Clear Sky Science · en
Impact of subanesthetic ketamine delivered via AmyloLipid nanovesicle (ALN)-based intranasal system on biobehavioral responses in an animal model of PTSD
Helping the Brain Heal from Trauma
Post-traumatic stress disorder (PTSD) can linger for years, leaving people trapped in cycles of fear, anxiety, and intrusive memories. Many current treatments work only partially or very slowly. This study explores a new way to deliver a well-known drug, ketamine, directly to the brain through the nose using tiny fat-and-starch bubbles. The hope is to boost benefit, cut side effects, and better support the brain’s own ability to adapt and recover after trauma.
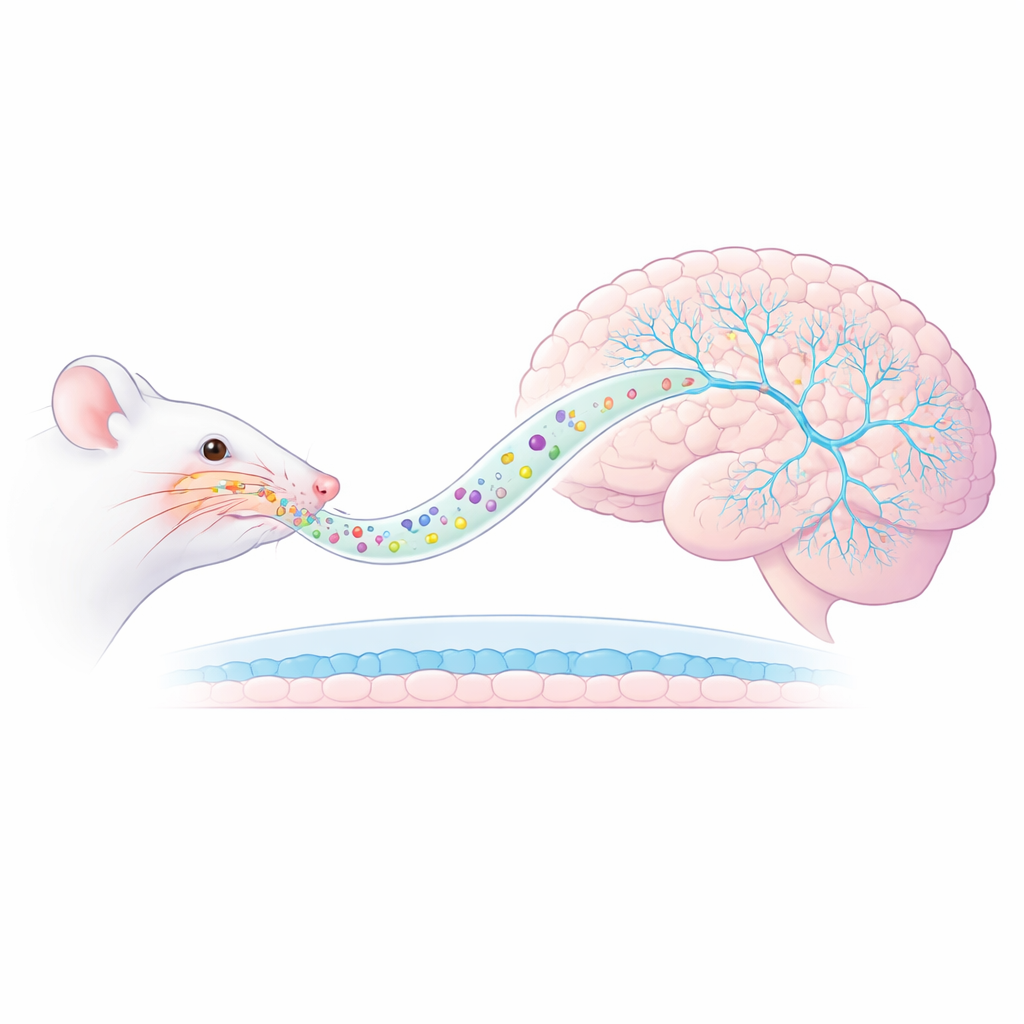
A New Nose-to-Brain Pathway
The researchers focused on a rat model of PTSD in which animals are exposed to a predator scent, triggering long-lasting anxiety and exaggerated responses to reminders of the threat. Instead of giving ketamine by injection, they packaged it inside AmyloLipid nanovesicles—nanometer-scale droplets with a fatty core wrapped in a modified starch shell. When sprayed into the nose, these particles are designed to travel along nerve pathways directly into the brain, skirting the usual blood–brain barrier that limits drug entry. Earlier measurements showed that this approach greatly increased ketamine levels in brain tissue while keeping blood levels low, suggesting more targeted delivery and potentially fewer whole-body side effects.
