Clear Sky Science · en
Modulation of oxidative stress and P53/PCNA signaling by glucosodiene-loaded nanoferrites (GLONF) in ehrlich solid tumor–induced hepatotoxicity
Why this research matters
Cancer treatments often damage healthy organs, especially the liver, which must break down many drugs and waste products from tumors. This study explores a new, tiny, magnetically guided carrier that delivers a sugar-based anticancer compound directly toward tumors, while also shielding the liver from harm. For readers, it offers a glimpse of how nanotechnology and smart chemistry might one day make cancer treatment more effective and less toxic.
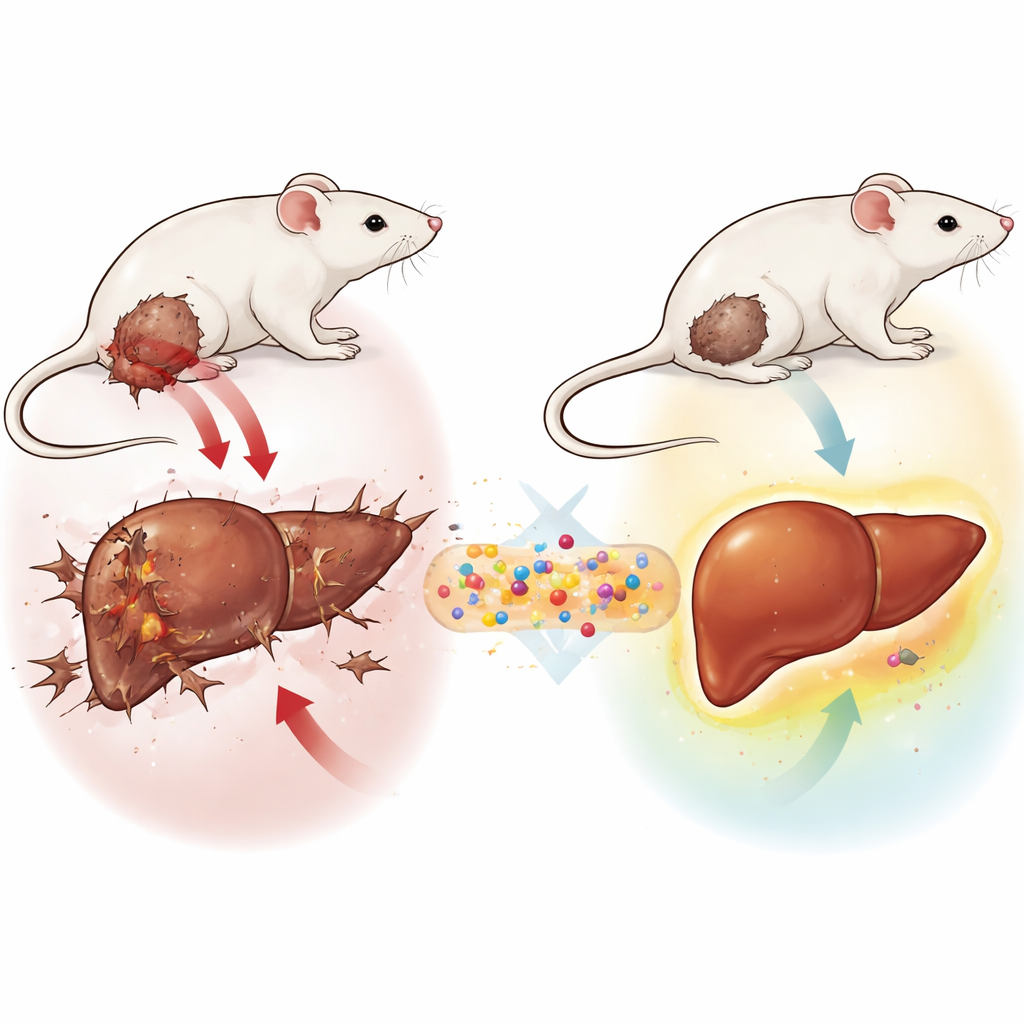
A tiny delivery system with a big job
The researchers built a three-part nano-sized system called GLONF. At its center is a magnetic mineral core made of a special iron-based material. Around that core they added a natural sugar-based shell called chitosan, often used in medical materials for its safety and stability. Finally, they attached a new sugar-derived molecule called glucosodiene, designed to interfere with cancer cells’ hunger for glucose and to soften the harsh, acidic environment around tumors. Together, these components create a miniature vehicle that can, in principle, be guided by magnets, travel safely in the body, and release an anticancer agent where it is needed most.
Testing in a mouse model of breast-like cancer
To see whether GLONF could protect the liver during cancer, the team used a well-established mouse model called Ehrlich solid tumor. This tumor, originally derived from breast tissue, grows quickly under the skin and mimics many features of aggressive human cancers. Female mice were divided into groups: some remained healthy, some received only the nano-formulation, some developed tumors without treatment, and others received GLONF either at the same time as tumor implantation or after the tumor had formed. The scientists then measured tumor size, standard blood markers of liver health, and a battery of chemical and microscopic signs of damage and repair inside the liver.
