Clear Sky Science · en
The evolution to hepta-refractory myeloma involves sequential loss of CD38, BCMA and GPRC5D
When Cancer Outsmarts Even the Newest Drugs
Multiple myeloma, a cancer of plasma cells in the bone marrow, has benefited from a wave of new medicines, including sophisticated immune-based treatments. Yet some patients now reach a stage where their disease has escaped every major drug type available. This study looks closely at that end stage—called “hepta‑refractory” myeloma—to understand how the cancer evolves to evade so many therapies and to explore how doctors might still find useful options.
A New Extreme Stage of Myeloma
The researchers define hepta‑refractory myeloma as disease that no longer responds to seven key treatment categories: two kinds of immune‑modulating pills, two types of proteasome‑blocking drugs, an antibody against a surface marker called CD38, and two newer immune therapies aimed at targets known as BCMA and GPRC5D. In a group of 37 such patients treated at several centers, survival was poor despite intensive care. After reaching this stage, patients typically lived about 13 months on average, and each new line of “salvage” therapy held the disease at bay for only about three months. Still, outcomes varied: a few individuals did noticeably better, hinting that the underlying biology is not the same in every case.
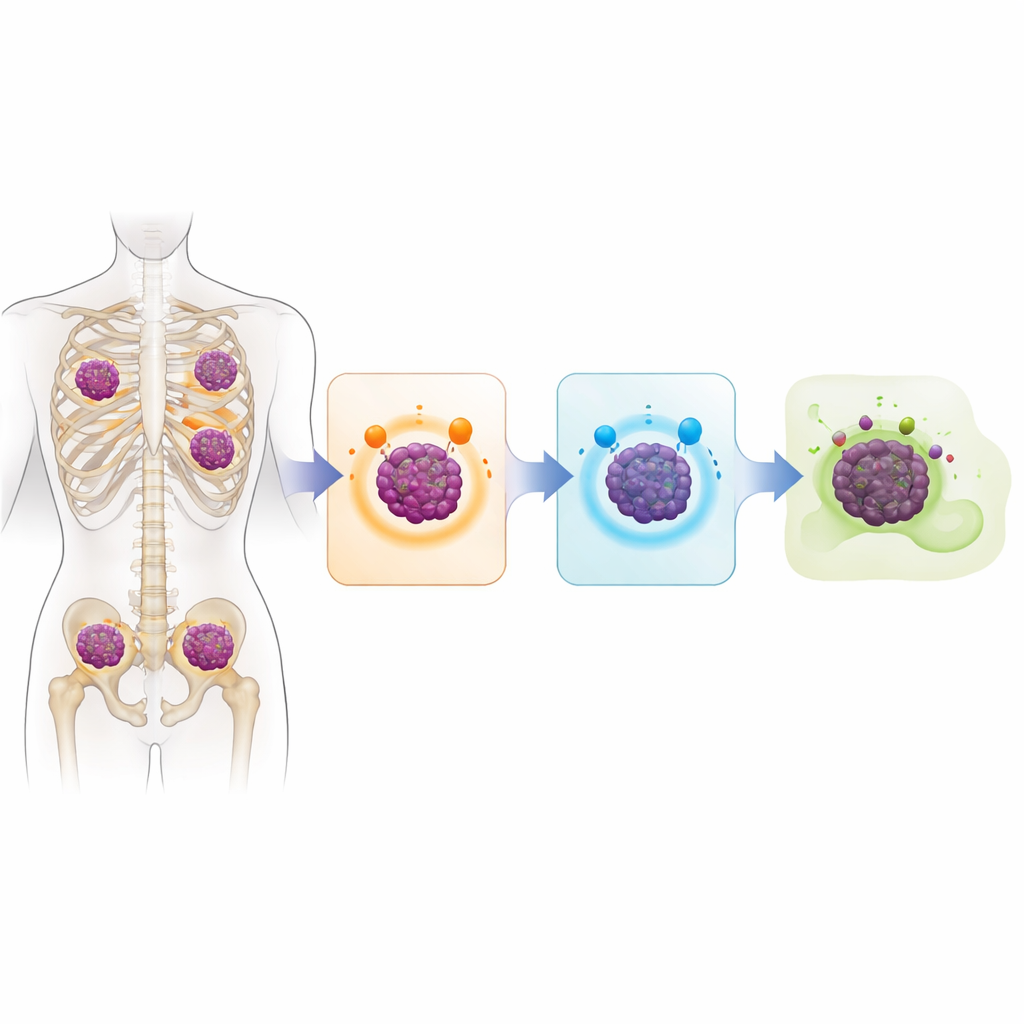
A Cancer Genome Full of Warning Signs
To see what set these cancers apart, the team used whole genome sequencing, a technique that reads essentially all of a tumor’s DNA. In 17 patients with available samples, they found highly damaged and rearranged genomes with many hits to classic safeguard genes that usually restrain cell growth or trigger cell death. Common problem areas included TP53 and CDKN2C, among others often linked to especially aggressive disease. Many of