Clear Sky Science · en
Klebsiella pneumoniae LPS drives stromal-mediated repression of p53 and colorectal cancer chemoresistance
Why Your Gut Bacteria Matter for Cancer Treatment
Colorectal cancer is usually treated with powerful chemotherapy drugs, but many tumors learn to survive these attacks. This study explores an unexpected partner in crime: certain gut bacteria. The researchers show how molecules from these microbes can silently weaken one of our body’s main cancer-fighting safeguards inside tumors, making standard treatments less effective—even when the cancer cells themselves are not directly touched by the bacteria.
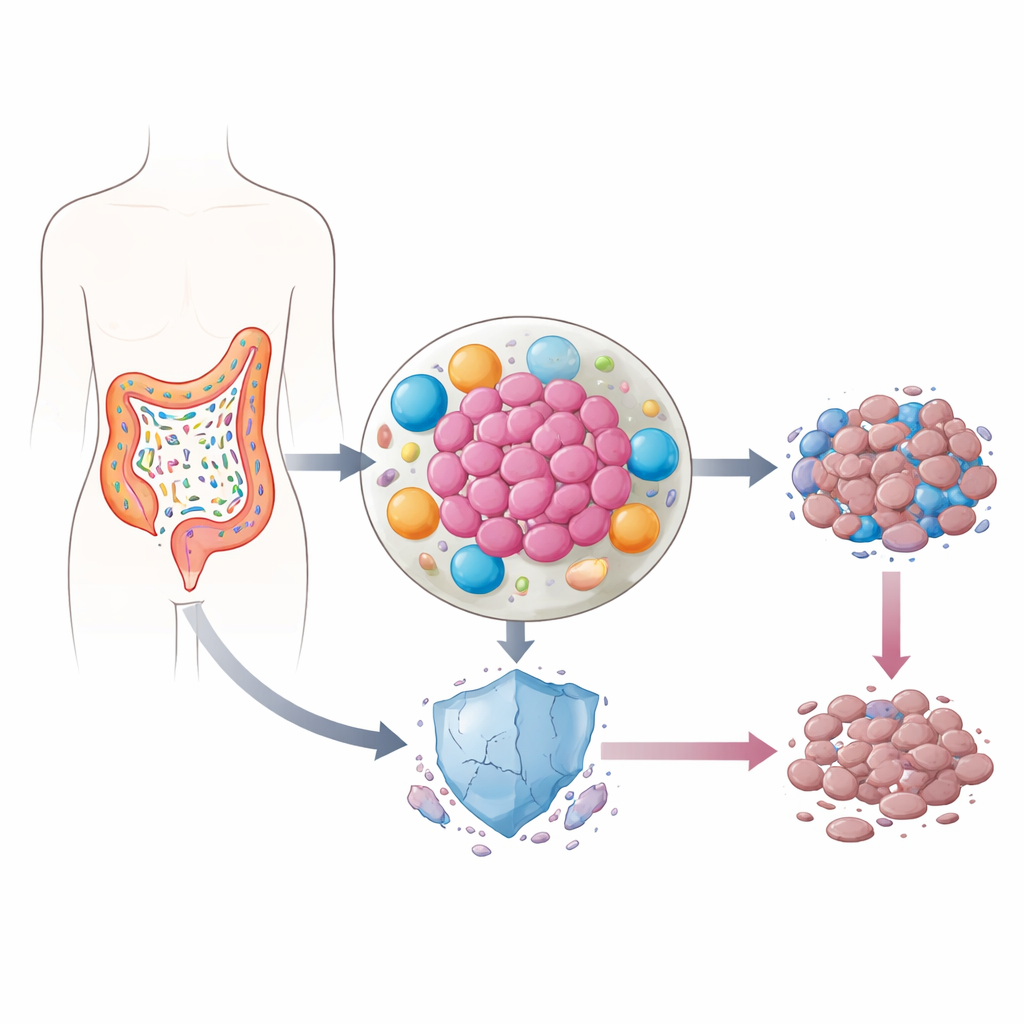
Germs Living Inside Tumors
Our intestines are home to trillions of bacteria, most of them harmless or even helpful. In colorectal cancer, however, the balance of species often shifts. Some bacteria move into the tumor itself, where they release inflammatory molecules and reshape the neighborhood of cells that surround the cancer. Earlier work suggested that in these inflamed pockets, levels of p53—a protein often called the “guardian of the genome” because it helps damaged cells self-destruct—are unusually low. Since p53 is crucial for how cancer cells respond to chemotherapy, the authors asked whether gut bacteria could be quietly sabotaging this safeguard.
A Hidden Support Network Around Tumor Cells
The team focused on lipopolysaccharide (LPS), a strong inflammatory molecule found on the surface of common gut microbes such as Klebsiella pneumoniae and other Enterobacteria, which are often increased in colorectal cancer. Surprisingly, when they added LPS directly to colorectal cancer cells, nothing much happened: the cells did not become more drug-resistant and p53 levels stayed the same. The picture changed completely when they first exposed immune cells called macrophages or connective-tissue cells called fibroblasts to LPS, and then transferred the liquid these cells had been growing in to the cancer cells. Under these conditions, chemotherapy killed far fewer tumor cells, and p53 protein failed to build up as it normally would after treatment.
