Clear Sky Science · en
ATPase N-ethylmaleimide-sensitive factor mediated calcium dependent fusion pore closure and endocytosis
How Cells Reuse Their Secret Packages
Every second, your brain cells, hormone-secreting cells, and immune cells are releasing tiny packets of chemical messengers and then quickly taking the emptied packets back in for reuse. This rapid ship-and-recycle system keeps thinking, blood sugar control, and immunity running smoothly. The study behind this article uncovers a missing piece of that recycling puzzle: how the small opening that lets cargo out – the “fusion pore” – knows when and how to close so the empty packet can be pulled back into the cell.
The Life Cycle of a Secret Packet
Many cells communicate by loading signals such as neurotransmitters, hormones, or inflammatory molecules into bubble-like packages called vesicles. These vesicles move to the cell surface and briefly join with the outer membrane, forming a tiny water-filled tunnel – the fusion pore – through which their contents escape. Often, instead of fully merging, the vesicle performs “kiss-and-run”: it touches the surface, opens a pore just long enough to release its cargo, then the pore closes and the vesicle is retrieved to be used again. This fast, economical cycle depends critically on timing: open the fusion pore too little and not enough signal gets out; fail to close it and the recycling system stalls. 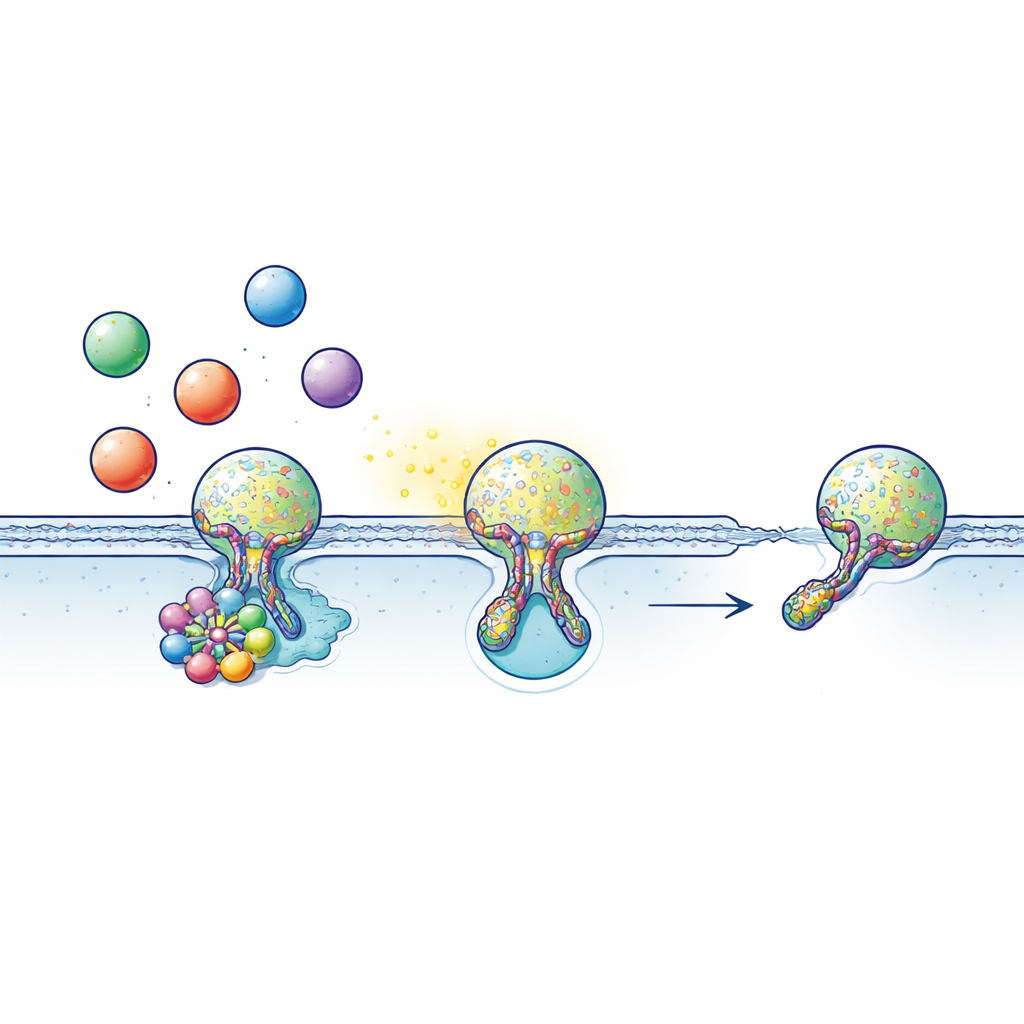
A Molecular Motor at the Pore
Vesicle fusion and retrieval are driven by proteins that grip the vesicle and the cell membrane and pull them together like a drawstring. These “SNARE” proteins form tight bundles that bring the two membranes into contact and open the fusion pore. The new study focuses on another protein, NSF, an enzyme that uses energy from ATP – the cell’s fuel – to take SNARE bundles apart after they have done their job. Using electrical recordings sensitive to changes in cell surface area together with live confocal imaging, the authors watched single vesicles fuse and be retrieved in rat adrenal hormone-secreting cells, sensory neurons, and insulin-producing cells. By chemically blocking NSF or removing it genetically, they could test whether its activity is needed not just before fusion, but also for closing the pore afterward.
