Clear Sky Science · en
ASCC3 promotes chemosensitivity in colorectal cancer cells
Why this research matters
Chemotherapy is a mainstay treatment for advanced colorectal cancer, yet many tumors eventually learn to survive these harsh drugs. This study explores a lesser-known protein called ASCC3 and reveals that it unexpectedly makes colorectal cancer cells more vulnerable to common chemotherapy drugs. By uncovering how ASCC3 links DNA damage, cell metabolism, and stress signals inside the cell, the work points to possible new ways to predict which patients will respond to treatment—and how resistant tumors might be disarmed.
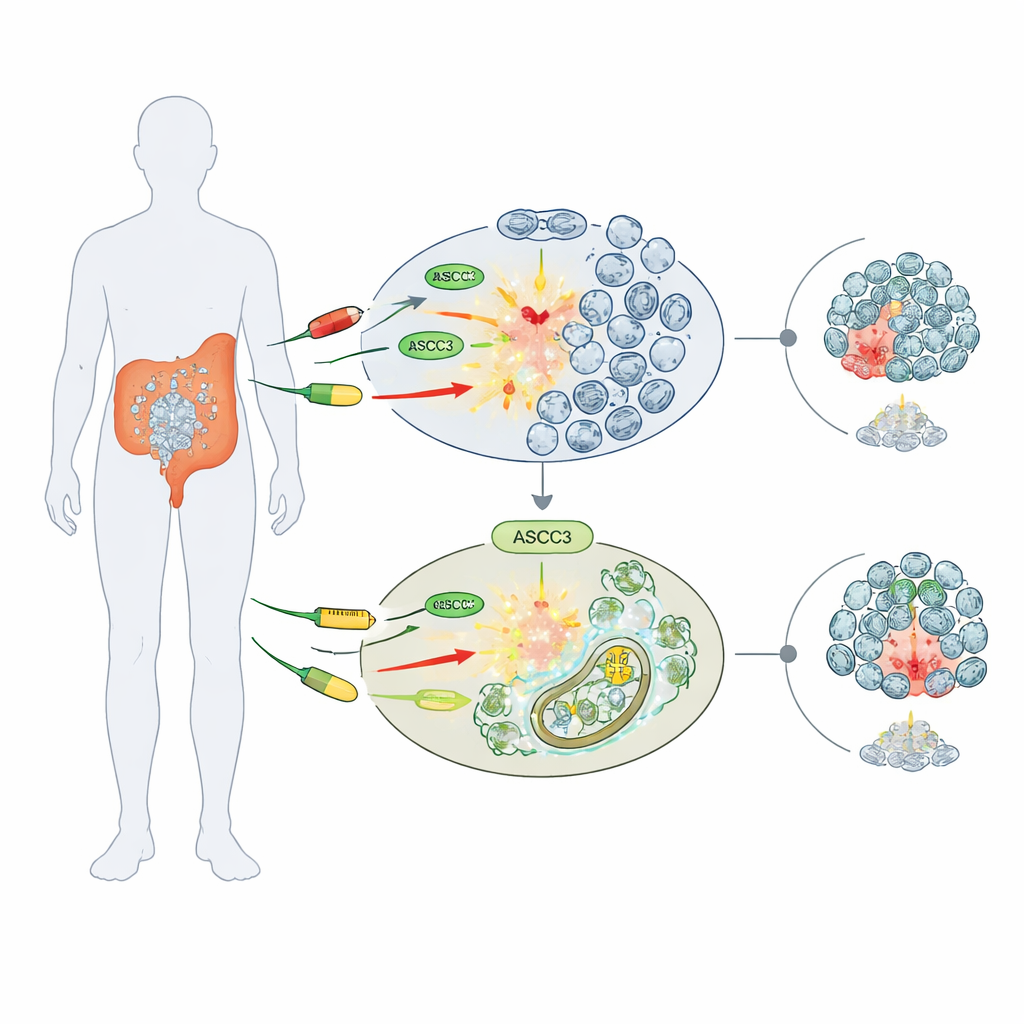
A gatekeeper for drug sensitivity
The researchers focused on three widely used chemotherapy agents that damage DNA or block its production: 5-fluorouracil (5-FU), cisplatin, and hydroxyurea. Working with several human colorectal cancer cell lines, they compared normal cells with cells genetically engineered to lack ASCC3. Surprisingly, cells without ASCC3 survived these drugs better, forming more colonies and retaining higher viability than their normal counterparts. This "chemoresistance" effect was specific: in bone cancer (osteosarcoma) cells, removing ASCC3 did not change drug sensitivity, suggesting that its role depends on cancer type.
More DNA damage, yet more survival
At first glance, the improved survival of ASCC3-deficient cells seems paradoxical. Earlier work showed that ASCC3 helps cells cope with stalled DNA replication, preventing broken chromosomes. In this study, when colorectal cancer cells were treated with chemotherapy, the loss of ASCC3 led to more signs of DNA damage—bright nuclear foci of damaged DNA and extra bits of chromosomes called micronuclei. In other words, the cells were genetically less stable, yet they were harder to kill. This disconnect suggested that other protective pathways, beyond simple DNA repair, were allowing the damaged cells to persist.
