Clear Sky Science · en
GPR124 regulates hyaloid blood vessel regression and is associated with endothelial-mesenchymal transition
Why tiny eye vessels must disappear
Before birth, the inside of the eye is laced with a temporary web of blood vessels that feed the growing lens and surrounding tissue. For sharp vision later in life, this scaffold must vanish on schedule and be replaced by the permanent retinal circulation. When this cleanup fails, children can develop serious eye problems, including retinal detachment and blindness. This study asks a basic but crucial question: what molecular signals tell these temporary vessels when and how to disappear?
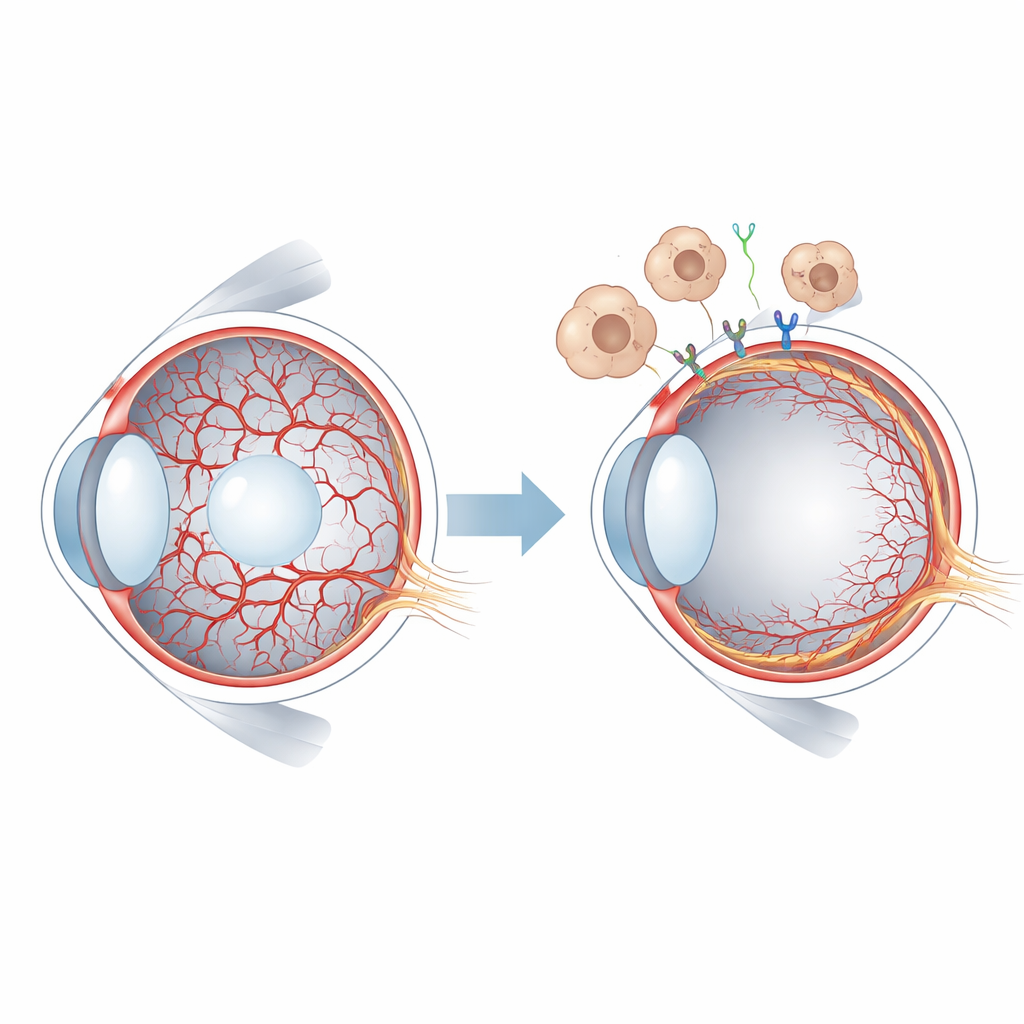
A short-lived blood supply in the young eye
In mammals, early eye tissue is nourished by the hyaloid vessels, a delicate network that spans the gel-like center of the eye and wraps around the lens. As the retina starts to build its own blood supply, the hyaloid system must regress and clear out. In humans this happens before birth, while in mice it unfolds after birth, offering a convenient window for experimental study. When hyaloid vessels persist, as in a condition called persistent hyperplastic primary vitreous, vision can be severely compromised. Researchers already knew that a family of signals called WNT, and in particular the protein WNT7B released by immune cells called macrophages, helps trigger hyaloid vessel regression. But the specific receptors on blood-vessel cells and the downstream events were not well understood.
Pinpointing a key molecular gatekeeper
The team focused on GPR124, a receptor-like protein known to act as a co-receptor for WNT7A and WNT7B in the developing brain, where it promotes blood-vessel growth and blood–brain barrier formation. They engineered mice in which GPR124 could be deleted specifically in endothelial cells—the cells that line blood vessels—shortly after birth. When GPR124 was removed from hyaloid endothelial cells, the central eye vessels regressed much more slowly than in normal littermates. The hyaloid network remained denser for longer, even though the same signal appeared unnecessary for building the retinal vessels along the inner surface of the eye. This compartment-specific effect shows that GPR124 is critical for dismantling the temporary hyaloid circulation but largely dispensable for constructing the permanent retinal network.
