Clear Sky Science · en
β-catenin mutation reprograms ketone body metabolism to drive hepatocellular carcinoma metastasis and resistance to ketogenic therapy via transcriptional activation of OXCT1
Why this matters for cancer and diet
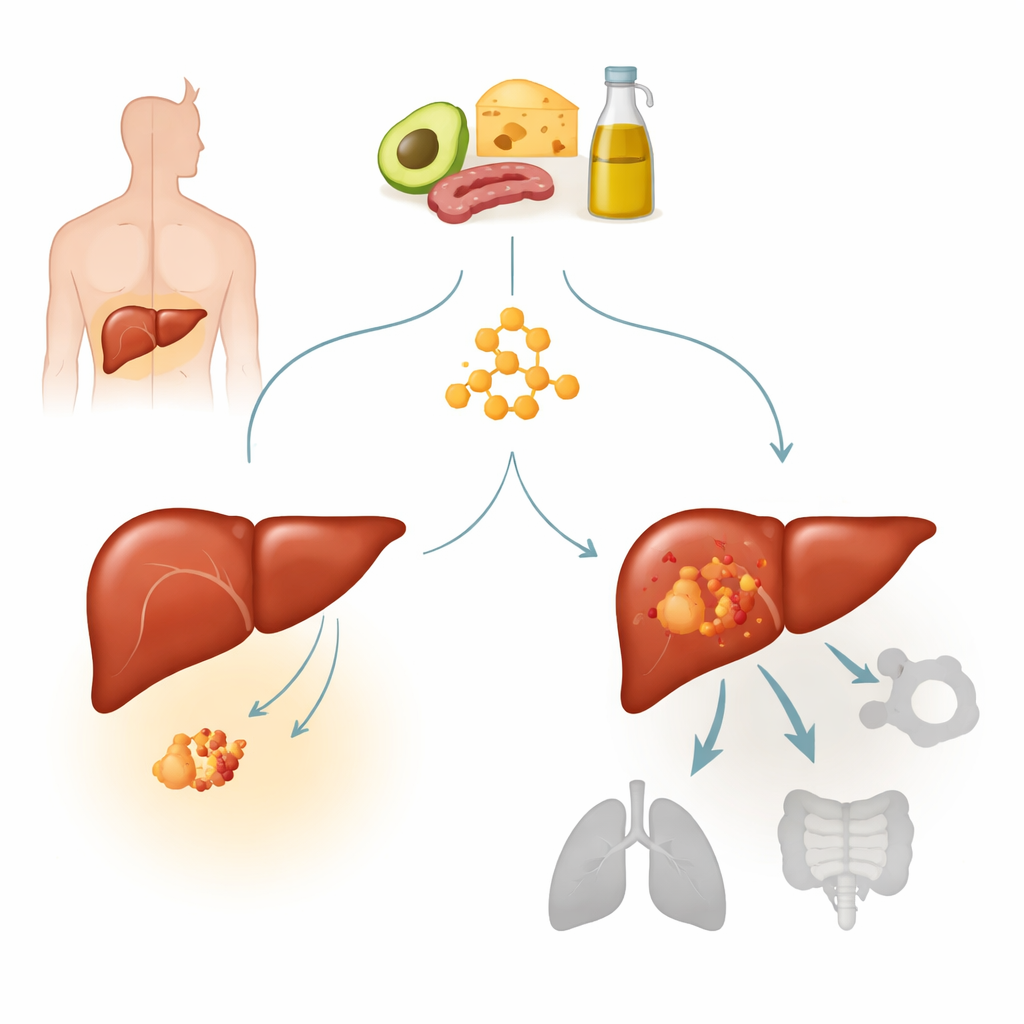
Ketogenic diets, which are high in fat and very low in carbohydrates, are being explored as a way to “starve” tumors by changing what fuel the body uses. This study shows that for an important subgroup of liver cancer, a common genetic change lets tumor cells hijack ketone bodies as an energy and growth source. Instead of being weakened by a ketogenic diet, these cancers may actually become harder to treat and more likely to spread, highlighting why cancer therapies based on diet must be matched to each tumor’s biology.
A closer look at liver cancer and a key mutation
Hepatocellular carcinoma is the most common form of liver cancer and a leading cause of cancer death worldwide. Many of these tumors carry activating mutations in a gene called beta-catenin, which normally helps control how cells grow and respond to signals. One hotspot mutation, named S33Y, keeps the beta-catenin pathway permanently switched on and defines a distinct subtype of liver cancer. The authors had previously found that tumors with strong ketone body metabolism seemed less responsive to ketogenic diets. Here, they focused on beta-catenin–mutant tumors to ask whether this mutation makes liver cancers intrinsically resistant to ketogenic therapy and, if so, how.
When a “low sugar” diet fails to slow tumors
Using human liver cancer cells grown in mice, the researchers compared tumors with and without the S33Y beta-catenin mutation under either a normal diet or a ketogenic diet. In tumors without the mutation, the ketogenic diet clearly slowed growth: tumors were smaller and lighter, even though the animals maintained body weight and showed the expected rise in blood ketones and drop in blood sugar. In sharp contrast, tumors driven by the beta-catenin mutation grew aggressively under both diets and showed no benefit from the ketogenic regimen. The team observed a similar pattern in patient-derived liver tumor grafts: ketogenic feeding shrank beta-catenin–normal tumors but did not curb growth of beta-catenin–mutant tumors, indicating true resistance rather than a model-specific effect.