When NSF Stalls, Pores Refuse to Close
Under normal conditions, many vesicles in these cells undergo kiss-and-run: the fusion pore opens, stays open for a short, well-defined time, then closes so the vesicle can be pulled back inside. When the researchers inhibited NSF’s ATP-driven activity with drugs or knocked it out, they saw a striking shift in behavior. Vesicles still reached the membrane and pores could still open, but a much smaller fraction of events completed pore closure. Instead, vesicles tended to remain attached to the surface with pores that stayed open for far longer than usual. This change went hand in hand with a strong slowdown or block of several flavors of endocytosis, including very fast, slower, and “overshoot” retrieval in which extra membrane is taken back. The pool of vesicles ready for rapid release was also refilled more slowly, showing that pore closure and vesicle recycling are tightly linked steps.
Calcium-Guided Recycling Choices
Cells can recycle vesicles in both calcium-dependent and calcium-independent ways. In sensory neurons of the dorsal root ganglion, the team compared these two routes. With calcium present, blocking NSF again slowed retrieval after vesicle fusion, indicating that NSF is essential for calcium-triggered recycling. But when the scientists used a stimulation pattern that provokes a calcium-free, voltage-driven recycling mode, NSF blockers had little effect. This contrast suggests that cells reserve NSF as a specialized tool for handling bursts of calcium-driven activity, such as those occurring during intense signaling, while leaving a separate, calcium-independent pathway to support quieter, background recycling.
How NSF Pulls the Strings
