Clear Sky Science · en
Selection of porin and LPS mutants during infection by phage T4 facilitates the emergence of β-lactam resistance in Escherichia coli
Why this matters for everyday medicine
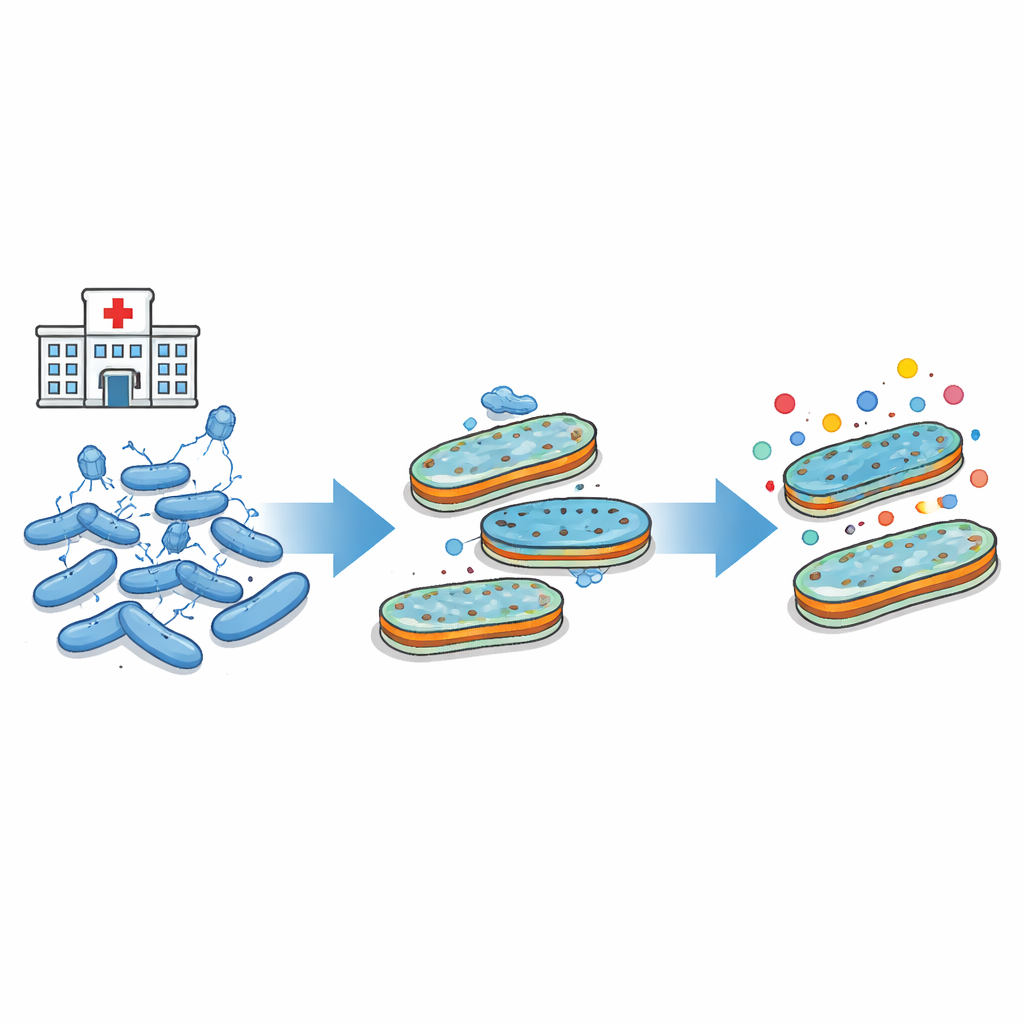
Doctors are running out of options to treat infections caused by hard-to-kill bacteria, and new ideas like using viruses that attack bacteria, called phages, are gaining attention. This study asks a simple but crucial question: when we use such viruses to fight bacteria, could we accidentally make it easier for those bacteria to become resistant to antibiotics, especially powerful drugs like carbapenems that are often used as a last resort?
