Clear Sky Science · en
LincRNA-EPS alleviates osteoclastogenesis under inflammatory microenvironment through preventing excessive iron metabolism
Why this matters for sore gums and fragile teeth
Many adults live with bleeding gums and loose teeth caused by periodontitis, a chronic infection that quietly eats away the bone holding teeth in place. This study looks inside that process and discovers how a little-known RNA molecule helps keep bone-eating cells and iron use in balance, pointing to new ways to protect the jawbone during gum disease.
Bone eaters at work in inflamed gums
In periodontitis, bacteria around the teeth stir up a strong immune response in the gums. This inflammation wakes up special bone-eating cells called osteoclasts, which dig holes in the jawbone and can eventually lead to tooth loss. The authors used a mouse model of gum disease created with bacterial components tied around the teeth, and confirmed by 3D scans that inflammation led to clear bone loss. They focused on a molecule named lincRNA-EPS, a noncoding RNA already known to calm inflammatory signals in other tissues, to see whether it also reins in osteoclast activity in this harsh environment.
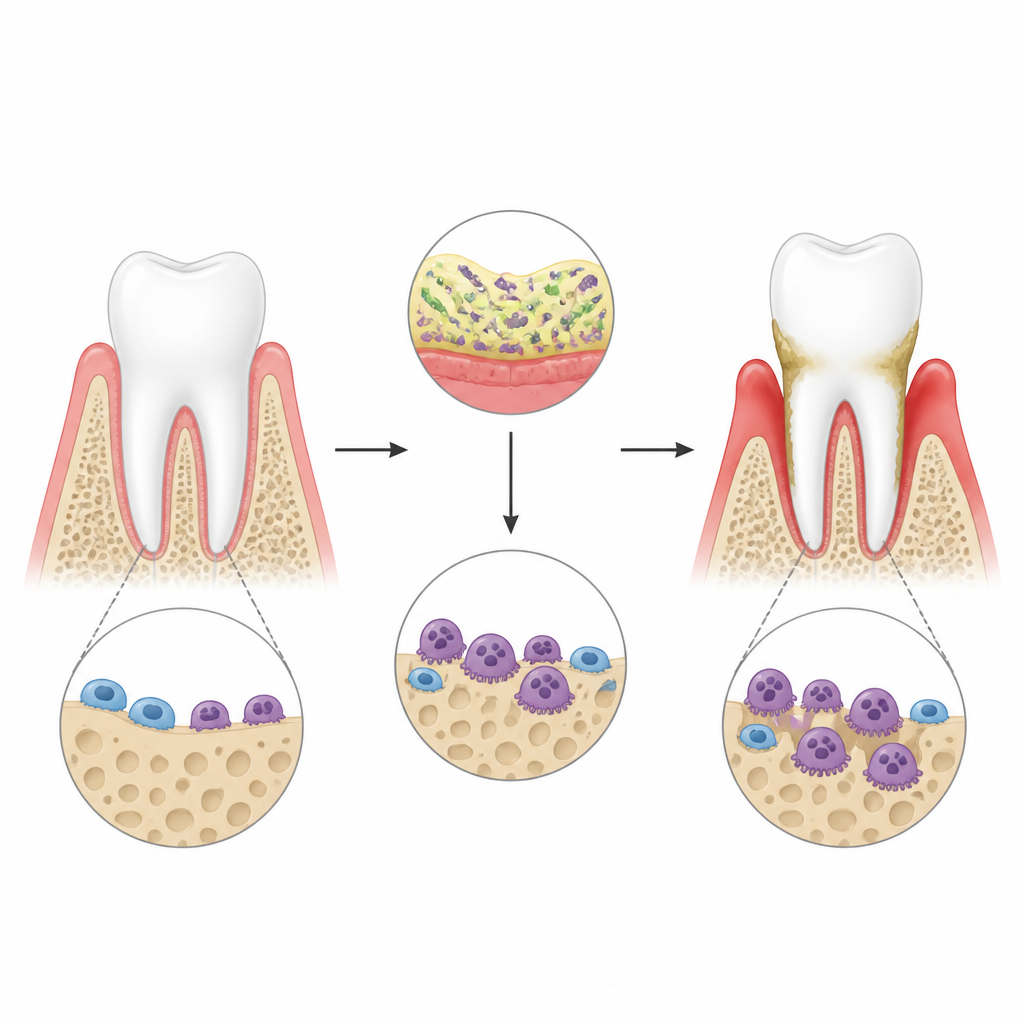
A protective RNA that keeps bone loss in check
When the researchers removed lincRNA-EPS from mice, bone loss around the teeth became noticeably worse after gum infection. The jawbone surface showed more and larger osteoclasts, and the pits they carved into bone slices in the lab were deeper and wider. In cell culture, bone marrow cells from normal and knockout animals were first nudged toward becoming osteoclasts, then exposed to an inflammatory trigger that mimics bacterial toxins. Cells lacking lincRNA-EPS turned into aggressive osteoclasts more readily and switched on bone-resorbing genes and proteins at higher levels. These findings suggest that lincRNA-EPS normally acts like a brake on osteoclast growth and function under inflammatory stress.
Iron handling as the hidden link
To understand how this RNA exerts its effect, the team compared gene activity and protein patterns in precursor cells and mature osteoclasts from normal and knockout mice. They found not only stronger osteoclast programs, but also striking changes in pathways that control iron use, storage and transport. One secreted protein, lipocalin-2 (Lcn2), which can shuttle iron in and out of cells, stood out as strongly increased when lincRNA-EPS was missing, especially after inflammatory stimulation. Further tests showed that in tissues and cultured cells without lincRNA-EPS, Lcn2 levels stayed high, iron-handling proteins remained switched on for longer, and mobile iron inside osteoclasts stayed elevated, all signs of an overactive iron economy that feeds the energy-hungry bone eaters.