To get a closer look at what NSF is doing at the fusion pore, the researchers built a fluorescent sensor that glows when SNARE proteins form a bundle and dims when that bundle is pulled apart. During stimulation, they saw these signals appear where vesicles fused and then fade within a few seconds as the pore closed and the vesicle was retrieved. When NSF was blocked, the glowing signal lingered much longer, indicating that the SNARE bundle remained assembled instead of being dismantled. Together with measurements of ATPase activity – NSF’s fuel-burning action – these results support a picture in which NSF actively disassembles SNARE bundles at a late stage of fusion. That disassembly appears to help tighten and finally pinch off the pore, finishing the kiss-and-run cycle. 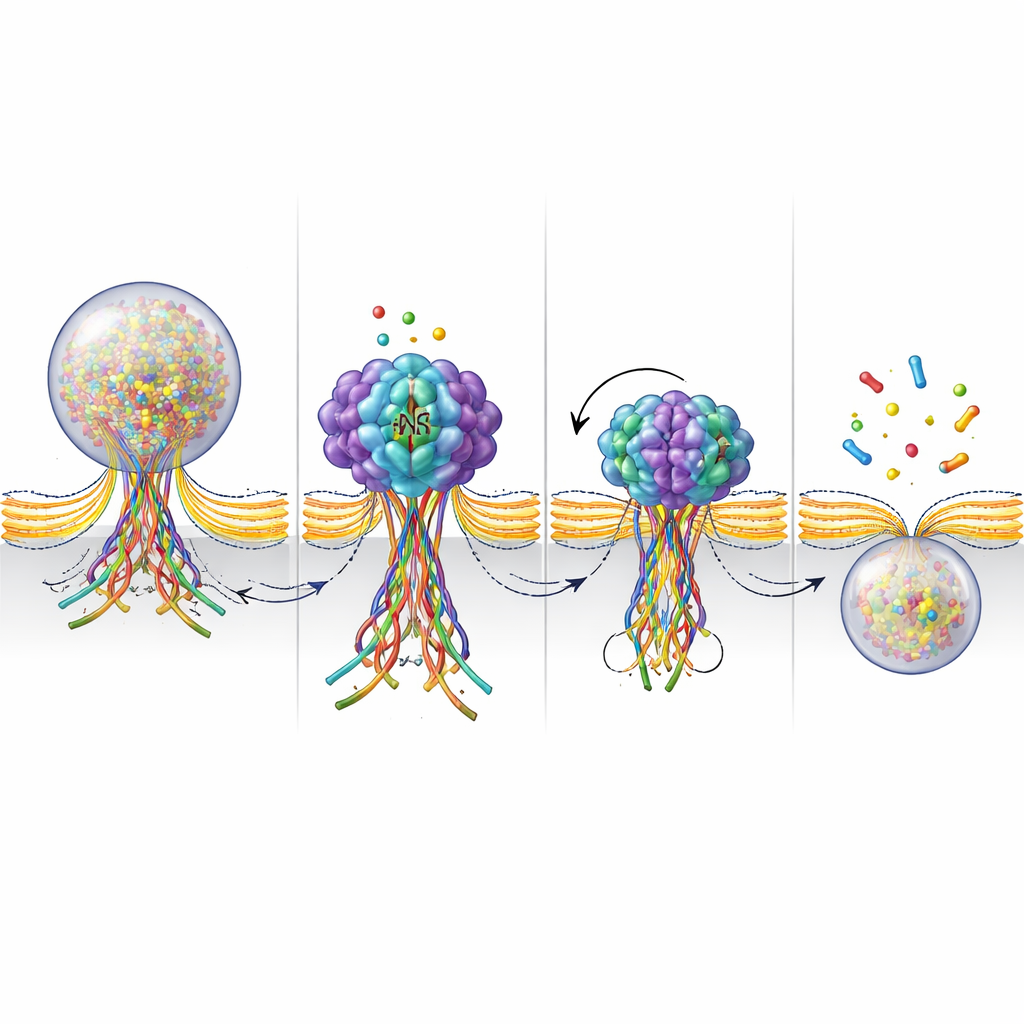
Why This Matters for Health and Disease
By showing that NSF’s energy-driven action is a central switch for closing the fusion pore and triggering vesicle recycling, this work ties a well-known molecular motor to the fine control of cell-to-cell communication. In hormone-secreting cells, adrenal chromaffin cells, and certain neurons, this mechanism ensures that vesicles can be reused rapidly and reliably whenever calcium surges. If NSF function is disrupted, pores may fail to close properly, recycling slows, and cells may struggle to keep up with demand, potentially contributing to problems in stress responses, pain signaling, or blood sugar control. Understanding this closing step opens new avenues for exploring how subtle defects in vesicle recycling might underlie neurological and endocrine disorders, and highlights NSF as a potential target for future therapeutic strategies.
Citation: Wang, Y., Ma, S., Song, S. et al. ATPase N-ethylmaleimide-sensitive factor mediated calcium dependent fusion pore closure and endocytosis. Commun Biol 9, 495 (2026). https://doi.org/10.1038/s42003-026-09743-3
Keywords: vesicle recycling, fusion pore, endocytosis, calcium signaling, NSF ATPase