Clear Sky Science · en
A selective and augmentable butyrate-FFAR2 signal circuitry programs the cellular identity of enteroendocrine L-cells
How Gut Fiber Talks to Hunger Hormones
Why does a high‑fiber diet help people feel full and avoid weight gain? This study digs into that question by zooming in on a special group of gut cells that release appetite‑controlling hormones. The researchers show how a molecule made when our gut microbes digest fiber can reprogram these cells to favor peptide YY (PYY), a hormone that curbs hunger, and suggest ways this natural pathway could be amplified for future obesity treatments.
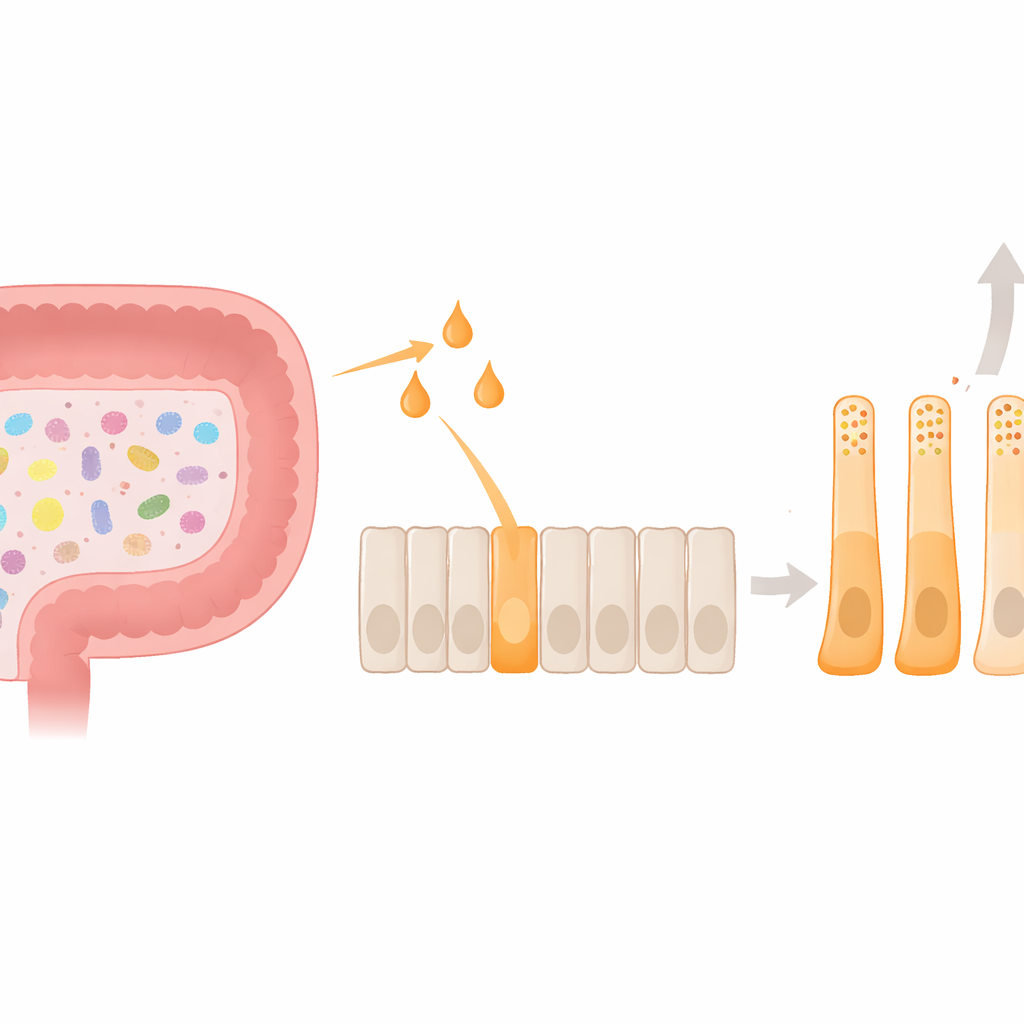
A Key Player in the Gut’s Hunger Circuit
Deep in the lining of the large intestine live enteroendocrine L‑cells, tiny hormone factories that release glucagon‑like peptide‑1 (GLP‑1) and PYY after we eat. These hormones signal the brain to reduce appetite and help balance blood sugar. The gut microbiome adds an extra layer to this system: when bacteria ferment dietary fiber, they produce short‑chain fatty acids such as acetate, propionate, and butyrate. These small molecules, in turn, activate a surface receptor on L‑cells called FFAR2. Until now, most work had focused on how this signaling triggers short bursts of hormone release; much less was known about how it might permanently shape the identity and behavior of the L‑cells themselves.
