Clear Sky Science · en
Usage of the adaptive quasi-linear viscoelastic model to predict load-unload, stress-relaxation, and sine load of porcine liver
Why the squishiness of liver matters
When you slam on the brakes in a car crash, or when a surgeon pulls on an organ during an operation, your liver does not behave like a simple rubber band. It slowly stretches, relaxes, and dissipates energy in ways that are tricky to predict. This study explores how well a popular mathematical model can capture that complex behavior in pig liver and asks a deceptively simple question: are the model’s parameters true material properties of liver, or do they change with how quickly and in what way the tissue is loaded?
How scientists currently model soft organs
Soft organs such as liver are viscoelastic: they resist being deformed like an elastic solid, but they also flow and relax like a thick fluid. For decades, researchers have used families of models called quasi-linear viscoelastic (QLV) models to describe this behavior. An improved version, the adaptive quasi-linear viscoelastic (AQLV) model, represents tissue as combinations of springs and dashpots whose response can change with strain. It is attractive because it has analytic formulas for common types of loading and can be calibrated from relatively simple tests. However, the standard way to calibrate the AQLV model uses slow stretching and holding of the tissue, leaving open whether the same parameters can be trusted when the liver is loaded much faster, as happens in impacts or rapid surgical maneuvers.
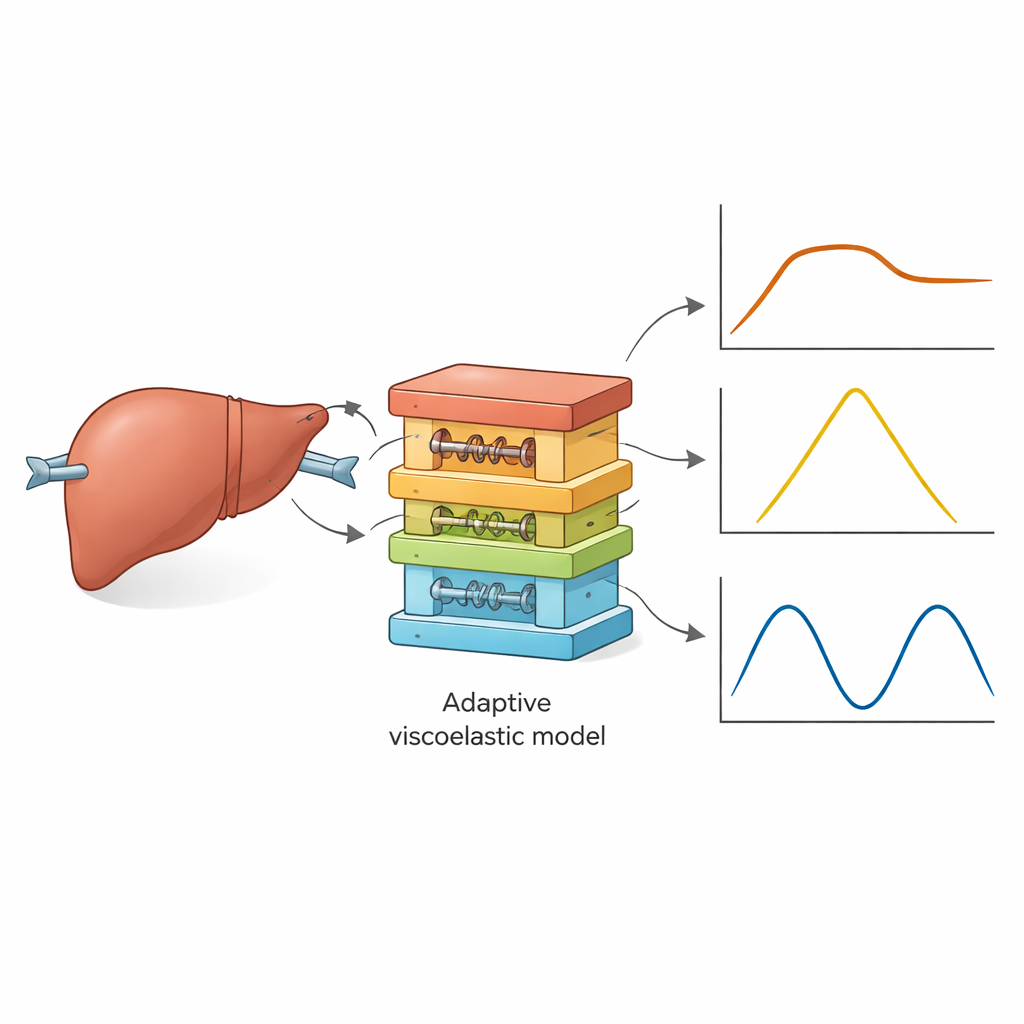
Putting the liver model to the test
The authors used AQLV parameters from earlier slow tests on porcine liver and asked the model to predict three very different experiments performed in a separate study: a quick stretch followed by a hold (stress-relaxation), a triangular load-unload cycle, and a back-and-forth sine-wave loading at several frequencies. In each case, measured strain from the experiments was fed into the model to generate predicted stress, which was then compared with the actual recorded stresses. Initially, the model struggled badly: error levels were large, some predictions showed impossible negative tension during unloading, and key energy measures differed significantly from experiment. This meant that the original parameter set, obtained at one slow strain rate, could not simply be reused under different loading histories.
