Clear Sky Science · en
Sphingosine-1-phosphate promotes CD8 T cell exhaustion in breast cancer via exosomal transfer of TGFBR2
Why This Matters for Cancer Treatment
Many modern cancer therapies aim to unleash the body’s own immune system, especially killer CD8 T cells, to attack tumors. Yet in many breast cancers, these cells become tired and ineffective, a state known as exhaustion, and immunotherapy often works only modestly. This study uncovers a previously hidden way that breast tumors chemically “sedate” CD8 T cells at a distance, revealing a new signaling route that might be blocked to help existing immunotherapies work better.
A Fat Molecule That Shapes the Tumor Neighborhood
The researchers focused on sphingosine-1-phosphate (S1P), a fat-like signaling molecule found at high levels in breast cancer. S1P is already known to help tumors grow blood vessels, spread, and resist treatment. Here, the team asked whether S1P also helps tumors disarm CD8 T cells in their surroundings. When they grew human breast cancer cells with S1P and then co-cultured them with human CD8 T cells, the T cells divided far less, even when they could not physically touch the cancer cells. This contact-free suppression hinted that the tumor cells were releasing some kind of message-carrying particles into the culture.
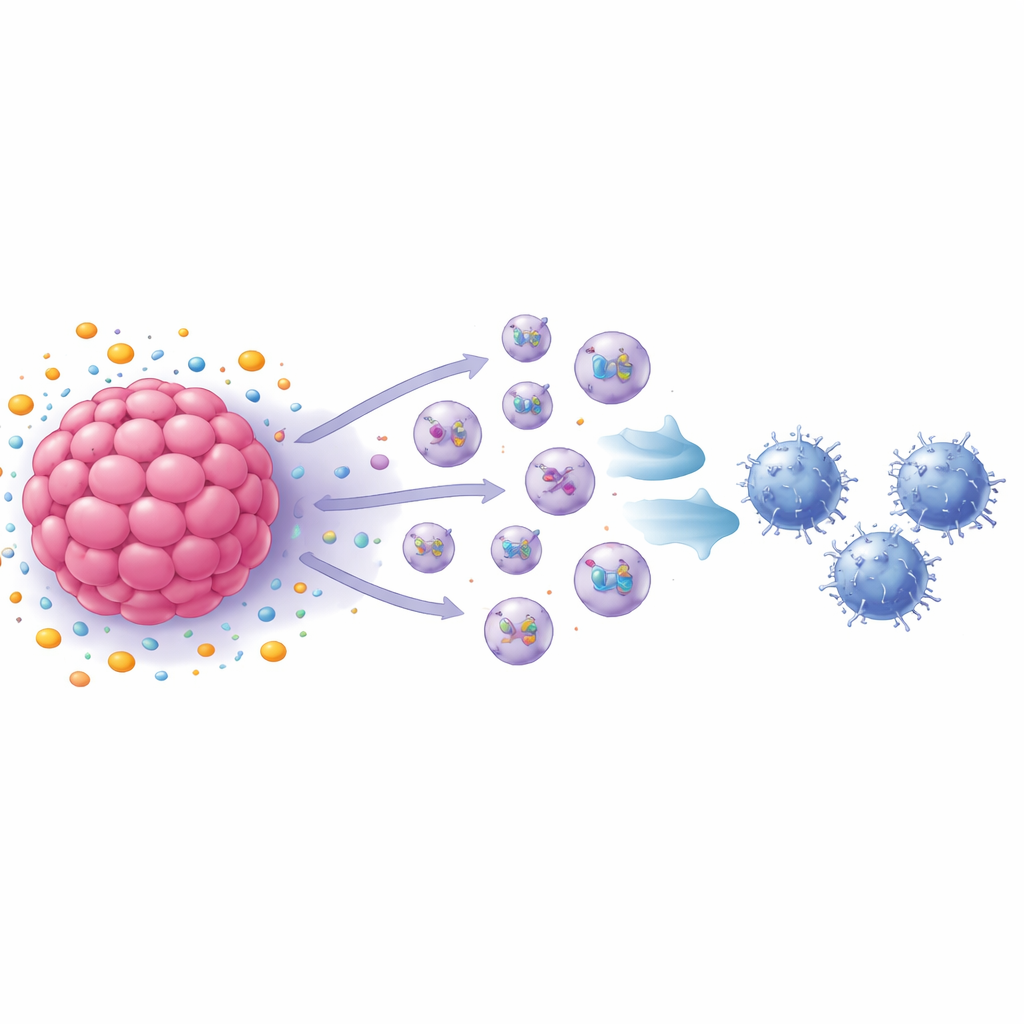
Tiny Vesicles as Messengers of Immune Suppression
The group next turned to exosomes—nanometer-scale vesicles shed by cells that ferry proteins and other molecules to distant targets. Blocking exosome production largely restored CD8 T cell growth, and analysis showed that S1P-treated breast cancer cells released many more exosomes than untreated cells. When