Clear Sky Science · en
mTOR-driven integrin β4-enriched extracellular vesicles from lenvatinib-resistant hepatocellular carcinoma fuel lung metastasis via fibroblast-niche formation
Why this research matters
Liver cancer is one of the deadliest cancers worldwide, and even powerful new drugs often lose effectiveness as tumors adapt. This study explores how liver tumors that have become resistant to the drug lenvatinib may actually become better at spreading to the lungs. By uncovering the hidden “messages” these resistant tumors send to distant organs, the researchers identify a weak point that could be targeted to both slow metastasis and restore drug sensitivity.
Drug resistance with a dangerous side effect
Lenvatinib is a frontline pill for advanced liver cancer that works by cutting off blood supply and blocking growth signals. Yet many patients initially respond only to later relapse as the tumor finds ways around the blockade. In mouse models, the authors compared original liver cancer cells with versions that had acquired resistance to lenvatinib. They found that resistant tumors not only survived the drug but also grew faster and produced far more lung metastases, shortening survival. This suggested that resistance was tied to a more aggressive, spreading behavior rather than merely a loss of drug effect.
Tiny messengers that prepare the lungs
To understand how resistant tumors gain this metastatic edge, the team focused on extracellular vesicles—tiny bubbles shed by cells that carry proteins and other cargo through the bloodstream. Using imaging and particle tracking, they showed that lenvatinib-resistant liver cancer cells released significantly more of these vesicles than their non-resistant counterparts, and that the vesicles preferentially accumulated in the lungs of mice. When healthy mice were “pre-treated” with vesicles from resistant tumors, later injections of liver cancer cells produced many more lung tumors, demonstrating that the vesicles actively prepare a welcoming “soil” for cancer seeds. 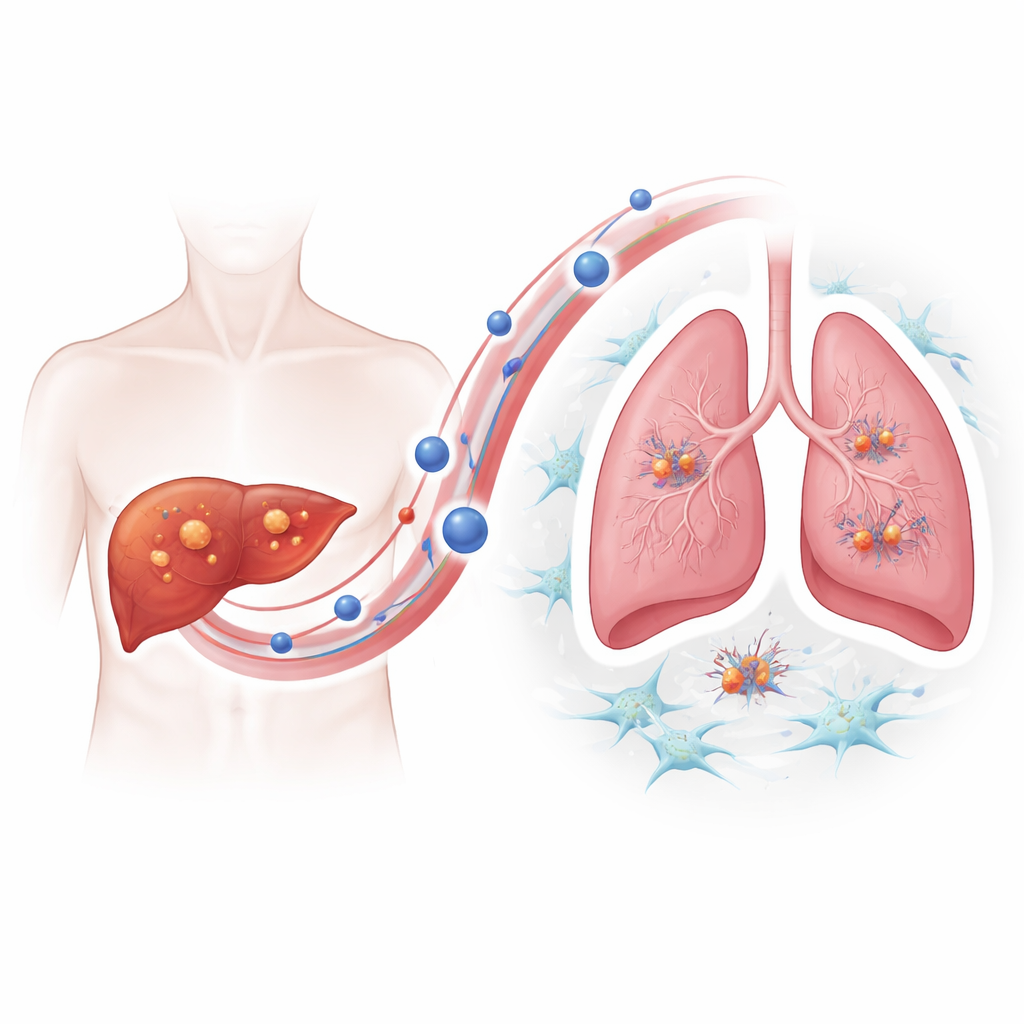
