Clear Sky Science · en
Homogeneous image-based digital immunoassays with high error tolerance
Why tiny particles and smart cameras matter
Many of today’s medical tests either demand a full laboratory with skilled staff or offer quick but rough answers on a simple strip. This paper presents a way to get the best of both worlds: lab‑grade accuracy from a small blood sample using simple hardware, with most of the heavy lifting done by clever image analysis. By watching how tiny gold particles behave in a drop of blood and letting machine‑learning software interpret those pictures, the authors show a path toward home and clinic tests that are both easy to use and hard to fool.
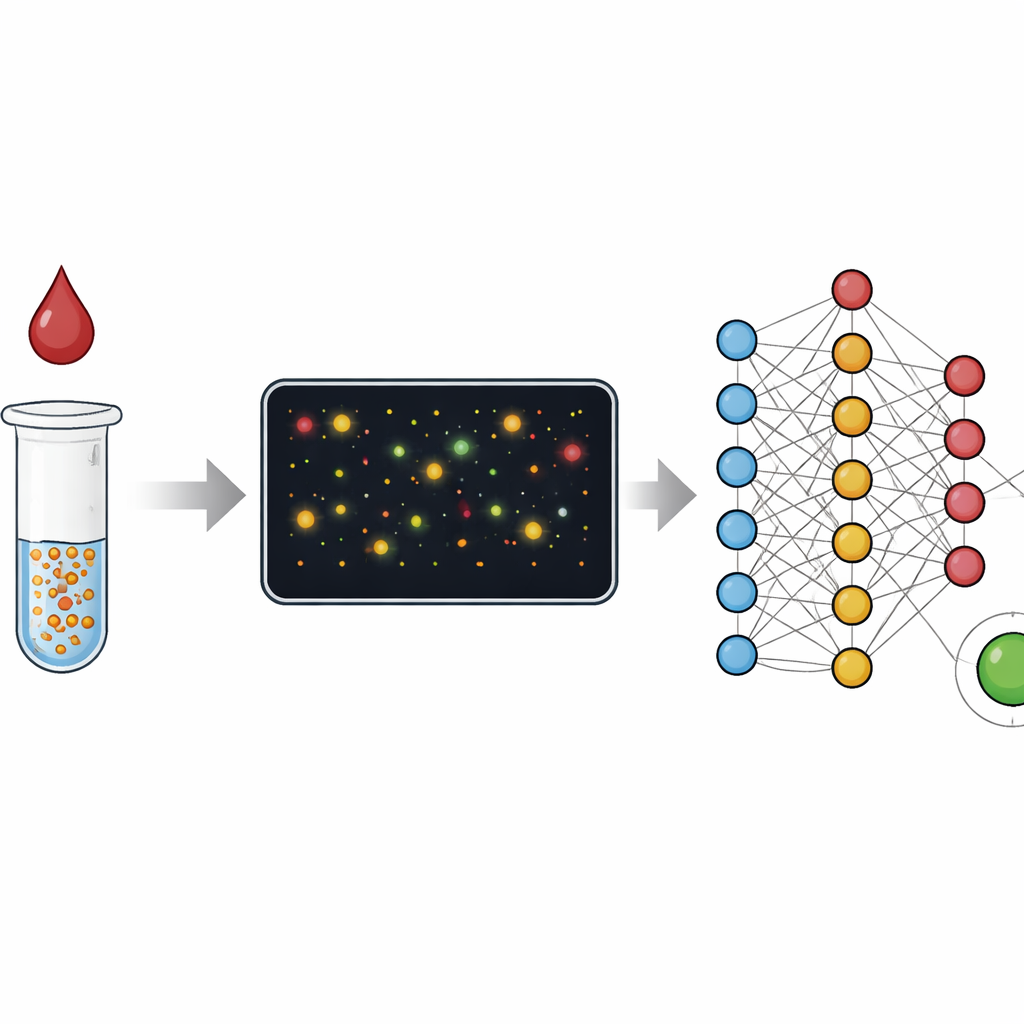
From color changes to counting particles
Traditional blood tests such as ELISA read out an average color change from millions of molecules in a well, much like judging a crowd by its overall noise level. Rapid strip tests, or lateral flow assays, work on a similar idea and trade accuracy for speed and simplicity. In this study, the researchers instead focus on individual gold nanoparticles that clump together when they find their target in a sample, such as C‑reactive protein linked to inflammation or antibodies against the virus that causes COVID‑19. Rather than measuring only the bulk color of the solution, they use dark‑field microscopy to image tiny fields full of these particles and clusters, then analyze each one. This switch from “analog” color to “digital” counting greatly widens the range of concentrations the test can measure and makes it easier to spot when something has gone wrong.
Seeing through the mess in real‑world samples
Real blood samples are messy: they contain red cells, debris, bubbles, and other sources of visual clutter that can confuse simple image‑processing rules. The team first shows that even with tuned conventional algorithms, they can extract a clean, nearly linear relationship between cluster size and C‑reactive protein levels, and they can do this directly in whole blood without spinning out the cells. To deal more systematically with artifacts, they then train a segmentation model that labels each pixel in an image as nanoparticle, cell, dust, or background. After filtering everything except the true particles, they again use cluster size as a readout. With this hybrid approach, they correctly classify COVID‑19 antibody status in stored patient sera with 96% specificity and 90% sensitivity, even though the images were collected under deliberately error‑prone conditions.