these exosomes were added directly to activated CD8 T cells, the T cells slowed their proliferation and started to display hallmarks of exhaustion, including several surface and internal markers associated with worn-out immune cells. In animal experiments, exosomes from S1P-treated mouse breast cancer cells sped tumor growth and increased exhausted CD8 T cells inside tumors, while exosomes from cells lacking a key receptor had the opposite effect.
A Receptor Packed into Exosomes Does the Damage
Digging into what made these exosomes so harmful, the scientists looked at immune-related proteins enriched in their cargo. They found that one protein, the transforming growth factor beta receptor 2 (TGFBR2), was strongly increased on exosomes after S1P treatment, while another well-known checkpoint protein, PD-L1, was not. The exosomes fused with CD8 T cells and delivered TGFBR2 to the T-cell surface, effectively equipping the T cells with an extra copy of a receptor that drives exhaustion programs inside the cell. When the team blocked TGFBR2 on the exosomes with an antibody or reduced its levels in the cancer cells, the exosomes could no longer suppress T-cell activity or induce exhaustion markers, and CD8 T cells regained their ability to produce key attack molecules.
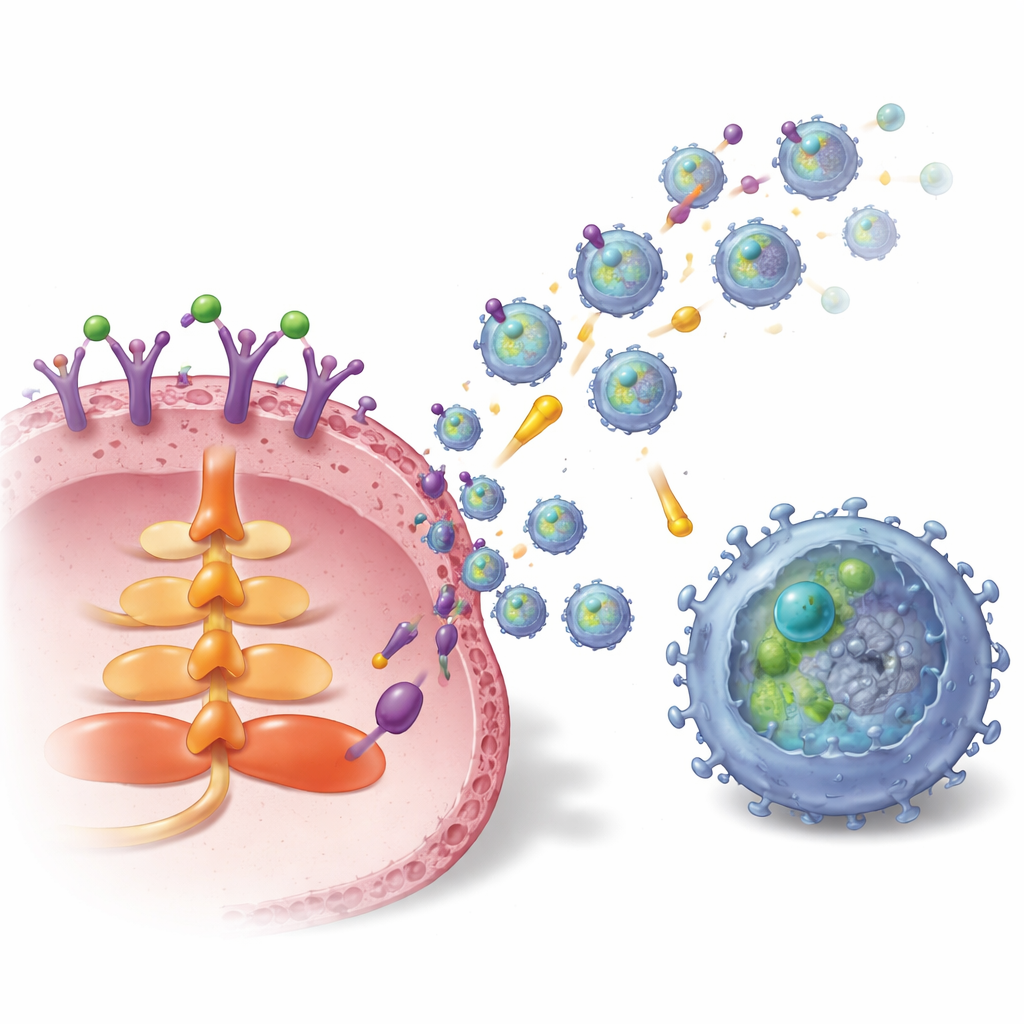
How the Tumor Loads and Launches These Vesicles
The study then mapped the internal wiring that connects S1P to exosome production and TGFBR2 loading. Activation of a specific S1P receptor on breast cancer cells, called S1P1, switched on the AKT signaling pathway and a trafficking protein named Rab27a, both of which are crucial for exosome release. At the same time, S1P1 boosted the