A molecular handshake that wakes up lung helpers
Proteomic analysis revealed that vesicles from resistant tumors were enriched in a surface protein called integrin beta-4. Integrins help cells latch onto their surroundings, and here integrin beta-4 on vesicles acted like a molecular handshake with laminin, a major component of lung tissue. The vesicles were taken up most efficiently by lung fibroblasts—support cells that can remodel tissue. Once activated by these vesicles, fibroblasts adopted a more muscular, contractile state and began secreting inflammatory molecules and growth factors. This process depended on a signaling cascade inside fibroblasts known as the PI3K–AKT–p65 pathway; blocking key steps in that cascade dampened fibroblast activation and reduced the pro-metastatic signals they produced. Vesicles lacking integrin beta-4 largely lost their ability to drive lung colonization.
An internal switch that boosts vesicle release
Why do resistant tumor cells release so many vesicles in the first place? The authors traced this to hyperactivation of a master growth regulator called mTOR, which also controls a cellular recycling system known as autophagy. In resistant cells, active mTOR impaired the normal degradation of multivesicular bodies—internal compartments that can either be broken down or fuse with the cell surface to release vesicles. When degradation was blocked, these compartments were shunted toward secretion, greatly increasing vesicle output. Treating resistant cells with rapamycin, a classic mTOR inhibitor, restored autophagic breakdown of these compartments, reduced vesicle release, and lowered the amount of integrin beta-4 carried by remaining vesicles. 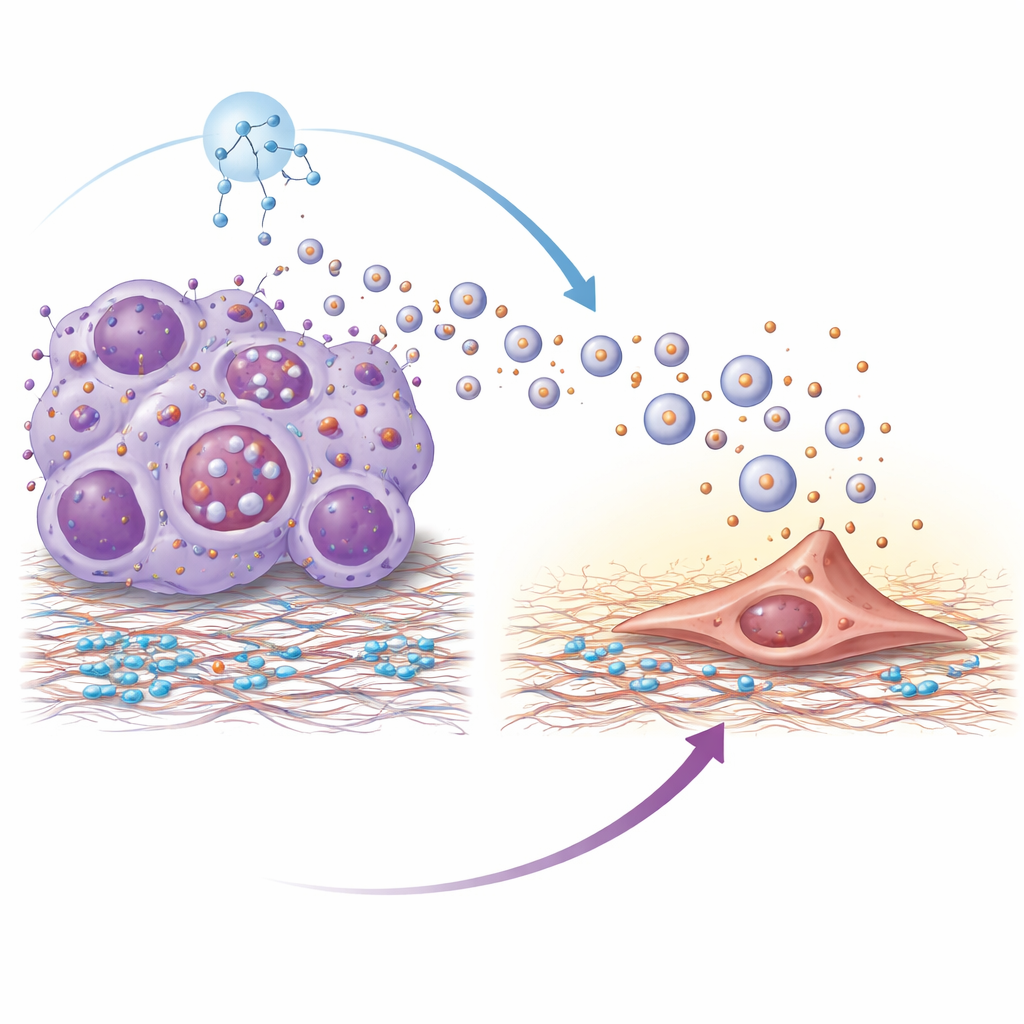
