Clear Sky Science · en
Tumor suppressor function of SHMT in a Drosophila RasV12DlgRNAi model: DNA damage and synergistic gene-nutrient interaction with PLP
Why this fruit fly story matters
Cancer often rewires how cells use nutrients, but the same molecules that fuel tumors can sometimes hold them back. In this study, researchers used fruit flies to uncover how a single enzyme that handles vitamin-linked chemistry inside cells can act as a brake on cancer. They show that when this enzyme falters, DNA becomes unstable, tumors grow more aggressively, and a common vitamin and an antioxidant can shift the balance between tumor growth and cell death.
The cell’s tiny carbon traffic hub
Inside our cells, a network known as one-carbon metabolism quietly supports life by feeding building blocks into DNA production and chemical tags that control gene activity. At the center of this network sits an enzyme called SHMT, which works together with vitamin B6 in its active form. SHMT helps turn one amino acid into another while handing off small carbon units needed to make DNA. In many human cancers, versions of this enzyme are found at high levels and are thought to help tumors grow. Yet in some cancers, low levels of one SHMT form are linked to worse outcomes, hinting that the enzyme can also act as a tumor suppressor, depending on context.
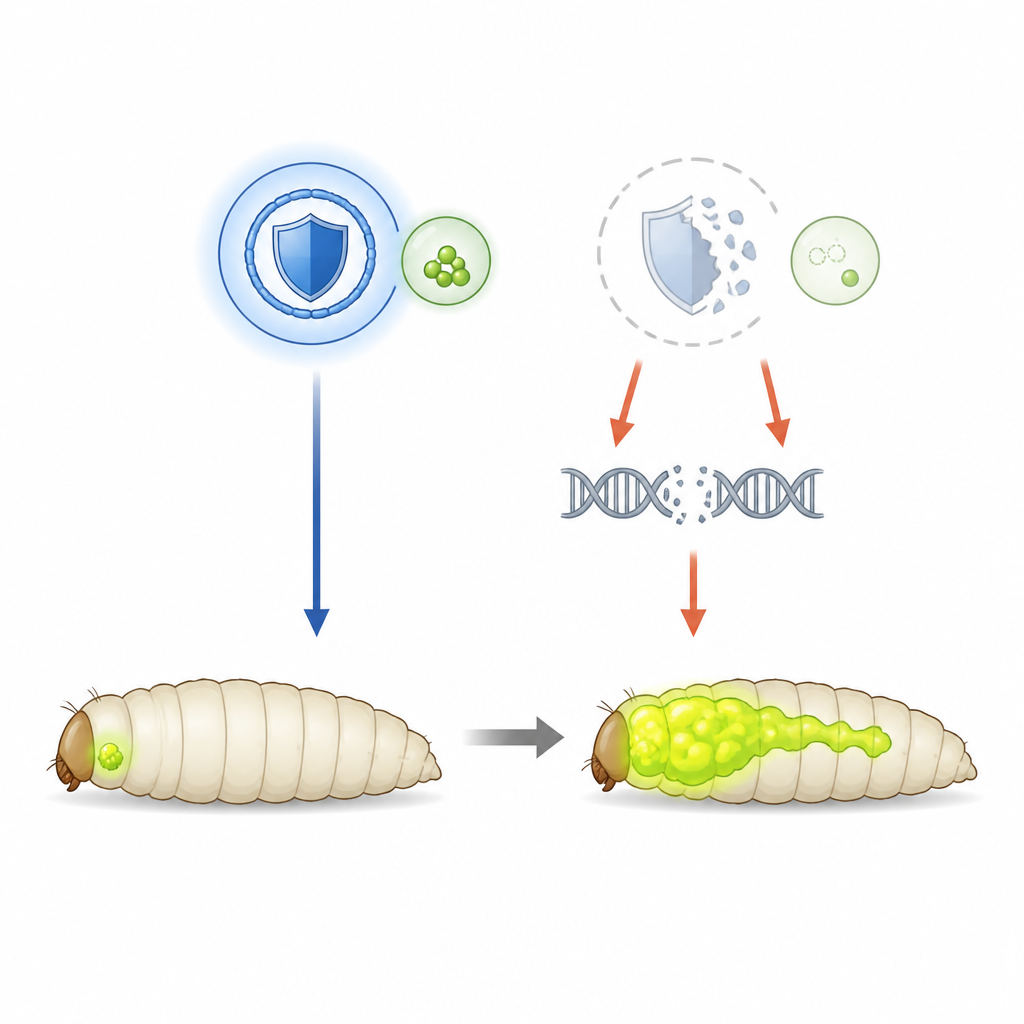
Using fruit flies to watch tumors grow
To untangle SHMT’s protective role, the team turned to fruit flies engineered to develop aggressive eye tumors driven by an overactive Ras gene and loss of a cell polarity gene. These tumors glow green, allowing scientists to track their size and spread in the larva. In this setting, SHMT levels are normal, so researchers could deliberately dial the enzyme down using RNA interference. When SHMT activity was reduced, the primary tumors grew larger and invaded the fly’s nervous system more often, showing that, here, SHMT acts as a brake on cancer progression rather than a fuel.
DNA building blocks, damage, and oxidative stress
SHMT normally feeds a pathway that produces thymidylate, an essential DNA building block. When SHMT was silenced, the supply of this building block faltered. The result was a surge in DNA double-strand breaks and visibly shattered chromosomes inside tumor cells, clear signs of genome instability. These cells also became unusually sensitive to DNA-damaging X-rays and to drugs that slow DNA copying, and they struggled to repair breaks once