matters as much as whether it dies at all. If death is immunogenic, each destroyed cancer cell can become a lesson for the immune system, helping it recognize and attack remaining cancer and possibly prevent relapse. To make this a practical reality for people with non‑small cell lung cancer, researchers still need clear markers to tell when ICD is happening in patients, better ways to match doses and combinations of therapies, and strategies to counteract the tumor’s many escape routes. If these challenges can be met, future treatment plans may routinely pair ICD‑inducing therapies with immune‑boosting drugs, transforming short‑lived responses into more durable control of the disease.
Citation: Liu, Z., Xu, X., Wang, M. et al. Integration of immunogenic cell death in the treatment landscape of non-small cell lung cancer: harnessing the power of the immune system. Cell Death Discov. 12, 155 (2026). https://doi.org/10.1038/s41420-026-03012-2
Keywords: immunogenic cell death, non-small cell lung cancer, cancer immunotherapy, chemoradiotherapy, tumor microenvironment