Clear Sky Science · en
Orai1 acts as a novel Ca2+ signal switch, balancing erythropoiesis through KLF1 regulation
Why a molecular switch for red blood cells matters
Every second, your body produces millions of new red blood cells to carry oxygen. For transfusions, blood shortages, and future cell therapies, scientists are eager to grow these cells efficiently in the lab. Yet the final steps of red blood cell maturation are surprisingly hard to control. This study reveals a hidden molecular “dimmer switch” that decides whether young blood cells keep developing or pause, opening new ways to boost lab-made blood production and better understand certain anemias. 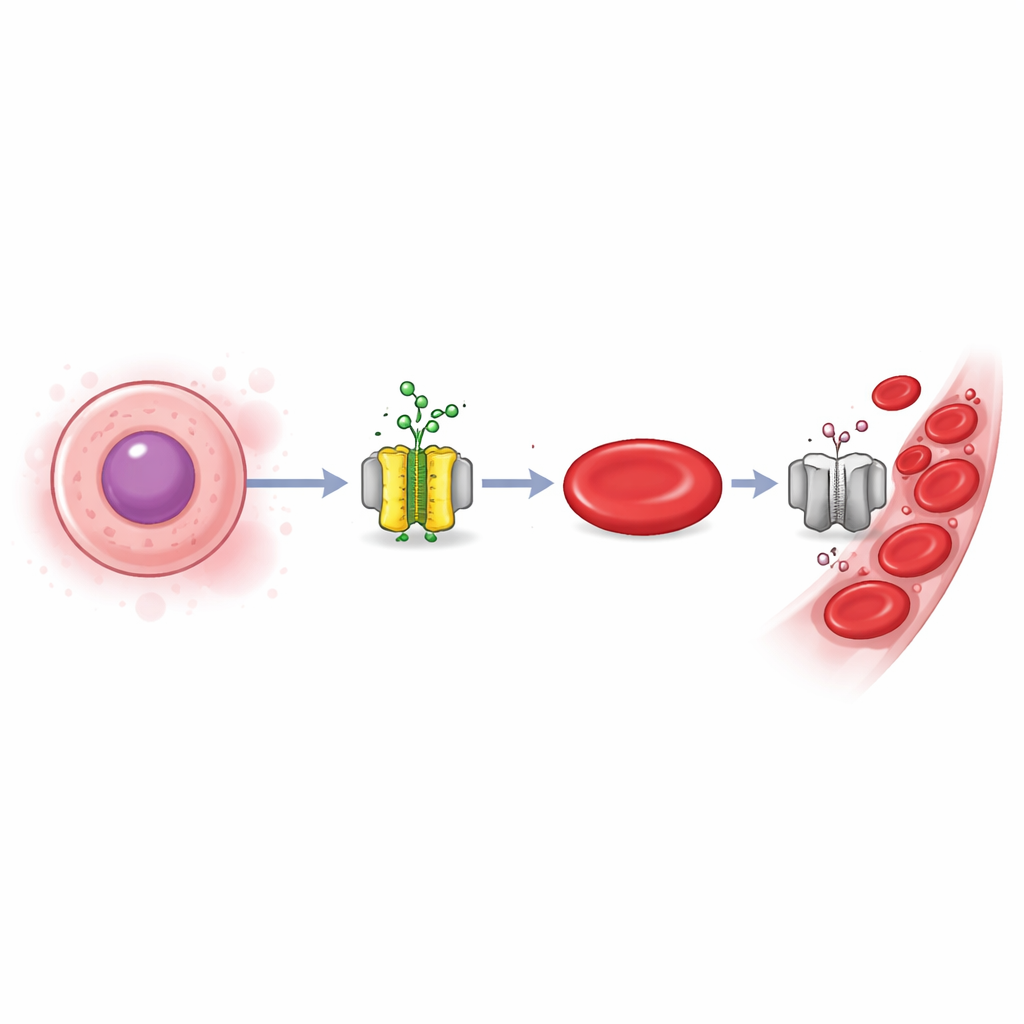
The final steps of red blood cell growth
Red blood cells are born from immature precursors that gradually shrink, fill with hemoglobin, and finally spit out their nucleus. This last stretch, called terminal maturation, is guided by the hormone erythropoietin, which tells cells when to survive, divide, or specialize. A key gene controller named KLF1 acts like a master program that turns on red blood cell–specific genes, including those for hemoglobin and enucleation. However, erythropoietin can both promote and slow down maturation, and how it times KLF1 activity at different stages has been unclear.
A calcium channel that slows early maturation
The authors focused on calcium, a universal signaling ion, and a membrane channel called Orai1 that lets calcium flow into cells. Using human erythroid cell lines, cord blood–derived cells, and red blood cells grown from pluripotent stem cells, they found that young precursors express high levels of Orai1 and show strong calcium entry, whereas more mature cells display much lower Orai1 and weaker calcium signals. Blocking general protein breakdown showed that Orai1 is removed mainly through an autophagy–lysosome route as cells mature. Functionally, when Orai1 was disabled by a mutant form or by CRISPR–Cas9 knockout, calcium entry fell, but KLF1 levels rose. This pushed cells to express more red blood cell genes, make more globin, mature faster, and enucleate more efficiently.