they occurred. At the same time, levels of reactive oxygen species, harmful oxygen-containing molecules, rose sharply. Treating larvae with thymidylate restored DNA building blocks and lowered both DNA damage and oxidative stress, while an antioxidant, N-acetyl cysteine, reduced reactive oxygen species, chromosome damage, and tumor growth. Together, these findings point to a chain of events in which poor DNA building block supply boosts oxidative stress, which then attacks DNA and helps tumors advance.
A vitamin cofactor tips the balance
SHMT depends on pyridoxal 5’-phosphate, the active form of vitamin B6, to function. The researchers lowered this vitamin inside flies using a dietary blocker and asked how it interacted with reduced SHMT. Cutting SHMT alone sped tumor growth and raised DNA damage. Lowering vitamin B6 alone had milder effects. Strikingly, combining both changes caused a dramatic spike in oxidative stress and DNA breakage, far more than either hit alone. Yet this did not produce proportionally larger tumors. Instead, many tumor cells switched on a key cell-death enzyme and underwent apoptosis, suggesting that extremely damaged cells were being eliminated. High-dose antioxidant treatment again reduced oxidative stress, DNA damage, and tumor size, underscoring the central role of reactive oxygen species in driving these outcomes.
What this means for cancer and diet
This work shows that, in a living animal, SHMT can act as a tumor suppressor by helping maintain DNA stability and keeping oxidative stress in check. When SHMT activity drops, especially together with reduced vitamin B6, tumor cells accumulate DNA damage that can either fuel cancer progression or, if pushed too far, trigger their own death. The study also reveals a direct gene–nutrient interaction between SHMT and vitamin B6 that shapes how tumors grow. While these findings come from fruit flies, they echo patterns seen in some human cancers and suggest that targeting SHMT, its vitamin cofactor, and reactive oxygen species together could inform future strategies to control tumor growth without relying solely on killing dividing cells.
Citation: Angioli, C., Ferriero, A., Pilesi, E. et al. Tumor suppressor function of SHMT in a Drosophila RasV12DlgRNAi model: DNA damage and synergistic gene-nutrient interaction with PLP. Cell Death Dis 17, 427 (2026). https://doi.org/10.1038/s41419-026-08602-7
Keywords: cancer metabolism, DNA damage, reactive oxygen species, vitamin B6, Drosophila tumors