A combination strategy with clinical clues
In mice, combining rapamycin with lenvatinib shrank resistant tumors more effectively, reduced lung metastases, and extended survival compared with either drug alone. Vesicles from rapamycin-treated resistant cells were far less capable of activating lung fibroblasts or fostering a pre-metastatic lung niche. In blood samples from liver cancer patients, higher levels of integrin beta-4 on circulating vesicles were linked to poor response to lenvatinib, more frequent lung metastasis, and worse survival, suggesting that this marker could help flag patients at risk of aggressive spread.
What this means for patients
To a lay observer, this work shows that drug-resistant liver tumors do more than just ignore treatment—they send out fleets of microscopic packages that “soften up” the lungs, turning local support cells into silent accomplices. By identifying mTOR as the internal switch that boosts these dangerous messages, and integrin beta-4 as a key address label guiding them, the study points to two practical opportunities: using vesicle integrin beta-4 levels in blood as an early warning signal, and combining mTOR-blocking drugs like rapamycin with lenvatinib to both restore drug sensitivity and curb lung metastasis.
Citation: Zou, T., Wang, X., Sun, H. et al. mTOR-driven integrin β4-enriched extracellular vesicles from lenvatinib-resistant hepatocellular carcinoma fuel lung metastasis via fibroblast-niche formation. Sig Transduct Target Ther 11, 138 (2026). https://doi.org/10.1038/s41392-026-02625-4
Keywords: hepatocellular carcinoma, drug resistance, extracellular vesicles, lung metastasis, mTOR signaling