Clear Sky Science · en
Multifaceted biological and computational assessment of aromatic and N-heteroaromatic non-substituted thiosemicarbazones
Why these small molecules matter
Cancer and parasitic infections remain two of the world’s most stubborn health problems, and many existing drugs either do not work well enough or cause strong side effects. This study explores a family of relatively simple, easily made molecules called thiosemicarbazones to see whether some of them can selectively harm cancer cells and disease‑causing parasites while sparing healthy human cells. It also examines how these molecules behave in the body and how they interact with blood proteins, using both laboratory tests and computer simulations.
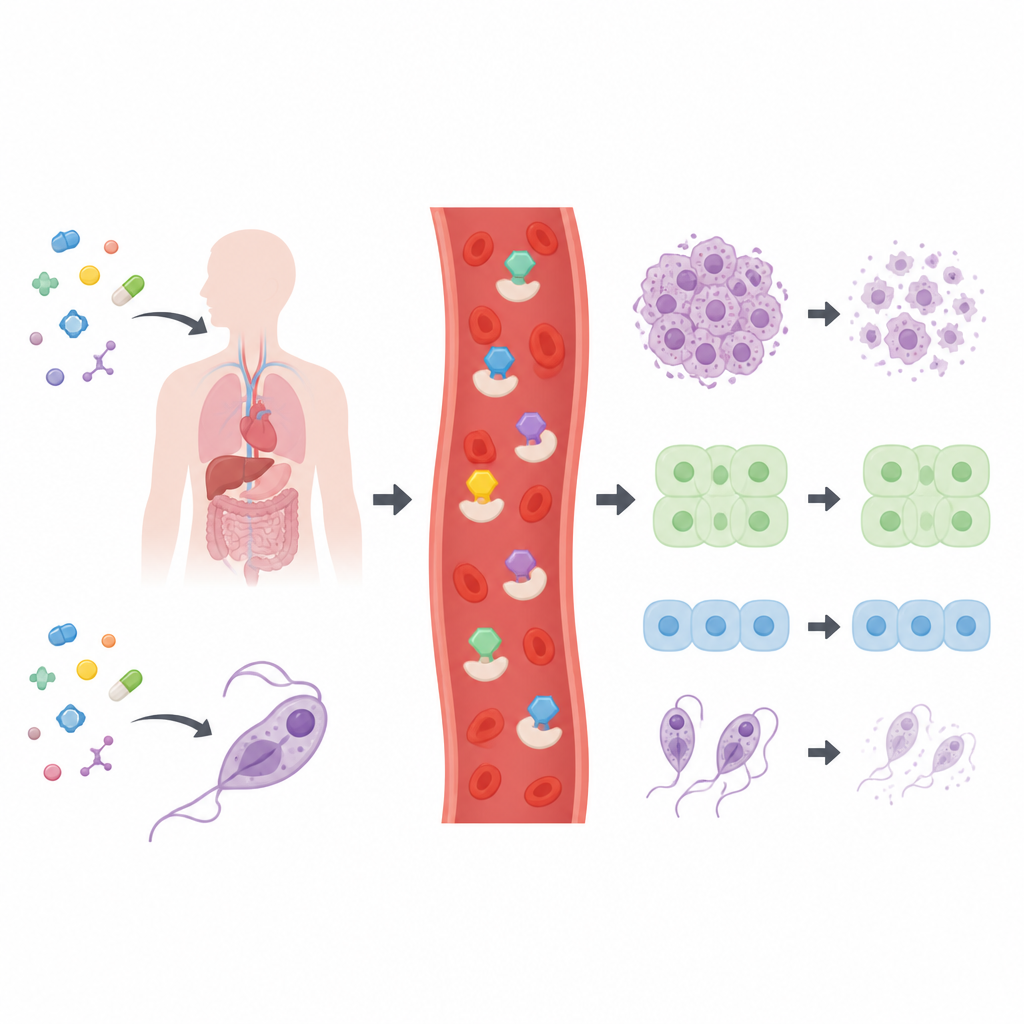
A library of candidates put to the test
The researchers created and characterized a set of 28 related thiosemicarbazone compounds, most built around ring‑shaped carbon frameworks known from many medicines. Before testing them on cells, they checked whether the molecules fit common “drug‑likeness” rules used in pharmaceutical research, which consider size, polarity, and flexibility. All of the compounds passed these basic filters, suggesting they are reasonable starting points for drug development. The team then exposed a wide panel of human cancer cell lines, two types of normal human cells, and four disease‑causing parasites to the compounds to map out which structures were most active.
Pinpointing the most promising molecules
Most of the library showed only modest effects, but six molecules stood out. Three were based on a pyridine ring, one on a quinoline ring, and one on an indole ring. These top candidates either slowed or killed cancer cells at low concentrations and, in several cases, also acted strongly against parasites that cause Chagas disease, African sleeping sickness, and leishmaniasis. The clear front‑runner, labeled 26, was especially effective against lung, liver, bone, and cervical cancer cell lines while showing essentially no toxicity toward normal lung and colon cells. That selectivity is crucial, because it hints at a window where tumor tissue can be attacked without comparable harm to healthy tissue.