Butyrate Tips Cells Toward a PYY‑Rich Identity
Using a human L‑cell model, the team compared how acetate, propionate, and butyrate affect hormone production and cell shape. All three molecules raised PYY gene activity, but butyrate had by far the strongest effect, and uniquely boosted actual PYY secretion. At the same time, butyrate reduced expression of the GLP‑1 precursor gene. Under the microscope, butyrate‑treated cells stretched into elongated forms with long processes, closely resembling PYY‑rich cells seen in human colon tissue. Inside, their hormone storage granules shifted: PYY granules became more numerous and made up a larger share of the total hormone pool. Together, these changes show that butyrate does more than trigger a quick hormone burst—it steers cells toward a stable, PYY‑biased state in both function and form.
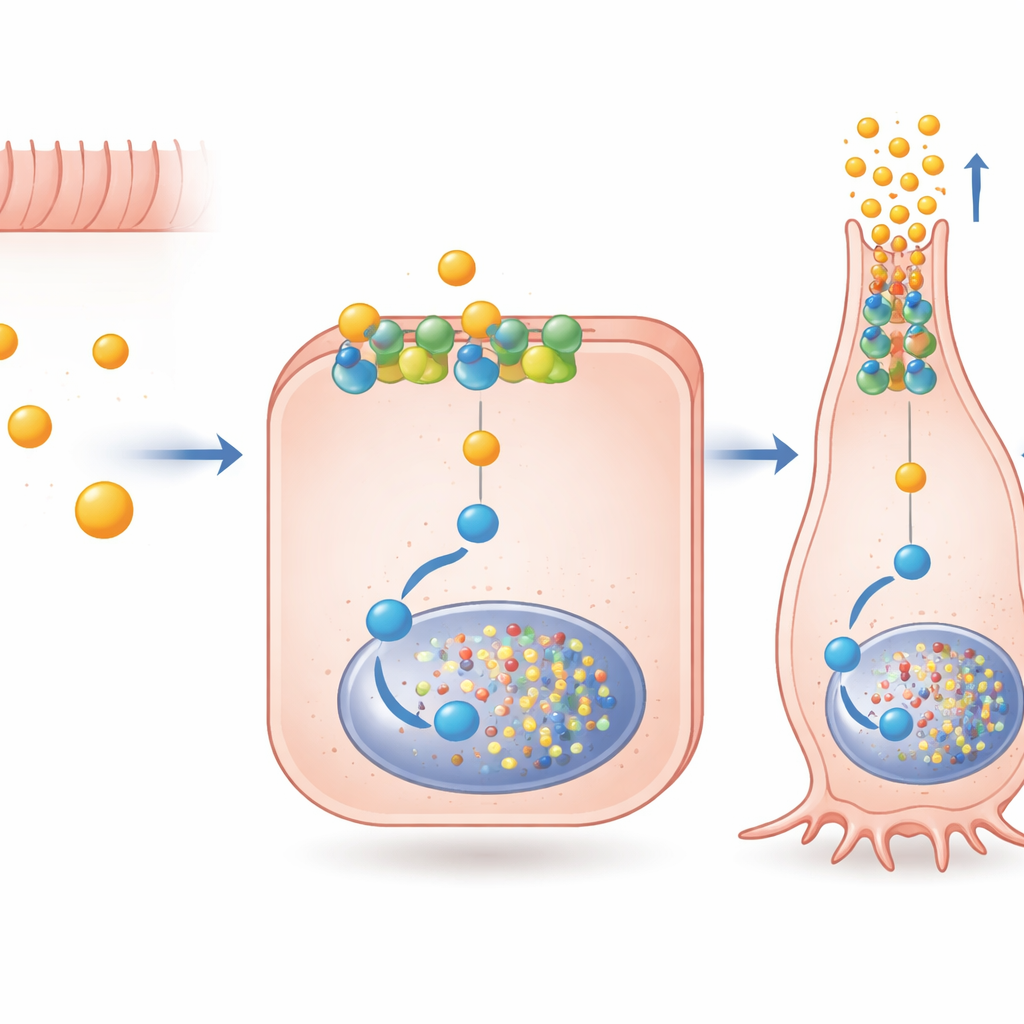
A Specialized Signal Pathway Inside the Cell
The authors then asked how butyrate sends such a specific message through FFAR2, a receptor that can connect to more than one internal signaling route. They found that acetate and propionate strongly activate both major FFAR2 pathways, while butyrate prefers one linked to a protein called Gαi. By blocking this pathway with targeted drugs, they showed that Gαi signaling through FFAR2 is required for much of butyrate’s boost in PYY gene activity and for switching on Pax6, a transcription factor important for late stages of hormone‑cell maturation. Importantly, this signaling did not depend on the receptor being dragged inside the cell; it could work directly from the outer membrane. An experimental molecule, AZ‑1729, which selectively enhances FFAR2‑Gαi signaling, further amplified butyrate’s effects on PYY and Pax6, hinting that pharmacological “boosters” could fine‑tune this natural circuit.
Guiding Stem Cells Toward Secretory Gut Cells