Tuning an iron shuttle to calm bone damage
The scientists then directly manipulated Lcn2 to see how it influences this system. Reducing Lcn2 in normal precursor cells dampened iron transport machinery and altered osteoclast shape, producing smaller cells with fewer nuclei, even though their numbers could rise. Adding extra Lcn2 had the opposite effect, helping cells fuse into larger, more powerful bone-resorbing units. In mice with gum disease, lowering Lcn2 in the tissues around the teeth reduced bone loss and osteoclast activity, but only when lincRNA-EPS was present. When lincRNA-EPS was absent, changing Lcn2 levels or adding extra Lcn2 had weaker and slower effects on iron levels and osteoclast behavior, suggesting that this RNA is required for a fast and stable adjustment of iron metabolism during inflammation.
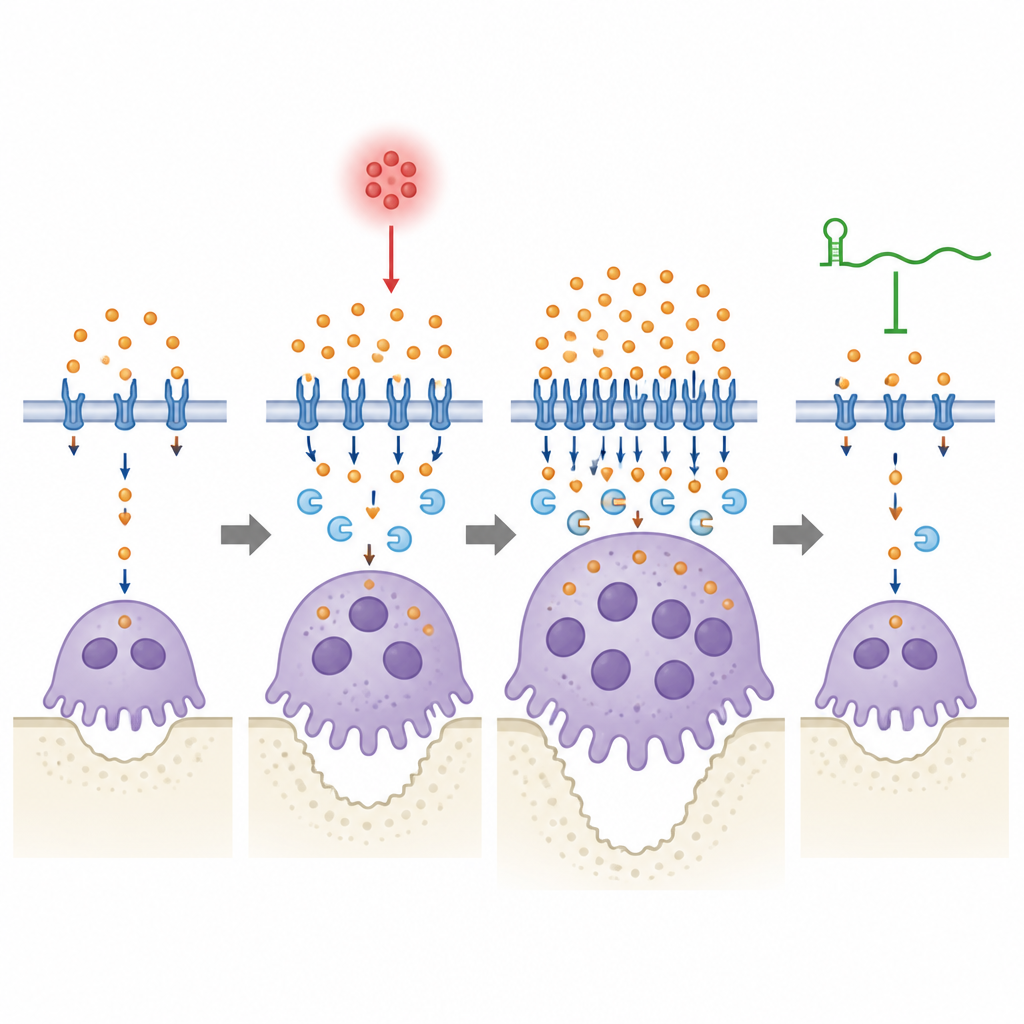
A fine-tuned balance between iron and bone
From a lay perspective, this work shows that a regulatory RNA, lincRNA-EPS, helps jawbone survive gum infection by preventing bone-eating cells from gorging on iron and staying overactive for too long. In an inflamed mouth, Lcn2 rises and pushes more iron into osteoclasts, helping them grow and chew through bone; lincRNA-EPS helps these cells sense and correct that surge so iron use returns to a safe level. When the RNA is missing, this safety valve fails, iron handling remains excessive, and bone loss worsens. Although the study is preclinical, it highlights iron regulation and the lincRNA-EPS–Lcn2 pair as potential targets for future therapies that aim to protect teeth by gently slowing bone resorption rather than shutting it down completely.
Citation: Wang, J., Wang, Y., Zhang, Z. et al. LincRNA-EPS alleviates osteoclastogenesis under inflammatory microenvironment through preventing excessive iron metabolism. Cell Death Dis 17, 444 (2026). https://doi.org/10.1038/s41419-026-08716-y
Keywords: periodontitis, osteoclasts, iron metabolism, lipocalin-2, long noncoding RNA