How the lead compound attacks cancer cells
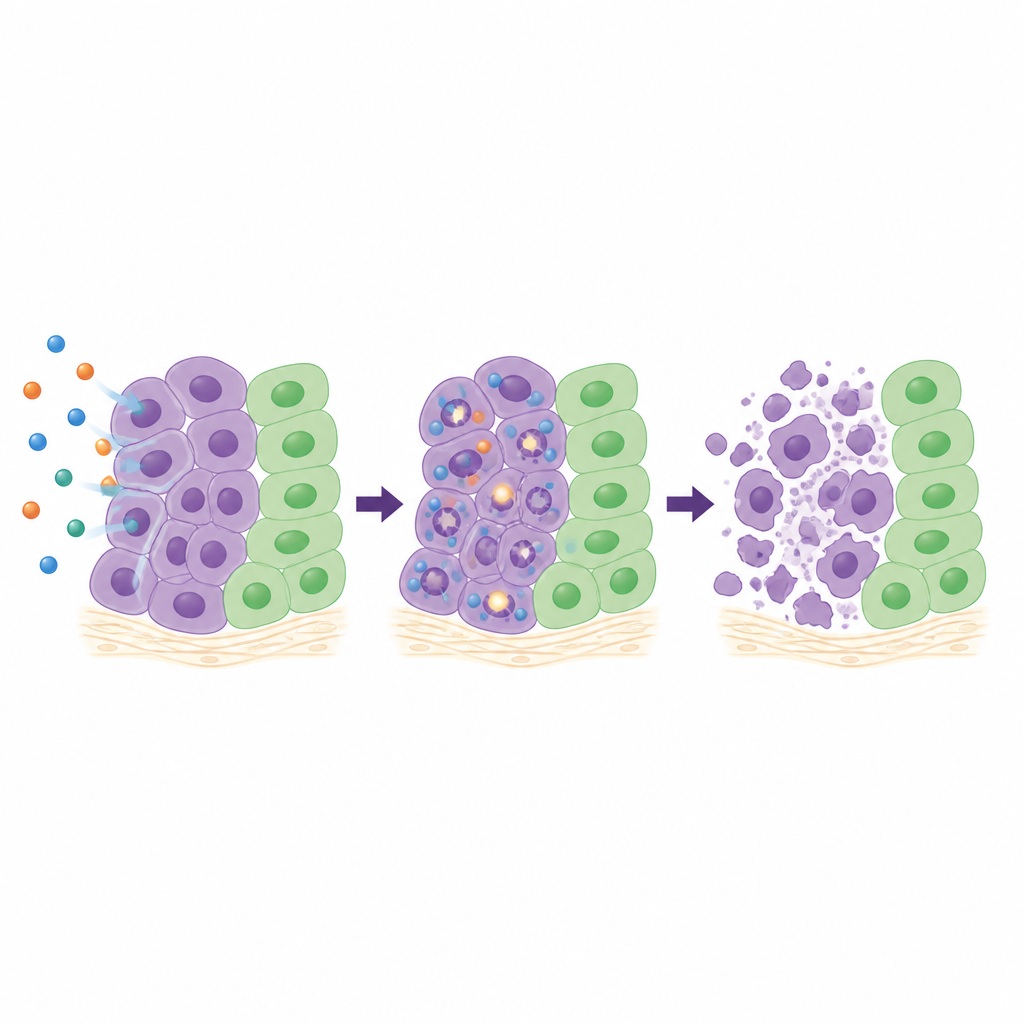
To understand what 26 does inside cells, the team looked in detail at four sensitive cancer lines. They followed how cells progress through the cell cycle, measured markers of DNA breakage, and tracked activation of internal “self‑destruct” enzymes. Depending on the cell type, 26 slowed cells at different stages of their growth cycle, but in all cases it caused increasing amounts of DNA damage over time and strongly turned on caspase‑3 and caspase‑7, key players in programmed cell death. In three‑dimensional clusters of lung cancer cells, which better mimic real tumors than flat cell layers, 26 sharply reduced spheroid growth and even shrank them at higher doses. In a chick egg membrane model where human tumor fragments grow with their own blood supply, treatment with 26 cut tumor area by roughly two‑thirds without visible damage to the surrounding tissue.
Clues from metal binding and blood transport
Why are some thiosemicarbazones active while others are not? Using quantum‑chemical calculations, the authors showed that the most effective compounds have electronic structures that favor grabbing hold of metal ions such as iron or copper at specific atoms. This ability to form tight “chelate” complexes is thought to underlie much of their biological activity. One standout, compound 27, behaves differently because its structure prevents this kind of metal binding, suggesting an alternative mode of action. The team also tested how readily the six key compounds might cross gut‑like membranes and how strongly they attach to human serum albumin, the main carrier protein in blood. All had rather poor passive gut permeability, hinting that formulation work will be needed for oral dosing. Compound 26, however, bound strongly to albumin, and computer simulations revealed a stable set of hydrogen bonds and hydrophobic contacts that help anchor it in a known drug‑binding pocket.
What this means for future medicines
Taken together, the experiments and simulations show that one member of this molecular family, compound 26, can selectively damage cancer cells by causing DNA breaks and triggering controlled cell death, while leaving tested normal cells largely unharmed. At the same time, related molecules show activity against several important parasites. Although their current form is not ideal for absorption through the gut, their simple structures, clear structure‑activity relationships, and defined interactions with blood proteins make them attractive building blocks for future drug design. With better delivery systems and animal testing, these thiosemicarbazones could contribute to new treatments for both cancer and parasitic diseases.
Citation: Macijewska, N., Ristić, P., Kallingal, A. et al. Multifaceted biological and computational assessment of aromatic and N-heteroaromatic non-substituted thiosemicarbazones. Sci Rep 16, 14911 (2026). https://doi.org/10.1038/s41598-026-44568-4
Keywords: thiosemicarbazones, anticancer agents, antiparasitic compounds, DNA damage, drug design