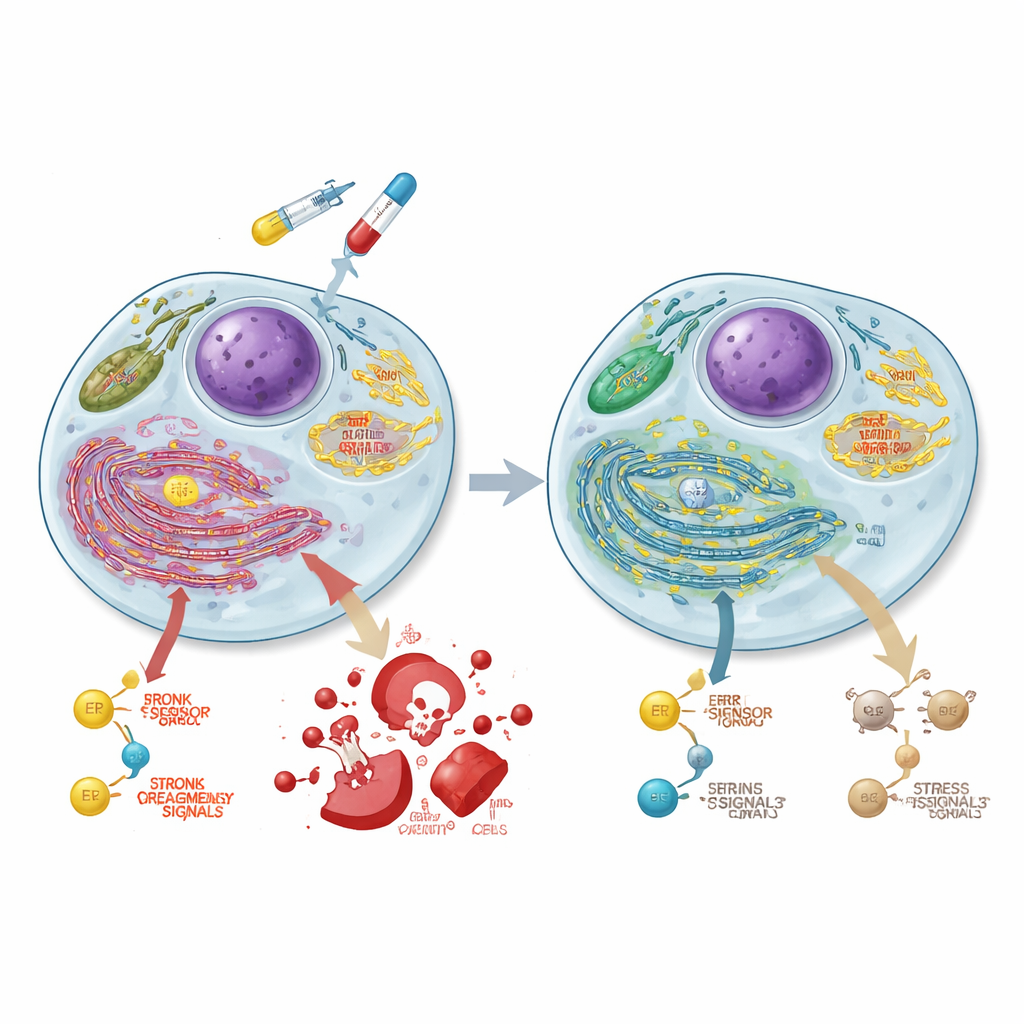
Rewiring how cancer cells make energy
To understand what else changes when ASCC3 is lost, the team measured gene activity across the entire genome. They found that ASCC3 normally boosts sets of genes involved in sugar breakdown (glycolysis), a growth-related signaling hub called mTORC1, and protein-folding pathways in the cell's endoplasmic reticulum. Using specialized metabolic tests, they showed that cells lacking ASCC3 shifted away from glycolysis and instead leaned heavily on their mitochondria, the cell's power plants, increasing oxygen-based energy production. Tracer experiments with labeled glucose confirmed that these cells channeled more sugar into the tricarboxylic acid (TCA) cycle and related amino acids, while reducing its use for certain building blocks like alanine and serine. ASCC3 also supported the cell's ability to make serine from scratch and to grow when this amino acid was scarce, though the exact mechanisms varied between cell lines.
Blunting a self-destruct stress signal
The endoplasmic reticulum (ER) monitors whether proteins are properly folded; when overloaded, it triggers stress responses that can either help cells adapt or push them toward death. One key ER sensor, PERK, can eventually activate a pro-death program involving the proteins CHOP and caspase 3. The researchers found that ASCC3 is essential for building up PERK protein during ER stress in colorectal cancer cells, even though it does not change PERK’s gene transcript or its breakdown rate. When cells were treated with 5-FU, normal cells showed strong increases in PERK, CHOP, and active caspase 3 over time, consistent with chemotherapy-induced cell death. In contrast, ASCC3-deficient cells failed to fully induce these proteins, and their death response was markedly reduced, helping explain their chemoresistance despite heavier DNA damage.
What this means for patients
Taken together, the study paints ASCC3 as a kind of internal ally of chemotherapy in colorectal cancer cells. When ASCC3 is present, it supports a more glycolysis-focused metabolism, robust serine production, and a strong ER-stress signal through PERK that can drive damaged cells toward self-destruction. When ASCC3 is lost, cells accumulate more genetic damage but survive by shifting their energy production toward mitochondria and blunting the PERK–CHOP death pathway. These insights suggest that ASCC3 levels could help predict which colorectal tumors will respond well to drugs like 5-FU and oxaliplatin, and that targeting the altered metabolism or stress responses of ASCC3-poor tumors may offer new strategies to overcome chemoresistance.
Citation: Tu, A., Martyn, R.D.C., Cui, S. et al. ASCC3 promotes chemosensitivity in colorectal cancer cells. Sci Rep 16, 13863 (2026). https://doi.org/10.1038/s41598-026-43173-9
Keywords: colorectal cancer, chemoresistance, cell metabolism, ER stress, PERK signaling