The outer armor of bacteria
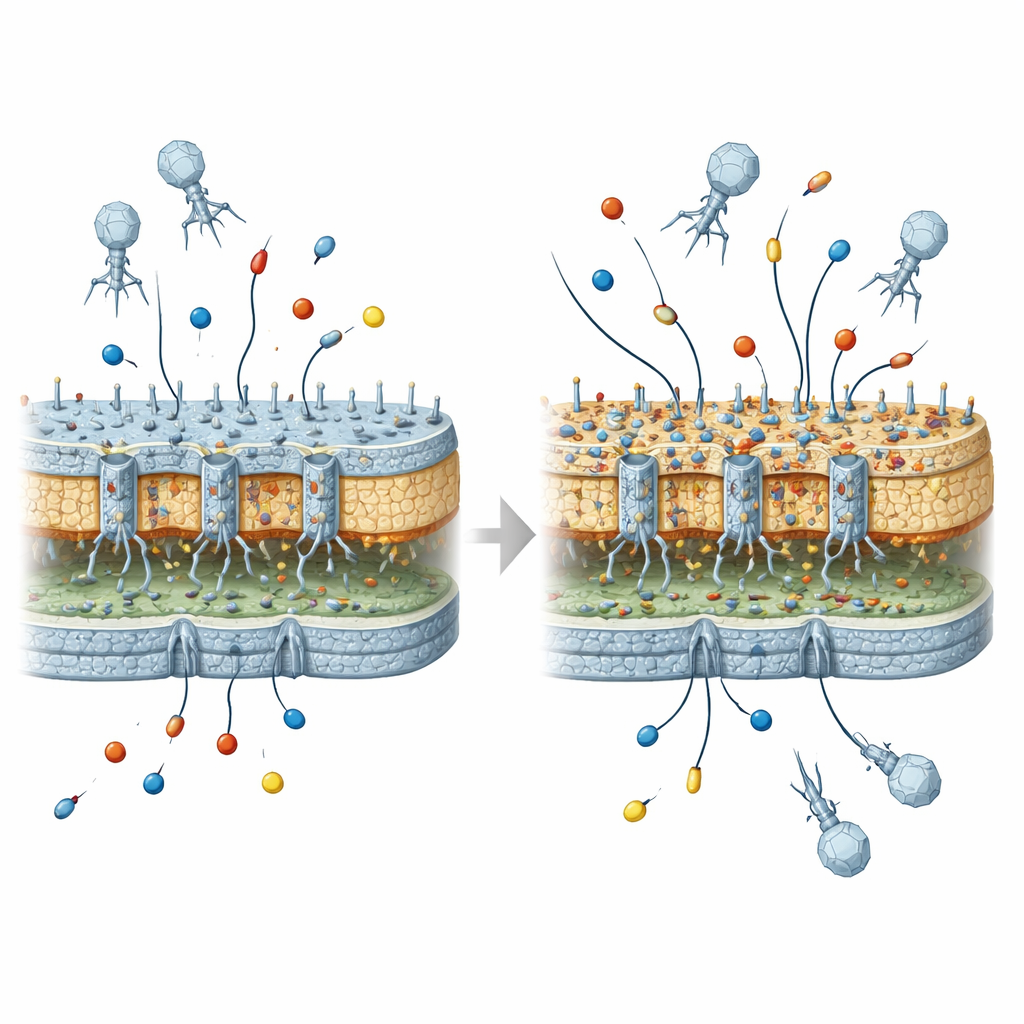
Many problematic bacteria, including Escherichia coli, are wrapped in a tough outer shell. This shell contains tiny channels, or pores, that let in nutrients and also allow many antibiotics to slip inside. It also carries sugary-fat molecules known as LPS that help form a dense barrier. Both the pores and the LPS layer are essential for the bacteria’s survival—but they also serve as landing pads for attacking phages. Because the same outer structures are used both to admit antibiotics and to admit phages, any changes that protect bacteria from one threat can affect how they respond to the other.
Viruses that shape bacterial defenses
The researchers focused on a well-known lab strain of E. coli and a classic virus that infects it, called T4. This virus needs a specific pore (called OmpC) and part of the inner LPS core to latch onto and start an infection. By exposing large numbers of bacteria to this virus under different conditions, and then sequencing the DNA of survivors, the team found that resistance to the virus almost always involved changes in the pore, the inner LPS core, or both. Sometimes regulatory systems that control how pores are produced were also altered. In all cases, the end result was a remodeled outer shell that no longer allowed the virus to attach easily.
When virus resistance sets the stage for drug resistance
Next, the team asked how these virus-resistant bacteria behaved when exposed to antibiotics. On their own, most of the mutations that blocked the phage did not make the bacteria strongly resistant to important β-lactam antibiotics such as carbapenems or advanced cephalosporins. But when the bacteria were given genes for common β-lactamase enzymes—molecules that break down β-lactam antibiotics—a different picture emerged. A small but important subset of virus-selected mutants, those with both a damaged OmpC pore and a deeply altered inner LPS core, showed noticeably higher survival against drugs like meropenem, cefepime, and ertapenem. These levels were still below full clinical resistance, but were clearly higher than in the original strain carrying the same enzymes.
Rebuilding the mechanism from the ground up
To be sure this was not a coincidence, the scientists engineered precise bacterial mutants with defined deletions in the pore gene and in selected LPS core genes, then measured how well carbapenems worked in these backgrounds with and without β-lactamases. They confirmed that losing the OmpC pore while also disrupting key inner LPS genes was sufficient to mimic the resistance pattern seen after virus exposure. Inner-core changes, not outer decorations, were critical. The authors suggest that these inner-core defects likely disturb the assembly or function of the remaining pores and trigger stress responses in the cell envelope, together lowering the overall permeability of the outer shell to antibiotics.
What this means for future phage treatments
To a lay reader, the central message is that viruses used to kill bacteria can unintentionally remodel the bacteria’s outer armor in ways that make them better prepared to withstand certain antibiotics—provided they later pick up the right resistance enzymes. On their own, the virus-selected changes do not create highly drug-resistant strains, but they create a “permissive” background that can turn modest resistance genes into more serious clinical problems. As phage therapy moves closer to routine medical use, this work argues that we must carefully consider these evolutionary side effects, designing virus treatments that cure infections while minimizing the risk of helping bacteria take the next step toward antibiotic resistance.
Citation: Le-Boulch, J., Charneau, E., Chevallereau, A. et al. Selection of porin and LPS mutants during infection by phage T4 facilitates the emergence of β-lactam resistance in Escherichia coli. npj Antimicrob Resist 4, 21 (2026). https://doi.org/10.1038/s44259-026-00193-9
Keywords: phage therapy, antibiotic resistance, Escherichia coli, beta-lactamases, bacterial outer membrane