Clear Sky Science · en
Porphyromonas gingivalis promotes oral squamous cell carcinoma progression via the IL-6/EZH2/Snai2 axis
Why a gum germ matters for mouth cancer
Most people think of bleeding gums as a dental nuisance, not a cancer risk. This study shows how a common gum-disease bacterium, Porphyromonas gingivalis, can help drive the growth and spread of oral squamous cell carcinoma, a frequent and deadly form of mouth cancer. By tracing a chain of events from infection to runaway cell behavior, the researchers identify a molecular weak point that could be targeted to slow or prevent cancer progression.
A stubborn cancer with hidden helpers
Oral squamous cell carcinoma (OSCC) ranks among the ten most common cancers worldwide and is notoriously hard to cure; despite surgery, radiation, chemotherapy, and newer immunotherapies, fewer than half of patients are alive five years after diagnosis. In recent years, scientists have realized that the communities of microbes in our mouths do more than cause cavities or gum disease. When their balance is disturbed, certain bacteria can fan the flames of chronic inflammation, alter local immunity, and push cells toward more aggressive, mobile states. P. gingivalis, a key culprit in periodontitis, has repeatedly been linked to OSCC and to a cellular shift called epithelial–mesenchymal transition, in which orderly surface cells loosen, move, and invade nearby tissues.
From gum infection to powerful chemical signals
The authors began by asking whether P. gingivalis infection raises levels of the inflammatory messenger IL-6 in the mouths of patients. Measuring fluid from the crevice around the teeth, they found that IL-6 was much higher in both gum-disease patients carrying P. gingivalis and in people with OSCC than in healthy volunteers. They then isolated primary tumor cells from OSCC patients and exposed them to IL-6. Many of these cells increased their levels of the IL-6 receptor, making them more responsive to the signal. The receptor-high cells divided faster and migrated more readily, clear hallmarks of a more dangerous cancer cell population.
A three-step relay inside tumor cells