Testing Stress, Fear, and Startle
After the traumatic exposure, rats received intranasal treatments three times a week for two weeks: different doses of ketamine-loaded nanovesicles, empty nanovesicles, plain ketamine solution, or saline. The team then measured how anxious the animals seemed in an elevated maze, how strongly they startled at loud sounds, and how much they froze when re-exposed to a harmless reminder of the predator scent. They also sorted individual rats into groups that showed extreme, partial, or minimal disturbance, mimicking how people differ in their responses to trauma. Low-dose ketamine in nanovesicles—especially 0.6 milligrams per kilogram—stood out: it lowered anxiety-like behavior, reduced exaggerated startle responses, and cut the proportion of animals with strong PTSD-like reactions by nearly half compared with those given empty particles. In contrast, the highest dose increased freezing and anxiety, underscoring that more drug is not always better.
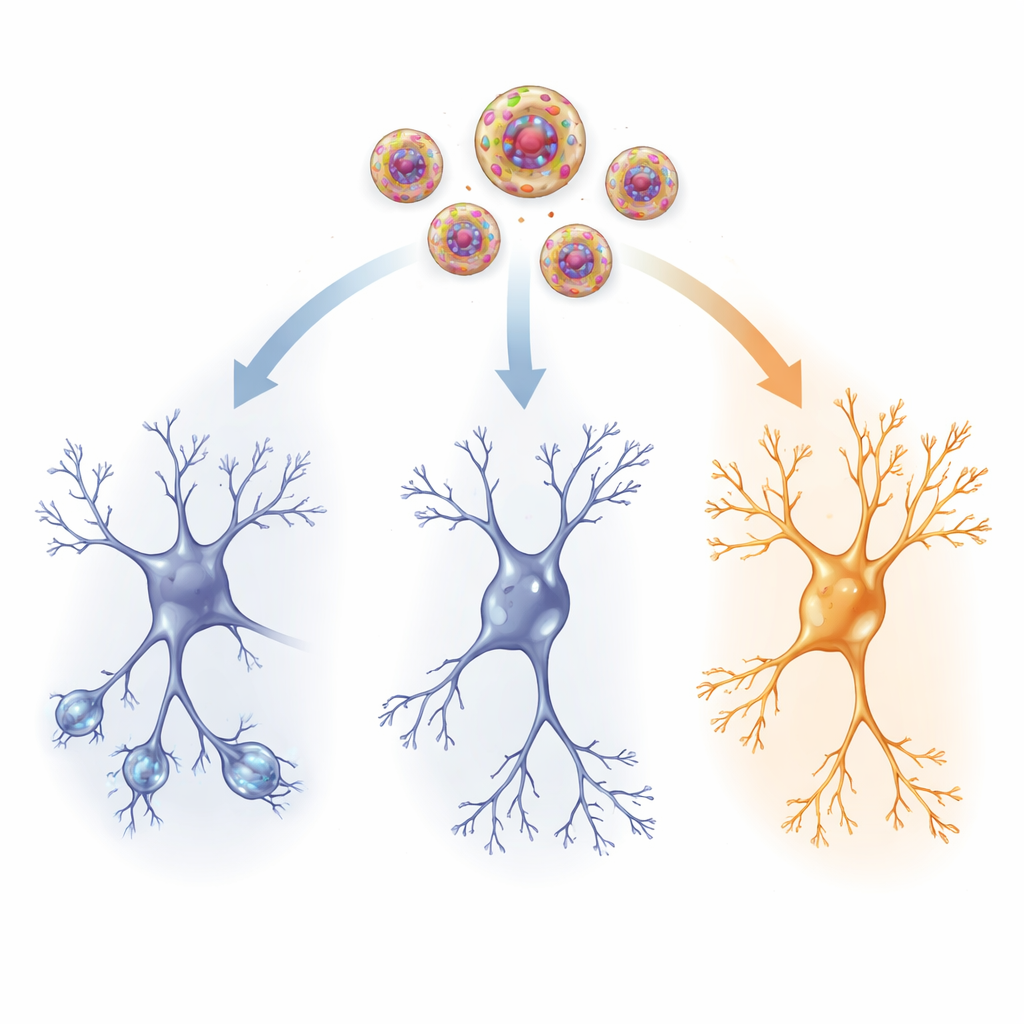
Inside the Brain’s Wiring
To see what was changing physically, the researchers examined a key memory region, the hippocampus, and a stress-control hub, the paraventricular nucleus of the hypothalamus. In traumatized rats that received no active drug, hippocampal nerve cells had fewer and shorter branches and fewer tiny spines where synapses form—signs of weakened connections. Levels of brain-derived neurotrophic factor (BDNF), a growth-supporting protein, and neuropeptide Y (NPY), a molecule linked to stress resilience, were also reduced. At the same time, there was an increase in HCN1 channels, proteins in the cell membrane that shape how easily neurons fire. Together, these shifts point to a brain network that is less flexible and less responsive after trauma.
Restoring Balance in Nerve Cells