these severe changes appeared in tumors that had spread outside the bone marrow into soft tissues, matching the patients’ fast‑moving illness. The DNA patterns also carried telltale “scars” of past treatments—such as changes associated with the chemotherapy drug melphalan—showing that years of therapy had shaped the cancer’s evolution.
How Tumor Cells Shed Their Targets
The central question was how myeloma cells become invisible to modern immune‑based drugs that are designed to recognize specific molecules on the cell surface. The study shows that, in many cases, tumor cells simply stop making these molecules. In about 40 percent of sequenced patients, the gene for BCMA was knocked out on both copies of the chromosome, often by outright deletion. Roughly a third had similar double hits affecting GPRC5D, usually through a mix of deletions and small damaging mutations. Some patients lost CD38 as well. Nearly one‑third of the analyzed group had lost both BCMA and GPRC5D, meaning they no longer displayed the very targets needed for several of today’s most powerful therapies to work.
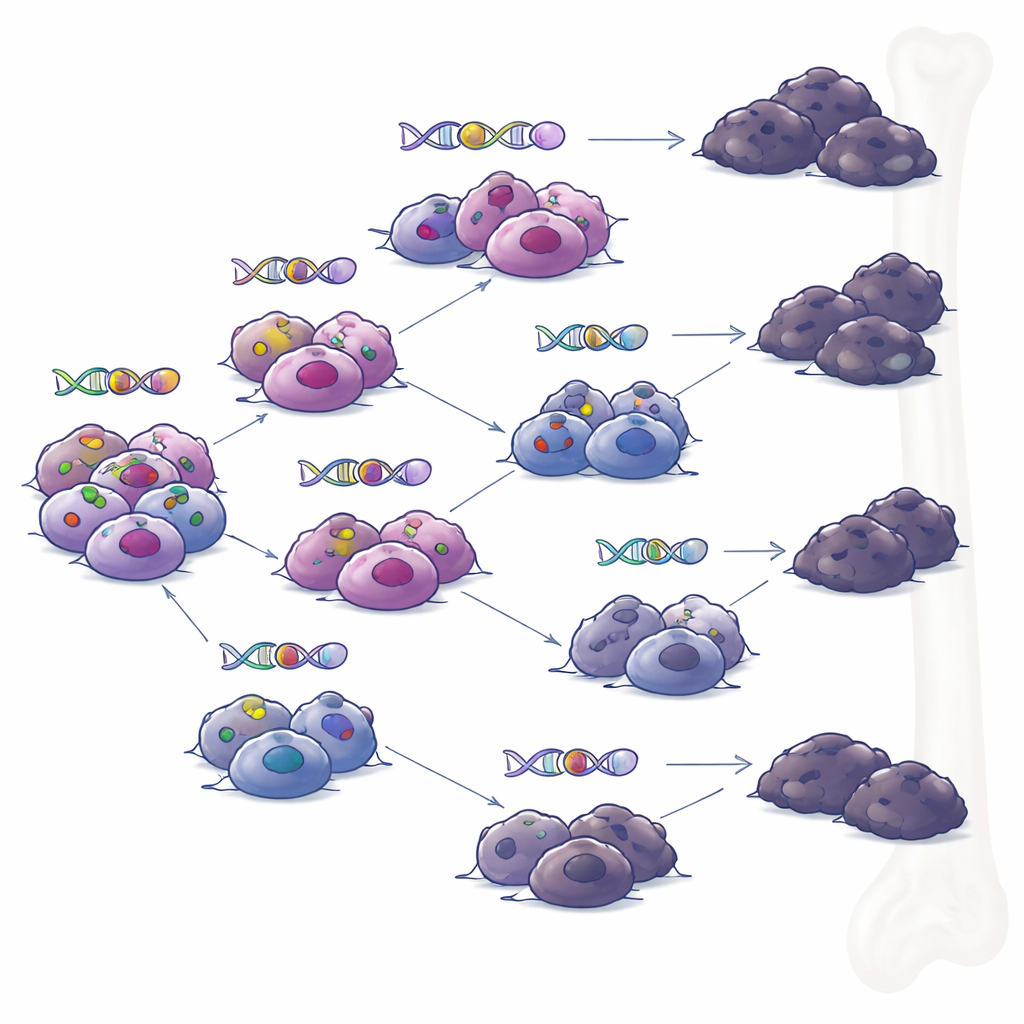
Branching Paths Rather Than a Single Rogue Clone
When the team examined samples taken from the same patients at different times, they reconstructed the tumors’ family trees. Instead of a single dominant clone steadily accumulating changes, they saw branching evolution: several related subgroups of cells that diverged and acquired different sets of mutations over time. Under pressure from targeted immune therapies, multiple branches independently developed ways to lose BCMA or GPRC5D. This pattern suggests that even when treatment appears to produce a deep remission, small pockets of genetically diverse cells can persist and later spark relapse, each with its own combination of resistance tricks.
Using Deeper Testing to Guide Second Chances