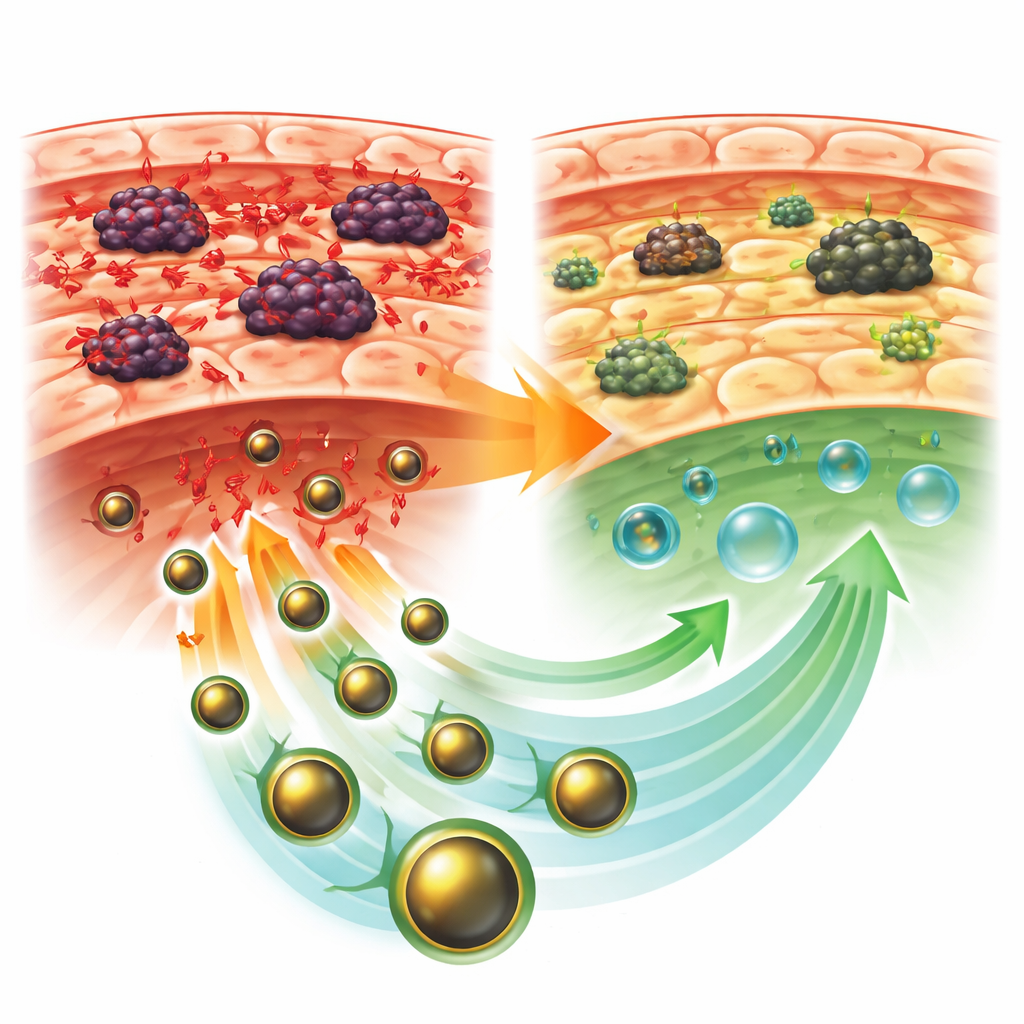
Protecting the liver from stress and damage
Mice bearing tumors without treatment showed a classic picture of liver injury. Enzymes that leak into the blood when liver cells are damaged rose sharply, while beneficial blood proteins dropped. Inside the liver, levels of damaging by-products of oxidation increased, and natural defenses such as protective enzymes and glutathione fell. Tissue sections under the microscope revealed distorted liver architecture, with swollen and dying cells, scar-like changes, and heavy inflammatory cell invasion. In contrast, mice treated with GLONF had smaller tumors and substantially healthier livers. Their blood tests moved back toward normal, oxidative damage was reduced, and antioxidant defenses rebounded, especially when GLONF was given after the tumor was allowed to form.
Calming runaway cell signals
The team also examined two key proteins inside liver cells. One, PCNA, is associated with cell proliferation, and the other, P53, responds to DNA damage and can trigger cell death. In tumor-bearing mice without treatment, both markers were strongly elevated, signalling intense stress and uncontrolled attempts at repair. GLONF treatment noticeably lowered the abundance of these markers in liver tissue, suggesting that the nano-system reduced the need for emergency repair and helped stabilize the cellular environment. This aligns with the biochemical and structural findings, pointing to a coordinated easing of oxidative stress and injury.
What this could mean for future cancer care
In summary, this work shows that GLONF, a magnetically responsive, sugar-based nano-carrier, can shrink tumors in mice while protecting the liver from serious harm. By combining targeted delivery with antioxidant and metabolic effects, the system offers a potential way to make cancer treatment gentler on vital organs. Although these results are still in animals and far from routine clinical use, they demonstrate how carefully engineered nano-structures might help future therapies attack tumors while sparing the body’s natural filters.
Citation: Tousson, E., Atrash, A.E., Abdelrasol, M.Y. et al. Modulation of oxidative stress and P53/PCNA signaling by glucosodiene-loaded nanoferrites (GLONF) in ehrlich solid tumor–induced hepatotoxicity. Sci Rep 16, 12351 (2026). https://doi.org/10.1038/s41598-026-45548-4
Keywords: nanomedicine, liver protection, breast cancer model, oxidative stress, drug delivery