Low-dose ketamine delivered via nanovesicles reversed many of these damage signs. Neurons in treated rats regained longer and more complex branches and more spines, especially in the uppermost layer of the hippocampus where distant inputs arrive. BDNF and NPY levels climbed back toward normal in both the hippocampus and the stress-control region, suggesting renewed support for cell survival, growth, and resilience. At the same time, the abnormal surge in HCN1 channels subsided, helping to restore a healthy level of electrical responsiveness. These structural and chemical changes closely matched the behavioral improvements, implying that targeted ketamine delivery helps the traumatized brain rewire itself in a more adaptive way.
Why Dose and Delivery Matter
Comparing nanovesicle-based ketamine with ordinary ketamine solution highlighted the importance of how and how much drug is given. At low doses, the nose-to-brain system outperformed standard ketamine in easing anxiety-like behavior, startle, and freezing. At the highest dose, however, ketamine in nanovesicles appeared to worsen stress responses, likely because more drug escaped into the bloodstream, was transformed into active breakdown products, and reached the brain by less targeted routes. This pattern suggests there is a narrow “sweet spot” where direct brain delivery at low dose maximizes helpful effects while minimizing unwanted ones.
What This Could Mean for Future Care
In simple terms, this work shows that gently sending small amounts of ketamine straight from the nose to the brain can make traumatized animals calmer and more resilient, while helping their brain cells regrow connections and regain healthy activity. Although the study was in rats, it points toward a strategy for treating PTSD that uses smart carriers and careful dosing rather than just higher amounts of drug. By tuning HCN1 channels and boosting growth and resilience molecules such as BDNF and NPY, intranasal ketamine in AmyloLipid nanovesicles may one day offer a more precise, longer-lasting, and safer way to help the brain heal from severe stress.
Citation: Levi, G., Sintov, A.C., Zohar, J. et al. Impact of subanesthetic ketamine delivered via AmyloLipid nanovesicle (ALN)-based intranasal system on biobehavioral responses in an animal model of PTSD. Transl Psychiatry 16, 230 (2026). https://doi.org/10.1038/s41398-026-03979-7
Keywords: PTSD, ketamine, intranasal delivery, nanoparticles, neuroplasticity