Clear Sky Science · en
Gut metabolite TMAO and its structural analogs bind to fibrinogen thereby enhancing clot formation: a rationale for atherosclerosis risk
From Gut Chemicals to Blood Clots
What happens in your gut does not stay in your gut. This study explores how a tiny molecule made by intestinal bacteria, called TMAO, can change the way blood clots form and break down. Because abnormal clots underlie heart attacks and strokes, understanding this hidden link between diet, gut microbes, common medicines, and clotting helps explain why some people face higher cardiovascular risk—and where new treatments might intervene.
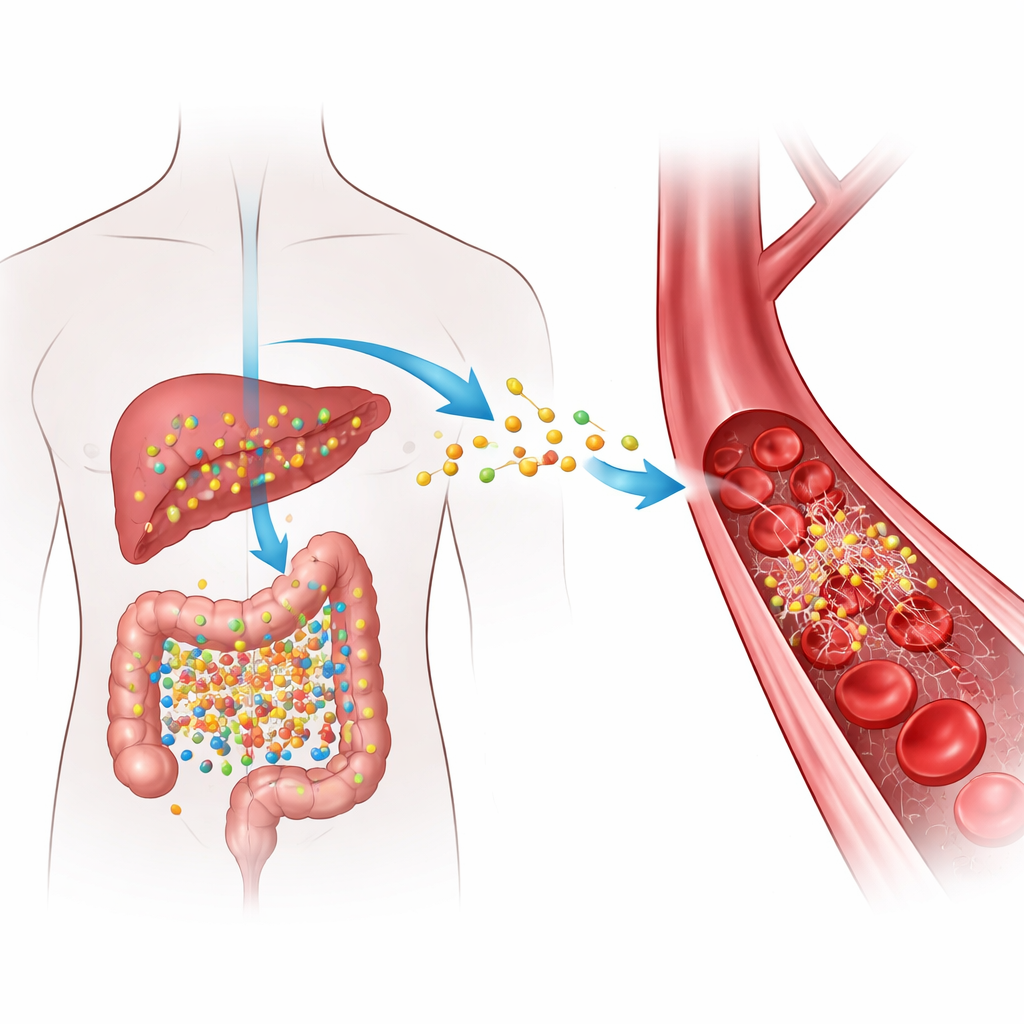
How Everyday Food Becomes a Risk Signal
When we eat foods rich in certain nutrients found in red meat, eggs, and high-fat dairy, gut bacteria convert these components into an intermediate compound that the liver then turns into TMAO. High levels of TMAO in the blood have been repeatedly associated with clogged arteries and a greater chance of dangerous clots, but the exact “how” has been unclear. The authors focused on fibrinogen, a soluble protein that circulates in blood and, when cut by the enzyme thrombin, turns into fibrin—the stringy material that helps seal injuries by forming clots. They asked whether TMAO physically interacts with fibrinogen in a way that could make clots form faster or become harder to dissolve.