Diving deeper, the team focused on two proteins inside cancer cells: EZH2, an enzyme that controls how DNA is packaged and which genes are turned on, and Snai2, a transcription factor known to push cells into a more mobile, invasive state. They found that OSCC cells already had more EZH2 and Snai2 than normal oral cells, and that IL-6 stimulation further boosted both proteins at the RNA and protein levels. When they artificially increased EZH2 in a standard OSCC cell line, the cells grew faster and moved more. Knocking down EZH2 had the opposite effect, but adding back EZH2 protein restored the aggressive behavior. Crucially, changes in EZH2 levels directly shifted Snai2 gene activity in the same direction, showing that EZH2 sits upstream of Snai2 in this relay.
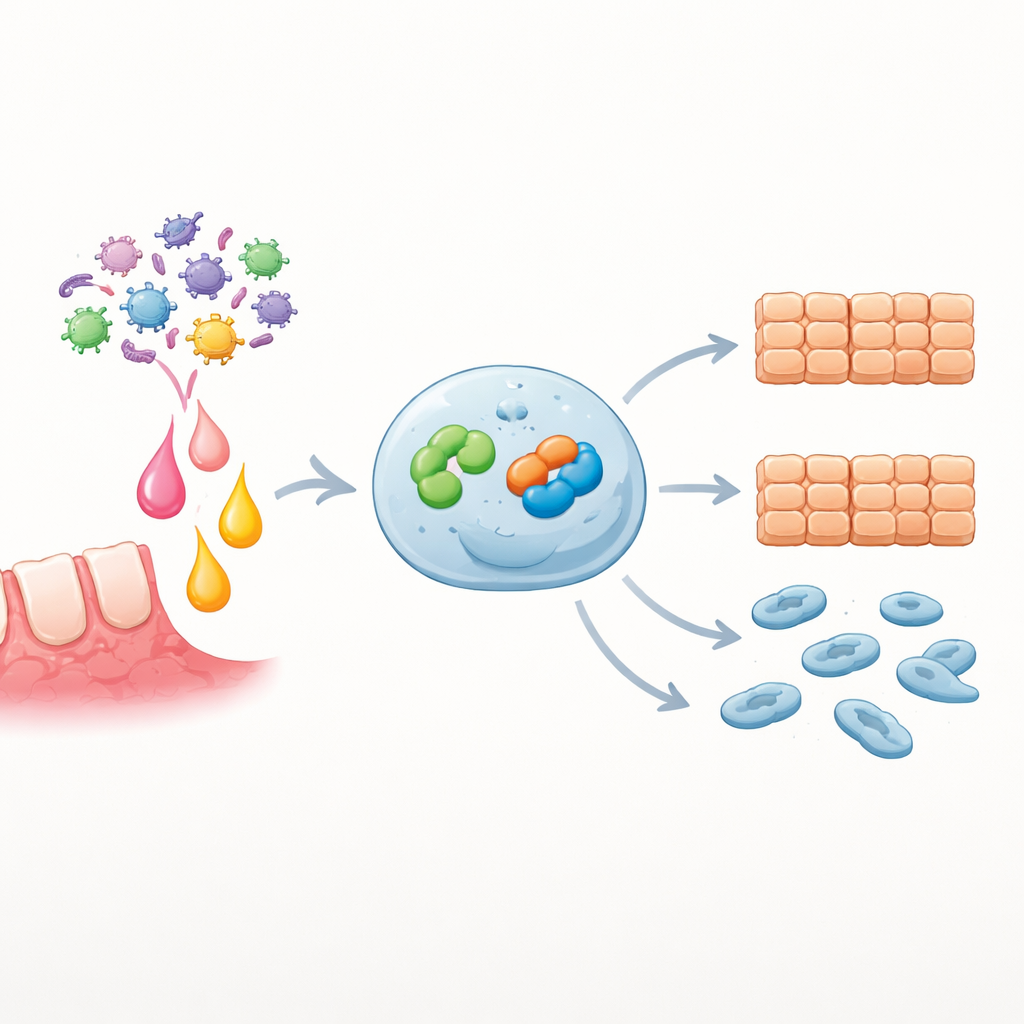
Blocking the relay slows cancer traits
To test whether Snai2 is required for EZH2’s effects, the researchers boosted EZH2 but simultaneously silenced Snai2. Under these conditions, cell growth, wound closure, and migration were all significantly reduced compared with EZH2 overexpression alone, indicating that Snai2 is a key engine of the aggressive traits driven by EZH2. In primary tumor cells that responded to IL-6, adding either a chemical inhibitor of EZH2 or small RNA molecules that knock down Snai2 sharply blunted IL-6-induced proliferation and migration. Bioinformatic analyses of large patient datasets reinforced these lab findings: high SNAI2 expression was enriched in pathways related to infection and cell movement, and patients whose tumors had more SNAI2 tended to have poorer survival.
What this means for patients and prevention
Taken together, the work outlines a clear story in simple terms: a gum-disease bacterium raises IL-6 levels in the mouth; IL-6 switches on EZH2 inside nearby tumor cells; EZH2 in turn cranks up Snai2, which helps cancer cells break away and spread. This IL-6/EZH2/Snai2 chain links everyday oral infections to life-threatening cancer behavior and highlights new intervention points. In the future, better control of chronic gum infection, drugs that dampen IL-6 signals, or targeted inhibitors of EZH2 or Snai2 might help slow OSCC progression. While these findings still need to be confirmed in animal models and clinical studies, they offer a concrete molecular bridge between oral hygiene and cancer risk—and a promising set of targets for more effective, biology-based treatments.
Citation: Liu, S., Guan, T., Xu, B. et al. Porphyromonas gingivalis promotes oral squamous cell carcinoma progression via the IL-6/EZH2/Snai2 axis. Sci Rep 16, 10296 (2026). https://doi.org/10.1038/s41598-026-41528-w
Keywords: oral cancer, gum disease, Porphyromonas gingivalis, inflammation, cancer signaling