How the switch works inside the cell
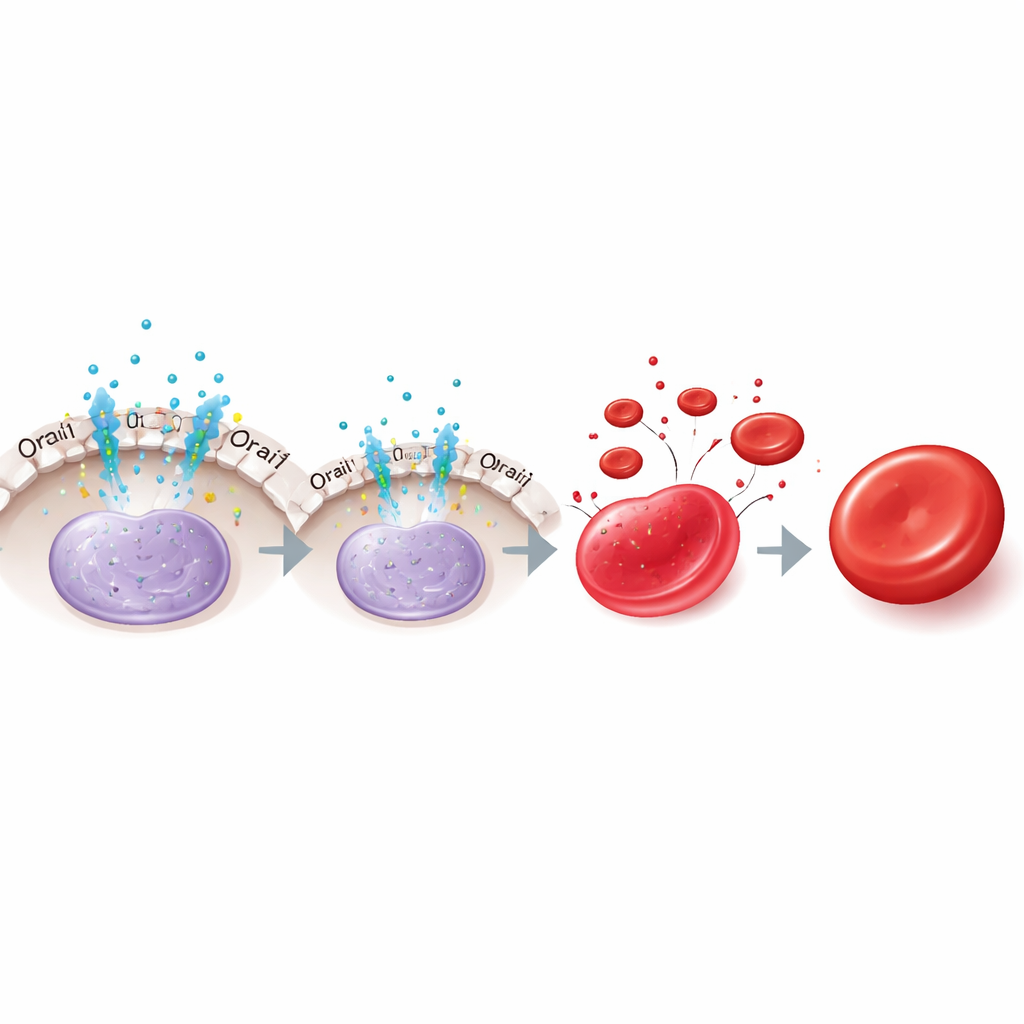
A handoff to a second hormone pathway
Interestingly, erythropoietin’s role changes over time. In early cells, removing erythropoietin or blocking Orai1 increased KLF1 and sped maturation, consistent with relief of NFAT2-mediated suppression. But in later-stage cells, taking away erythropoietin now reduced KLF1 and impaired maturation, even though Orai1 inhibition no longer had an effect. The authors traced this late support of KLF1 to another erythropoietin pathway involving the signaling protein STAT5 and its downstream partners TAL1 and DDX5, which help maintain KLF1 once the Orai1–NFAT2 brake has faded. Thus, erythropoietin first restrains KLF1 via Orai1–NFAT2, then later sustains KLF1 through STAT5 and its co-factors.
Toward better lab-grown blood
In simple terms, this work shows that Orai1 functions as a calcium-based switch that lets erythropoietin fine-tune when young red blood cell precursors should pause or proceed. When Orai1 is “on,” calcium signals activate NFAT2 to keep KLF1 and full maturation in check; when Orai1 is “off,” erythropoietin shifts to a STAT5 route that keeps KLF1 high and drives the final transformation into oxygen-carrying cells. By dialing down Orai1 or temporarily removing erythropoietin at the right stage, it may be possible to coax stem cell–derived precursors into becoming functional, enucleated red blood cells more efficiently, bringing the prospect of lab-grown blood for transfusions a step closer to reality.
Citation: Lee, Y.Y., Koh, H., Kim, J. et al. Orai1 acts as a novel Ca2+ signal switch, balancing erythropoiesis through KLF1 regulation. Exp Mol Med 58, 696–708 (2026). https://doi.org/10.1038/s12276-026-01651-0
Keywords: red blood cell maturation, calcium signaling, Orai1 channel, erythropoietin, KLF1