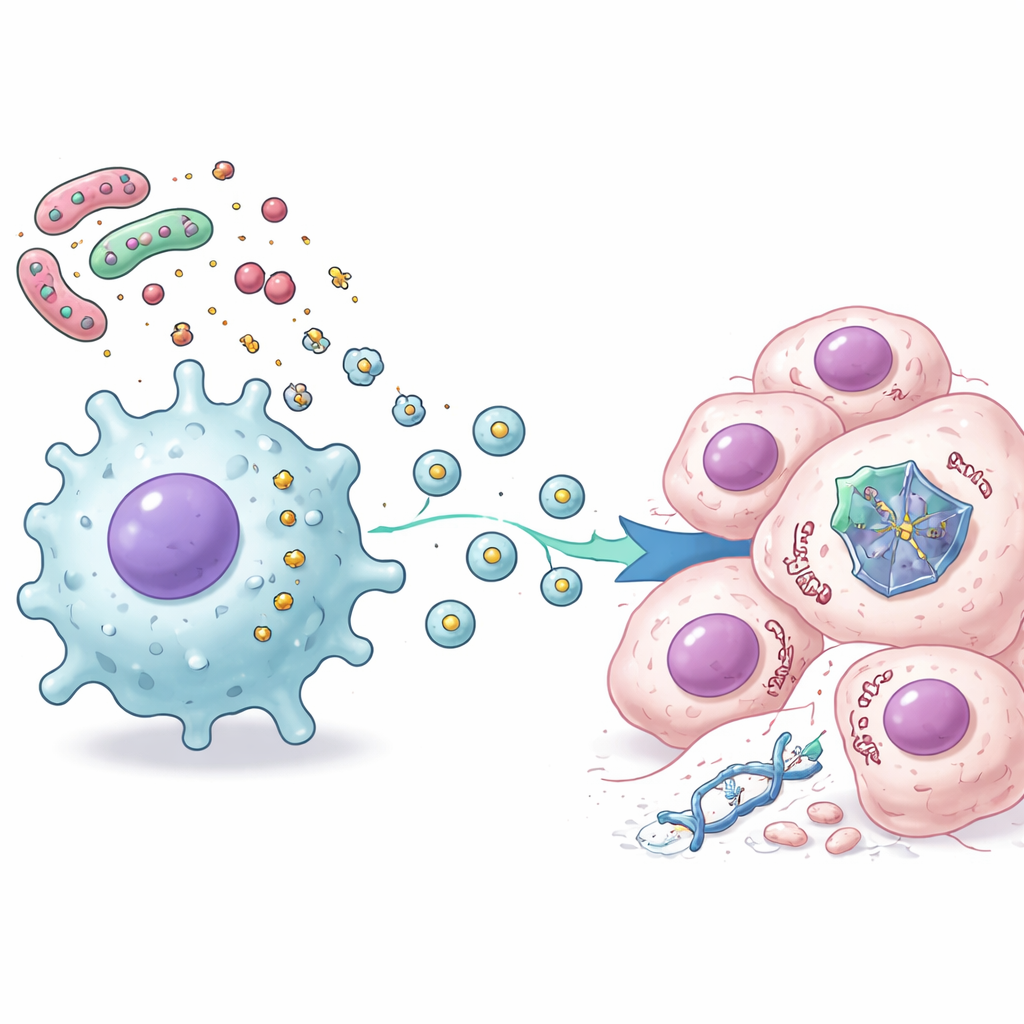
Messages Packaged in Tiny Vesicles
To understand what macrophages were sending, the researchers used advanced protein analysis to chart thousands of changes inside cancer cells treated with both chemotherapy and LPS-stimulated macrophage secretions. They saw a clear pattern: signals related to inflammation and immune activation went up, while many p53-controlled pathways involved in cell division and controlled self-destruction went down. Further experiments showed that this suppression did not stem from classic inflammatory hormones like interleukin-6. Instead, the key messengers were tiny membrane bubbles, known as extracellular vesicles, released by the activated macrophages. When these vesicles were isolated and added to cancer cells, p53 protein was destabilized and more rapidly destroyed by the cell’s own recycling machinery, shielding the cells from p53-driven death.
A Selective Silencing of the Cell’s Guardian
The authors discovered that this bacterial–stromal signal does not simply turn p53 off everywhere. Rather, it selectively dampens one group of p53-controlled genes, while leaving others—especially those linked to inflammation—unchanged or even boosted. When they looked at tumor samples from colorectal cancer patients, they found a matching pattern: in tumors where inflammation-related gene signatures were high, this “p53 activity” gene cluster tended to be low, particularly in cancers that still carried an intact TP53 gene. Importantly, patients whose tumors had strong expression of this p53-dependent cluster tended to fare better on chemotherapy than those with low expression, suggesting that non-genetic silencing of p53 by the inflamed tumor environment may worsen clinical outcomes.
What This Means for Patients and Therapies
Taken together, the work reveals a chain of events that links gut bacteria to chemotherapy resistance: bacterial LPS activates nearby stromal and immune cells, these cells release extracellular vesicles, and the vesicles enter cancer cells to destabilize p53 protein and blunt its life-or-death decisions after treatment. Even when the p53 gene itself is normal, this outside pressure can effectively mute its protective role. For patients, this suggests that the state of the gut microbiome and local inflammation could help explain why some colorectal tumors shrug off standard drugs. In the future, strategies that reshape gut bacteria, calm harmful inflammation, or block the production or action of these vesicles may help restore p53’s tumor-suppressing power and make existing chemotherapies work better.
Citation: Fragkoulis, K., Łasut-Szyszka, B., Végvári, Á. et al. Klebsiella pneumoniae LPS drives stromal-mediated repression of p53 and colorectal cancer chemoresistance. Cell Death Dis 17, 395 (2026). https://doi.org/10.1038/s41419-026-08756-4
Keywords: colorectal cancer, gut microbiome, chemotherapy resistance, p53 tumor suppressor, tumor microenvironment