amount and stability of TGFBR2 inside cancer cells. It did this in two ways: first by turning on a transcription factor, LEF1, that increases TGFBR2 gene expression, and second by raising levels of a deubiquitinase enzyme, USP8, through an ERK–CREB1 signaling route. USP8 slows the breakdown of TGFBR2, allowing more of the receptor to accumulate and be packaged into exosomes. In breast tumor samples from patients, both S1P1 and TGFBR2 were higher in tumor tissue and in tumor-derived exosomes than in neighboring normal tissue, and higher TGFBR2 on exosomes correlated with more exhausted CD8 T cells.
What This Means for Patients
To a non-specialist, the core message is that certain breast cancers create a chemical bath rich in S1P that instructs tumor cells to pump out tiny vesicles loaded with a receptor, TGFBR2, that turns off CD8 T cells. These vesicles act like stealth packages that reprogram nearby killer cells to become sluggish and tolerant of the tumor. By pinpointing the S1P–S1P1–TGFBR2 chain of events, including the internal switches that control exosome release and receptor buildup, this work suggests several new drug targets. Inhibiting S1P signaling, blocking S1P1, or preventing TGFBR2 from reaching or stabilizing on exosomes could reduce CD8 T cell exhaustion and make existing immunotherapies more effective for breast cancer patients.
Citation: Bhoumick, A., Ghosh, A., Singh, A. et al. Sphingosine-1-phosphate promotes CD8 T cell exhaustion in breast cancer via exosomal transfer of TGFBR2. Cell Death Dis 17, 278 (2026). https://doi.org/10.1038/s41419-026-08523-5
Keywords: breast cancer immunotherapy, CD8 T cell exhaustion, sphingosine-1-phosphate, exosomes, TGFBR2 signaling