Clear Sky Science · en
Molecular action of NZ2114, a superior plectasin derivative
Why tiny fungal molecules matter for future antibiotics
As more bacterial infections stop responding to our best antibiotics, scientists are hunting for new ways to kill germs without triggering rapid resistance. One promising lead comes from small defensive molecules made by fungi and animals. This study digs into how one such molecule, a lab‑tuned version of the natural peptide plectasin called NZ2114, fights dangerous bacteria like Staphylococcus aureus, including hard-to-treat MRSA strains. By uncovering its step‑by‑step action on bacterial cell walls, the work points toward smarter design of next‑generation antibiotics.
A natural shield turned into a stronger weapon
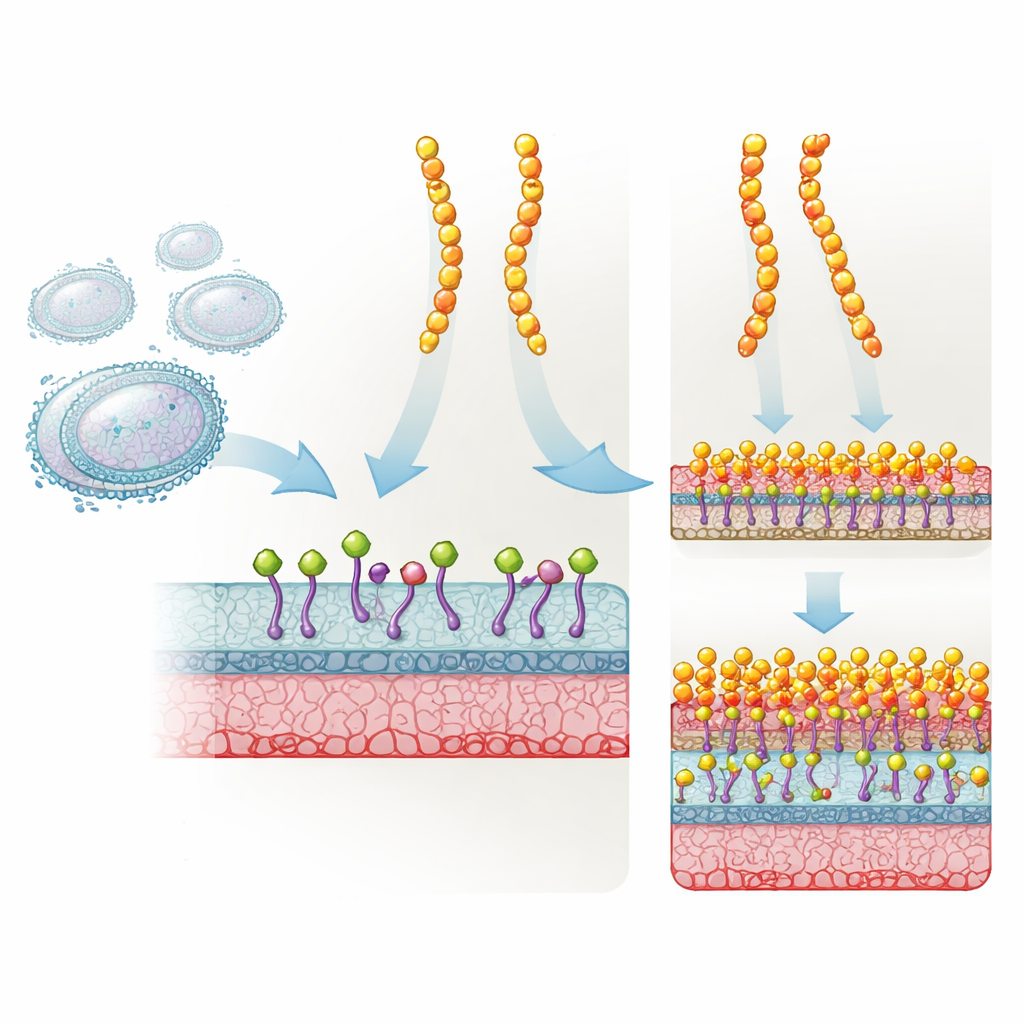
Plectasin is a short, sturdy protein made by a fungus that naturally defends against many Gram‑positive bacteria. It works by grabbing hold of Lipid II, a critical building block that bacteria use to make their cell wall. When Lipid II is locked up, the cell wall can no longer be properly built, and the bacterium eventually dies. Drug developers modified plectasin at three specific positions to create NZ2114, which proved much more effective in animal models against staphylococci and streptococci. Yet despite its success and clinical interest, no one really knew why this triple‑mutant worked so much better than the original.
Not tighter gripping, but a different way of holding on
A simple explanation had been that NZ2114 might stick more tightly to a “special” form of Lipid II found in Staphylococcus aureus, while plectasin would bind poorly to this altered target. The authors put this idea to a direct test using a battery of structural and binding experiments. They examined how both peptides interact with several versions of Lipid II, including those carrying chemical tweaks typical of staphylococci, and measured how strongly each peptide bound in different types of membranes. The surprising outcome was that both plectasin and NZ2114 bound all Lipid II variants very well, often with plectasin binding slightly more tightly. This ruled out target recognition as the main reason for NZ2114’s superior killing power.