Because gene sequencing only captures DNA, the researchers also used immunohistochemistry, a staining method that shows whether the BCMA protein is actually present on tumor cells. Every tumor with both BCMA gene copies lost showed complete loss of BCMA protein, but some tumors lacking obvious gene damage also had little or no BCMA on their surface, implying additional, more subtle resistance mechanisms. Crucially, in a small set of patients who had previously received BCMA‑targeting treatment, those whose tumors still carried the BCMA gene and showed at least some BCMA protein often responded again when retreated with another BCMA‑directed CAR T‑cell therapy. In contrast, patients whose tumors had fully lost BCMA did not benefit from such retreatment.
What This Means for Patients and Future Care
For people living with myeloma, this study underscores both a sobering and a hopeful message. On the one hand, some tumors can evolve into a highly drug‑resistant state by systematically discarding the very markers that advanced immune therapies rely on. On the other, detailed testing of tumor DNA and protein markers can reveal which targets remain and help avoid futile, side‑effect‑laden treatments when a target is gone. The authors argue that combining broad genomic analysis with staining‑based tests should become part of care for patients with very advanced myeloma. Such approaches could better match people to retreatment strategies that still have a chance to work, while guiding the search for new therapies that do not depend on a single, easily lost target.
Citation: Riedhammer, C., Truger, M., Lee, H. et al. The evolution to hepta-refractory myeloma involves sequential loss of CD38, BCMA and GPRC5D. Leukemia 40, 730–738 (2026). https://doi.org/10.1038/s41375-026-02889-3
Keywords: multiple myeloma, treatment resistance, immunotherapy, antigen loss, genomic profiling