How the signal reshapes and removes vessels
To understand how GPR124 influences cell behavior, the researchers measured activity of WNT target genes in hyaloid vessels. They found that loss of GPR124 markedly reduced the nuclear levels of LEF1 and other classic WNT-responsive proteins, indicating a dampened WNT/β-catenin pathway. Yet GPR124 did not control all forms of cell death. The number of isolated dying endothelial cells along otherwise intact vessels was similar with or without GPR124. Instead, GPR124 was required for coordinated “segmental” regression, where strings of neighboring endothelial cells along a vessel segment died together, causing the tube to collapse. This pattern suggests that GPR124 helps convert scattered signals into a concerted remodeling event that efficiently prunes unwanted vessel sections.
Cells in transition between two identities
Diving deeper, the team used single-cell RNA sequencing to profile individual cells from the hyaloid network. They discovered that endothelial cells with intact GPR124 showed a gene-expression pattern consistent with a partial endothelial-to-mesenchymal transition, a state in which they begin to lose some hallmark endothelial features while gaining traits associated with more mobile, matrix-producing cells. Classic endothelial markers such as VE-cadherin and von Willebrand factor were reduced, while mesenchymal markers like ZEB1 increased. Microscopy confirmed these changes at the protein level. At the same time, several genes promoting cell death were more active in GPR124-positive cells, and genes linked to transforming growth factor beta, another major developmental pathway, were also altered. Neighboring support cells called pericytes showed secondary stress responses, but careful imaging indicated that endothelial cells died first, followed later by pericyte loss and, ultimately, empty “ghost” strands of basement membrane.
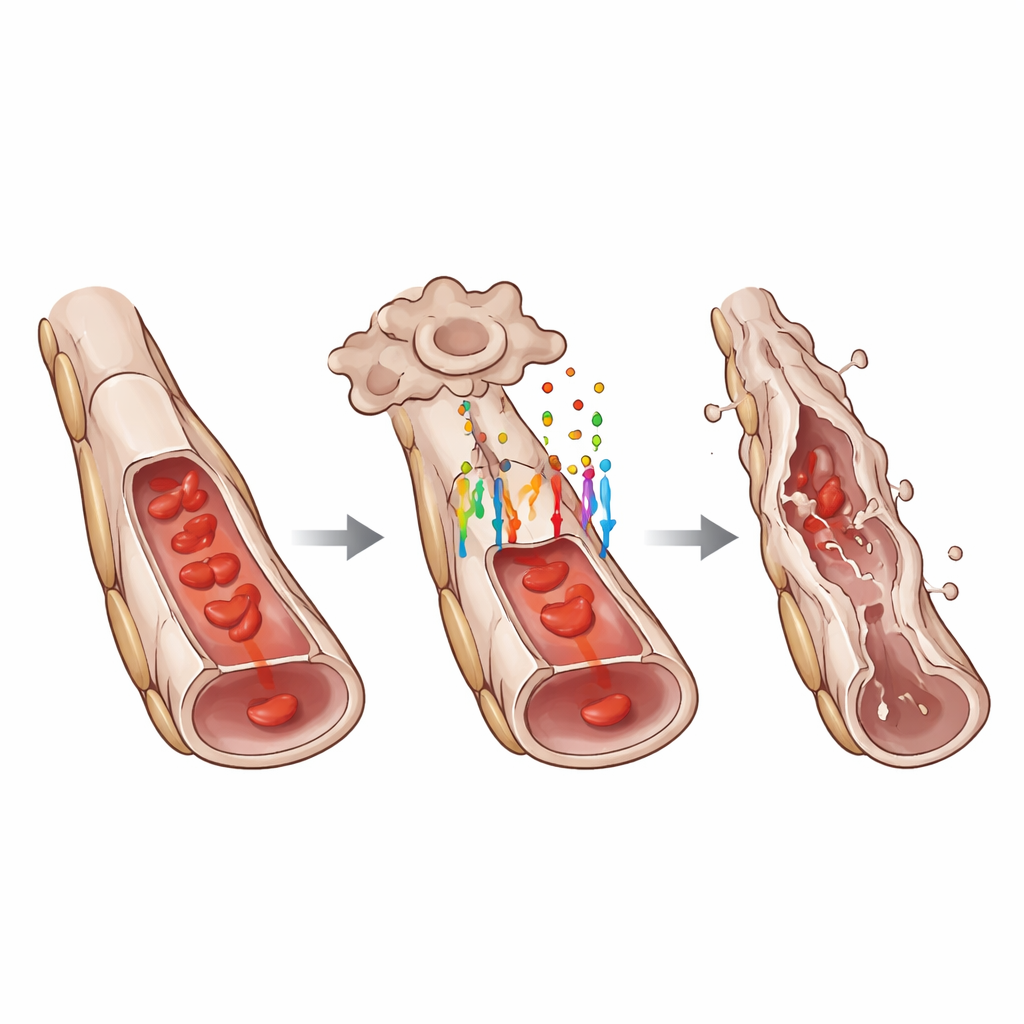
What this means for eye health and beyond
Taken together, the findings position GPR124 as a central coordinator of hyaloid vessel regression. By enabling WNT7-driven signaling, GPR124 pushes endothelial cells into a transitional state, loosens their junctions, primes them for apoptosis, and helps orchestrate the stepwise collapse and cleanup of obsolete vessels, all while leaving retinal vessel growth largely intact. Understanding this finely tuned program offers insight into how the developing eye remodels itself and highlights potential molecular targets for treating disorders where temporary vessels refuse to go away. Similar GPR124-dependent transitions may shape blood-vessel pruning in other organs, suggesting broader relevance for normal development and for diseases marked by abnormal vessel persistence or scarring.
Citation: Hannig, L., Heiden, R., Ergün, S. et al. GPR124 regulates hyaloid blood vessel regression and is associated with endothelial-mesenchymal transition. Sci Rep 16, 13765 (2026). https://doi.org/10.1038/s41598-026-50835-1
Keywords: eye development, blood vessel regression, Wnt signaling, GPR124, endothelial transition