Re-tuning the model for each type of loading
To probe further, the researchers re-calibrated the AQLV parameters separately for each loading case, using least-squares optimization while keeping the model structure unchanged. Once retuned, the model reproduced fast stress-relaxation curves almost perfectly, with errors dropping by orders of magnitude; it also gave realistic predictions for the rapid ramp itself. For load-unload cycles, re-calibration removed the negative-stress artifact and brought loading and unloading energies close to the experimental values. Under sinusoidal loading, the adjusted parameters allowed the model to match how stiff the liver appears (storage modulus) and how much energy it dissipates (loss modulus and loss tangent) across frequencies, with only minor discrepancies at the highest frequency tested. Crucially, the patterns of how individual spring stiffnesses and relaxation times changed made clear that the internal parameters systematically shift with strain rate and frequency.
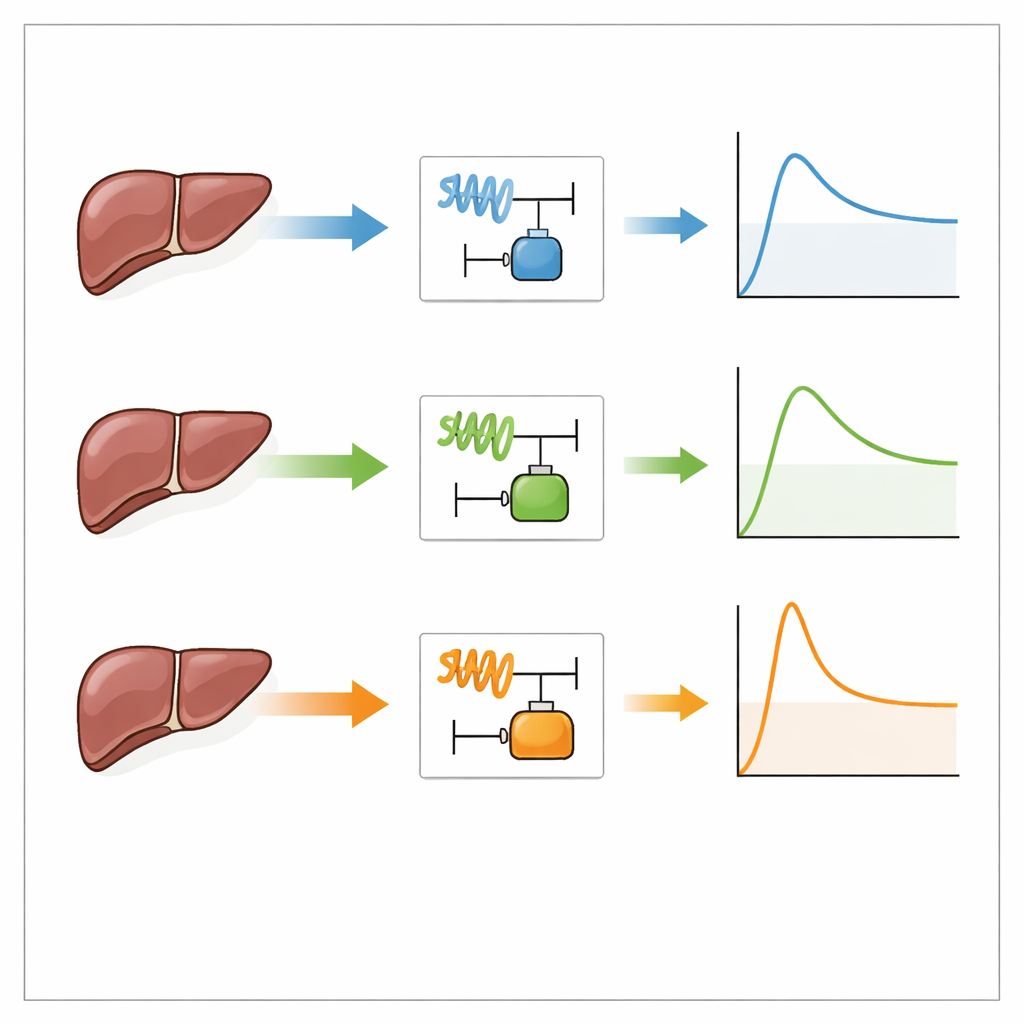
Can one calibration cover many situations?
The team then explored a practical shortcut: could they calibrate the model once in a fast ramp-hold test and reuse those parameters to predict other but related loadings? Using fast ramp-hold parameters to predict sine-wave tests at similar average strain rates worked reasonably well for the elastic part of the response: the storage modulus was close to experimental values across all frequencies. However, measures tied to energy loss, especially the loss tangent, still differed significantly. Applying the same parameter set to load-unload cycles produced larger stress errors and mismatched loading and unloading energies, even though the overall shape of the curves was captured. These results suggest that matching strain rate alone is not enough; the exact time pattern of loading also matters.
What this means for modeling real tissue
From a broader perspective, the study shows that parameters in the AQLV model are not fixed, universal fingerprints of liver tissue. Instead, they depend strongly on how the tissue is tested—how fast it is stretched, how long it is held, and whether the loading is a single pulse, a cycle, or continuous oscillation. The model can describe uniaxial liver behavior very well once tailored to a specific protocol, but it does not provide a single, all-purpose set of liver constants. For applications like car-crash simulations, surgical planning, or the design of realistic training phantoms, this means modelers must choose either to recalibrate for each loading scenario or to adopt more advanced fractional viscoelastic models that better cover a wide range of time scales with one parameter set. In everyday terms, liver does not have one “stiffness”; its apparent stiffness changes with how you poke, pull, or shake it, and our models must account for that.
Citation: Bittner-Frank, M., Aryeetey, O.J., Estermann, SJ. et al. Usage of the adaptive quasi-linear viscoelastic model to predict load-unload, stress-relaxation, and sine load of porcine liver. Sci Rep 16, 10675 (2026). https://doi.org/10.1038/s41598-026-45415-2
Keywords: viscoelastic liver, strain rate dependence, biomechanical modeling, soft tissue mechanics, adaptive quasi-linear viscoelasticity