Making Clots Faster and Harder to Clear
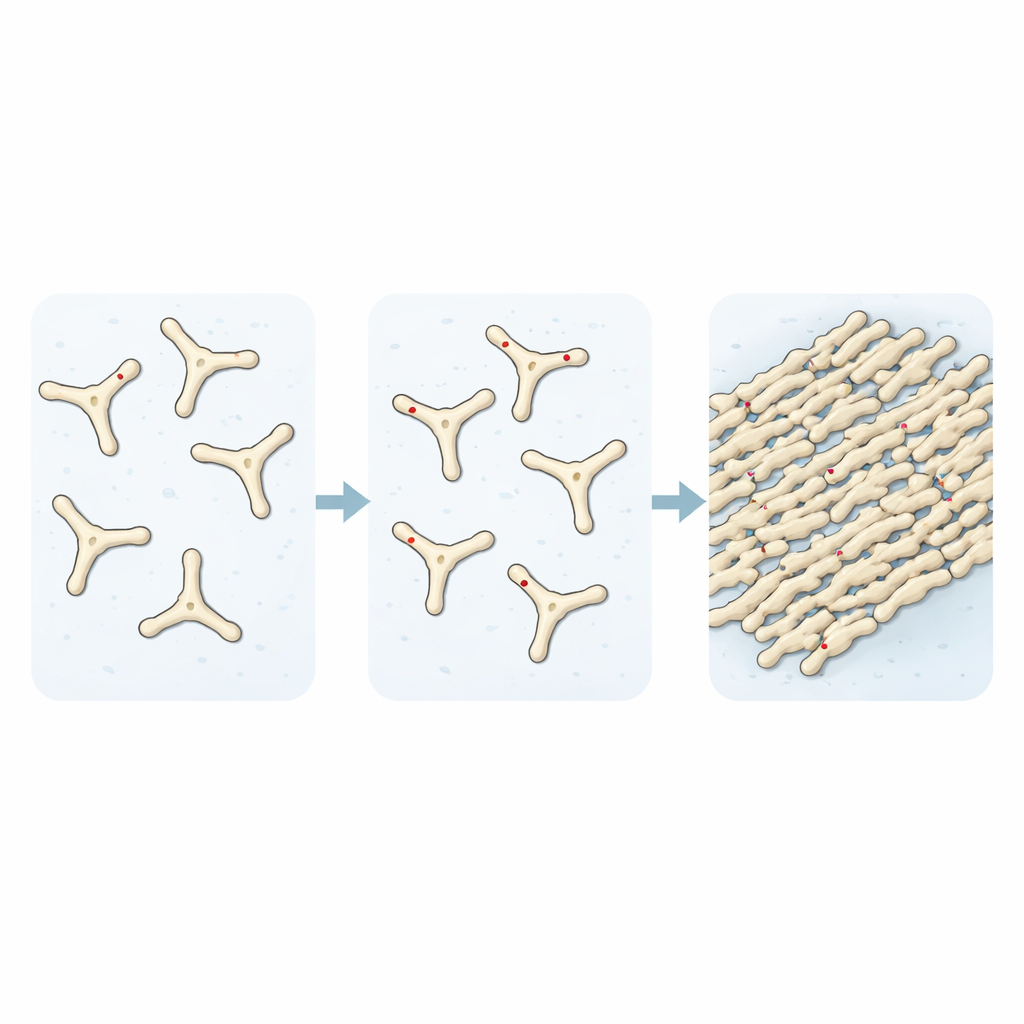
By following how solutions of fibrinogen turn into fibrin over time, the researchers showed that adding TMAO speeds up the assembly of early building blocks of clots. Light-scattering and fluorescent dye experiments revealed that, without TMAO, only about a third of the protein had joined into fibrils after a set period. With rising TMAO levels, that fraction climbed toward complete conversion, and microscope images showed a much denser web of fibers. When they examined how these clots broke down, they found another worrying feature: clots formed in the presence of TMAO were highly resistant to digestion by a protein-cutting enzyme, staying compact and largely intact while normal clots were almost completely degraded.
A Precise Docking Site on the Clotting Protein
To see whether this effect came from direct binding, the team measured heat changes as TMAO was mixed with fibrinogen and found clear evidence that the two interact with moderate strength at concentrations similar to those seen in people with heart disease. Spectroscopy methods indicated that fibrinogen’s shape became slightly loosened, exposing more sticky patches that favor protein–protein contact. Computer modeling and long simulations mapped where TMAO sits on fibrinogen and highlighted a specific region known as the beta1 calcium-binding site. When this site was occupied by natural calcium ions, TMAO could no longer exert its effect, and clot formation looked the same whether TMAO was present or not. This points to the beta1 site as a control point that tunes how easily fibrinogen molecules lock together.
Drug Metabolites Join the Story
The authors then asked whether other small molecules that resemble TMAO might behave in a similar way. Using chemical databases and docking software, they screened a set of related compounds and selected two that are N-oxide breakdown products of widely used antidepressant and nicotine-related drugs. In test-tube experiments, these metabolites also boosted the speed and extent of fibrin assembly, and produced thicker, denser fibrin networks, much like TMAO. This finding raises the possibility that some medications, once processed by the body, could subtly tip the balance toward more robust clot formation by acting on the same hotspot in fibrinogen.
What This Means for Heart and Stroke Risk
Putting all the evidence together, the work reveals a direct chain of events from gut-derived or drug-derived N-oxide molecules to changes in blood clot structure. By binding to a specific site on fibrinogen, TMAO and its chemical cousins nudge the protein into a conformation that encourages rapid fiber formation and produces clots that are tougher to break down. For a layperson, this means that certain microbial byproducts and drug metabolites may quietly harden the “scaffolding” of clots, helping to explain why elevated TMAO levels track with a higher chance of artery-blocking events. The beta1 site on fibrinogen emerges as a promising target for therapies aimed at softening clots and reducing complications from atherosclerosis without blocking clotting altogether.
Citation: Singh, K., Kumari, A., Bakhshi, R. et al. Gut metabolite TMAO and its structural analogs bind to fibrinogen thereby enhancing clot formation: a rationale for atherosclerosis risk. Sci Rep 16, 13676 (2026). https://doi.org/10.1038/s41598-026-43910-0
Keywords: gut microbiome, blood clots, TMAO, atherosclerosis, fibrinogen