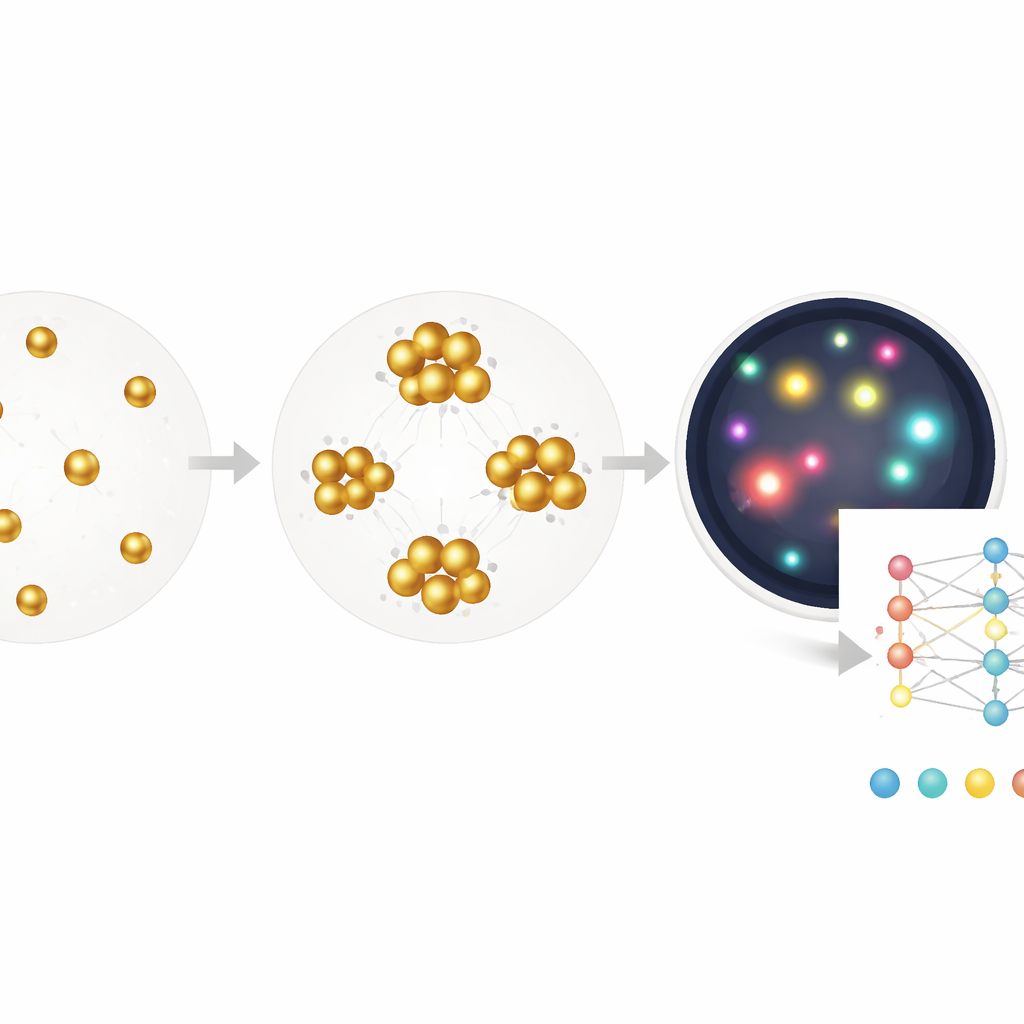
Letting the computer discover what matters
While the segmentation method helps, it still relies on human choices about which features to measure—such as how big or bright a cluster must be. The authors push further by training a deep neural network to go directly from a raw image to the concentration of anti‑viral antibodies, using only the known concentration as the label and no pixel‑level annotations. This end‑to‑end model, built on a standard ResNet architecture, effectively learns its own combination of clues: subtle shifts in color, density, cluster shape, empty spaces, and more. When tested on new samples, it estimates antibody levels over a range of more than three and a half orders of magnitude, with a detection limit approaching that of a clinical ELISA kit, yet requires just a single 30‑minute incubation and works on tiny volumes.
Designing tests that can be tuned like a microscope
Beyond demonstrating working assays, the study also asks how far this kind of digital particle counting could in principle be pushed. Using a mathematical and simulation framework, the authors show that the sensitivity of such tests is not fixed; it depends on how many particles are used, how uniform they are, and how well single and paired particles can be distinguished by brightness. By adjusting these knobs—especially the number of particles and their quality—they argue that one can, in theory, reach extremely low detectable concentrations, bounded mainly by fundamental counting statistics. Machine learning adds another boost by increasing how many particles can be confidently used and by improving discrimination between true binding events and random noise.
Toward smarter, more forgiving blood tests
Altogether, the work suggests that pairing simple optics with advanced image analysis can deliver tests that rival gold‑standard lab methods while tolerating the variability of real‑world use. Instead of building ever more precise hardware, the approach leans on the rich information already present in images and lets software sort signal from clutter. For patients and health systems, this could mean more reliable point‑of‑care and self‑administered tests for common blood markers and infectious diseases, without the need for complex instruments or expert operators.
Citation: McAffee, D.B., Hu, Q., Arnob, A. et al. Homogeneous image-based digital immunoassays with high error tolerance. npj Imaging 4, 30 (2026). https://doi.org/10.1038/s44303-026-00164-9
Keywords: digital immunoassay, nanoparticle imaging, machine learning diagnostics, point-of-care testing, SARS-CoV-2 antibodies