How small changes reshape motion and teamwork
To see what really changed, the team used nuclear magnetic resonance (NMR) and computer simulations to look at the three substituted amino acids in NZ2114. These mutations subtly rewired a network of hydrogen bonds near a negatively charged patch that can attract calcium and magnesium ions. That, in turn, altered the shape and flexibility of the peptide’s N‑terminal loop and a nearby segment that contacts Lipid II. Solid‑state NMR and high‑speed atomic force microscopy then revealed how these local tweaks played out on a membrane surface. At low calcium levels, NZ2114 formed small, highly mobile clusters on the bacterial membrane, while plectasin already spread into larger, carpet‑like assemblies. When calcium was abundant, NZ2114 underwent a global rearrangement and snapped into large, ordered carpets that closely resembled those formed by plectasin.
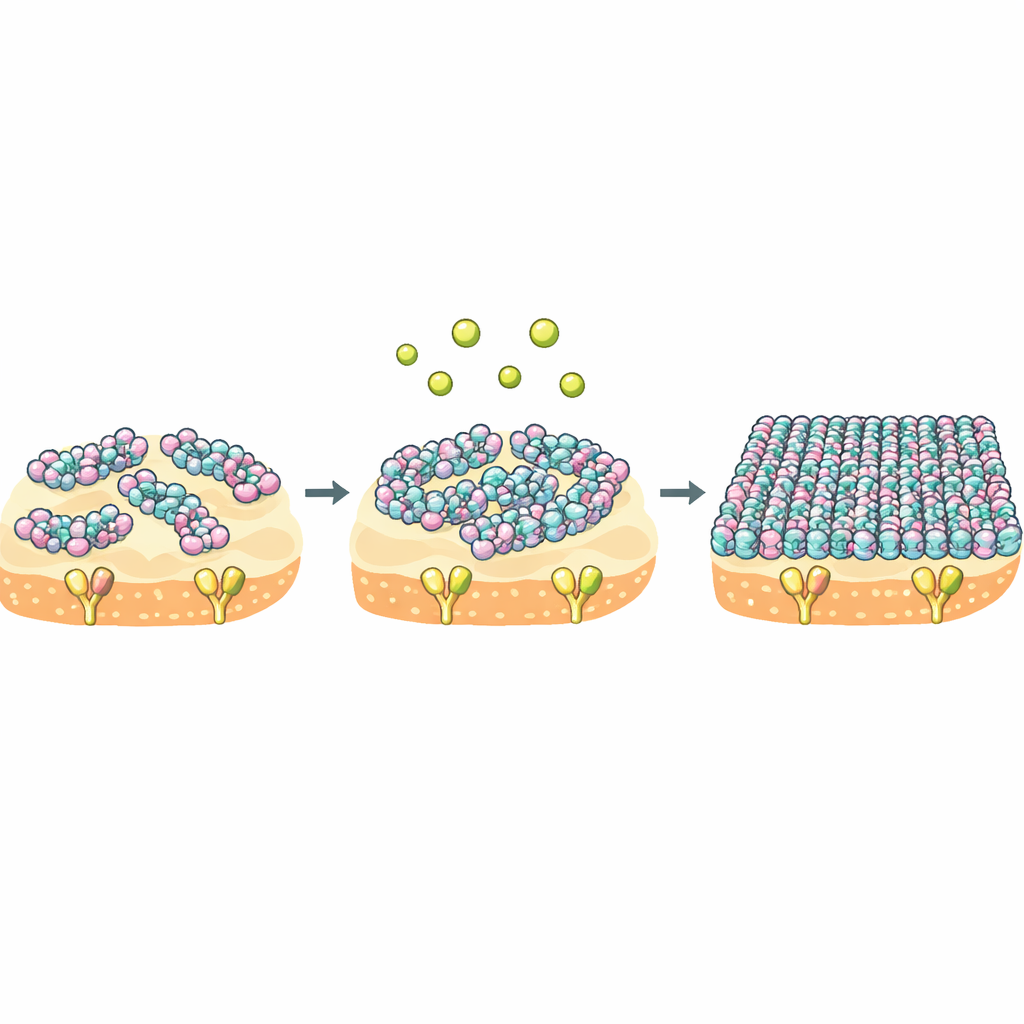
Calcium as a switch for molecular crowd behavior
The core insight is that NZ2114 and plectasin share the same target but differ in their “crowd behavior” once they reach the membrane. For NZ2114, calcium acts as a powerful switch: it shifts the peptide from a loose, dynamic state into a rigid, highly organized layer that blankets the membrane and traps Lipid II for a long time. Thermodynamic measurements show that NZ2114 pays far less of an entropy cost when binding its target, meaning the bound state remains more mobile and varied. Because binding and self‑assembly are coupled, this flexible yet calcium‑tunable behavior may allow NZ2114 to adapt its supramolecular structure to different environments on the bacterial surface, thereby enhancing its killing efficiency.
What this means for future antibiotic design
The study’s main message for non‑specialists is that NZ2114’s advantage does not come from grabbing a different lock, but from forming a more effective crowd around the same keyhole. Small, precisely placed mutations changed how the peptide responds to calcium ions and how it organizes with its neighbors on the bacterial membrane. By showing that Lipid II modifications in Staphylococcus aureus are not the culprit behind plectasin’s weaker performance, the work shifts attention toward controlling peptide shape, flexibility, charge, and ion sensitivity to tune collective behavior. These insights may guide the design of new peptide‑based antibiotics that are harder for bacteria to evade and more potent against resistant strains.
Citation: Derks, M.G.N., Jekhmane, S., Maity, S. et al. Molecular action of NZ2114, a superior plectasin derivative. npj Antimicrob Resist 4, 34 (2026). https://doi.org/10.1038/s44259-026-00196-6
Keywords: antimicrobial peptides, Lipid II, MRSA, calcium-dependent antibiotics, supramolecular assemblies