How cancer cells turn ketones into growth fuel
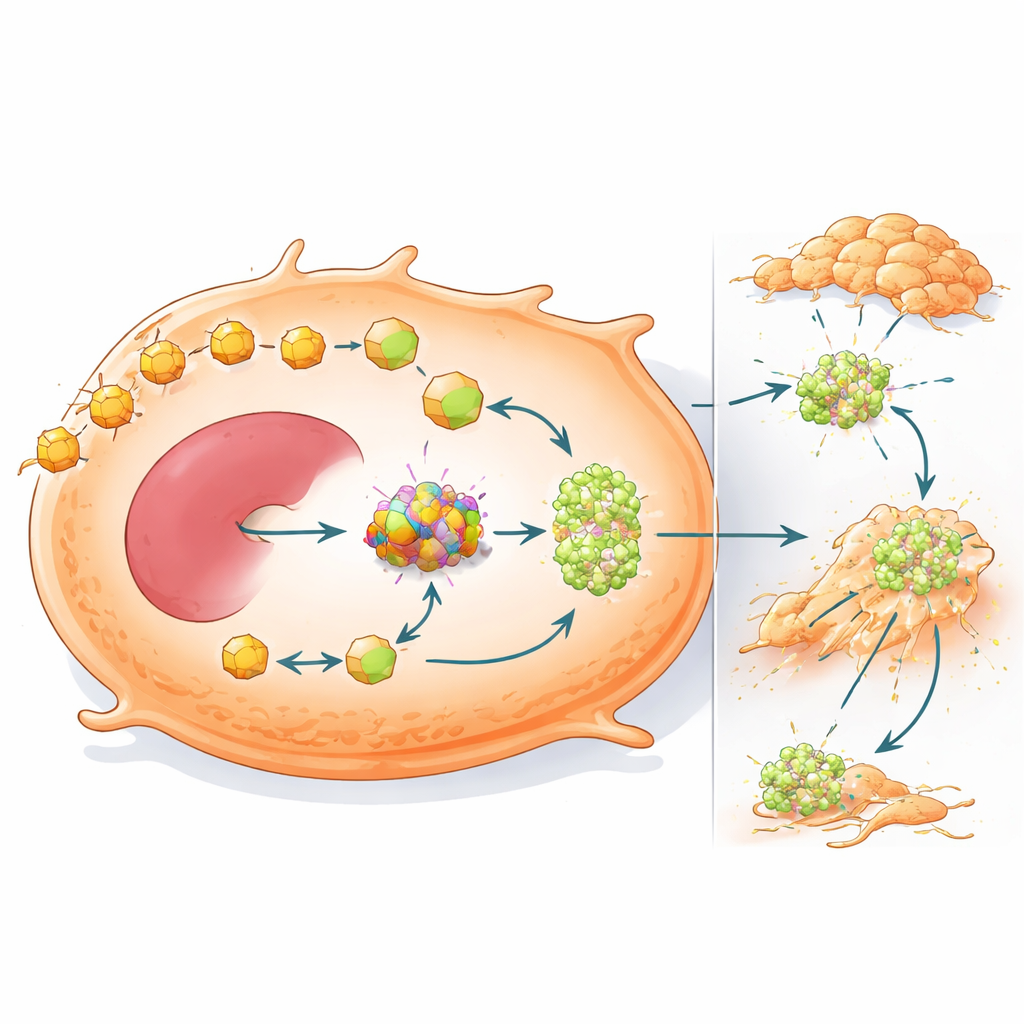
To understand this resistance, the authors examined how mutated beta-catenin reshapes ketone handling inside cancer cells. They focused on OXCT1, the main enzyme that lets cells break down ketone bodies—a process normally minimal in healthy liver tissue. Beta-catenin–mutant cells strongly boosted OXCT1 levels and other ketone-related enzymes, and metabolic tracing with labeled ketone bodies showed that these cells efficiently converted ketones into glutamate, a key building block and energy source. Detailed molecular experiments revealed that mutant beta-catenin teams up with a partner protein, LEF1, to bind the OXCT1 gene’s control region and switch it on. Blocking OXCT1 in mutant tumors reduced glutamate production and removed their resistance to the ketogenic diet, indicating that this enzyme is a critical metabolic “switch.”
Rewired fuel use also drives cancer spread
The study further shows that OXCT1 does more than help tumors survive a low-carbohydrate environment—it also makes them more prone to spread. When OXCT1 was raised in liver cancer cells, they migrated and invaded surrounding tissue more aggressively in lab tests and formed more metastatic nodules in the livers of mice. Gene analyses linked high OXCT1 to activation of a signaling protein called STAT3 and to an epithelial–mesenchymal transition program, in which cells loosen their attachments and gain a more mobile, invasive state. In animals, beta-catenin–mutant tumors had strong STAT3 activation and extensive metastasis, but knocking down OXCT1 largely reversed these changes and reduced both liver and lung colonization.
What this means for ketogenic therapy
In everyday terms, this work shows that some liver cancers carrying a common beta-catenin mutation can flip ketogenic therapy on its head. Instead of being deprived of fuel when carbohydrates are scarce, these tumors activate OXCT1 to burn ketone bodies, turn them into growth-supporting molecules, and engage pathways that help them spread. As a result, ketogenic diets alone may be risky or ineffective for patients with this tumor subtype. The findings suggest that testing tumors for beta-catenin and OXCT1 status, and developing drugs that block OXCT1, could make diet-based strategies safer and more effective by preventing cancers from turning an intended therapy into an advantage.
Citation: Li, H., Qian, L., Ji, Y. et al. β-catenin mutation reprograms ketone body metabolism to drive hepatocellular carcinoma metastasis and resistance to ketogenic therapy via transcriptional activation of OXCT1. Cell Death Dis 17, 301 (2026). https://doi.org/10.1038/s41419-026-08457-y
Keywords: ketogenic diet, liver cancer, beta-catenin mutation, tumor metabolism, OXCT1