To see whether butyrate also influences how new hormone‑secreting cells are made, the researchers turned to mouse colonic organoids—mini‑guts grown from stem cells. In this system, cell fate is heavily controlled by the Notch pathway, which tends to keep cells in a proliferating or absorption‑focused state and blocks them from becoming secretory cells like L‑cells. Using a fluorescent Notch reporter, the team showed that butyrate dampened Notch activity across different organoid types and time points, and gene‑expression profiling confirmed that Notch‑related programs were suppressed. They also saw an increase in rare PYY‑positive cells within these organoids. In the human L‑cell line, butyrate selectively increased markers of late endocrine maturation, especially NeuroD1 and Pax6, underscoring its role in pushing cells toward a fully developed, PYY‑producing identity.
From Microbial Metabolite to Future Therapies
This work paints a coherent picture: butyrate, a common product of fiber fermentation in the colon, can both rewire mature L‑cells and influence their developmental pathway to favor PYY‑rich, elongated cells that are poised to communicate with the nervous system and curb appetite. The signaling relies heavily on a butyrate‑activated FFAR2–Gαi circuit at the cell surface and can be further strengthened by a designer molecule that biases this pathway. For lay readers, the key message is that what our gut microbes do with fiber does not just trigger short‑lived hormone bursts—it can reshape the very make‑up of the gut’s appetite‑sensing cells, suggesting new strategies to harness diet and targeted drugs to support satiety and metabolic health.
Citation: Hirdaramani, A., Cheng, CW., Hanyaloglu, A.C. et al. A selective and augmentable butyrate-FFAR2 signal circuitry programs the cellular identity of enteroendocrine L-cells. Commun Biol 9, 606 (2026). https://doi.org/10.1038/s42003-026-09830-5
Keywords: butyrate, enteroendocrine L-cells, peptide YY, gut microbiome, FFAR2 signaling