Clear Sky Science · en
Pramipexole alleviates ferroptosis in HT22 cells induced by oxygen–glucose deprivation/reoxygenation via the Nrf2/SLC7A11/GPX4 pathway
Why protecting brain cells after stroke matters
When a person has an ischemic stroke, a blood clot starves part of the brain of oxygen and sugar. Doctors work urgently to reopen the blocked vessel, but ironically, the sudden return of blood can itself damage fragile brain cells. This study explores whether pramipexole, a drug already used for Parkinson’s disease, can shield nerve cells from this second wave of harm by calming a recently discovered form of cell death linked to iron and runaway oxidation.
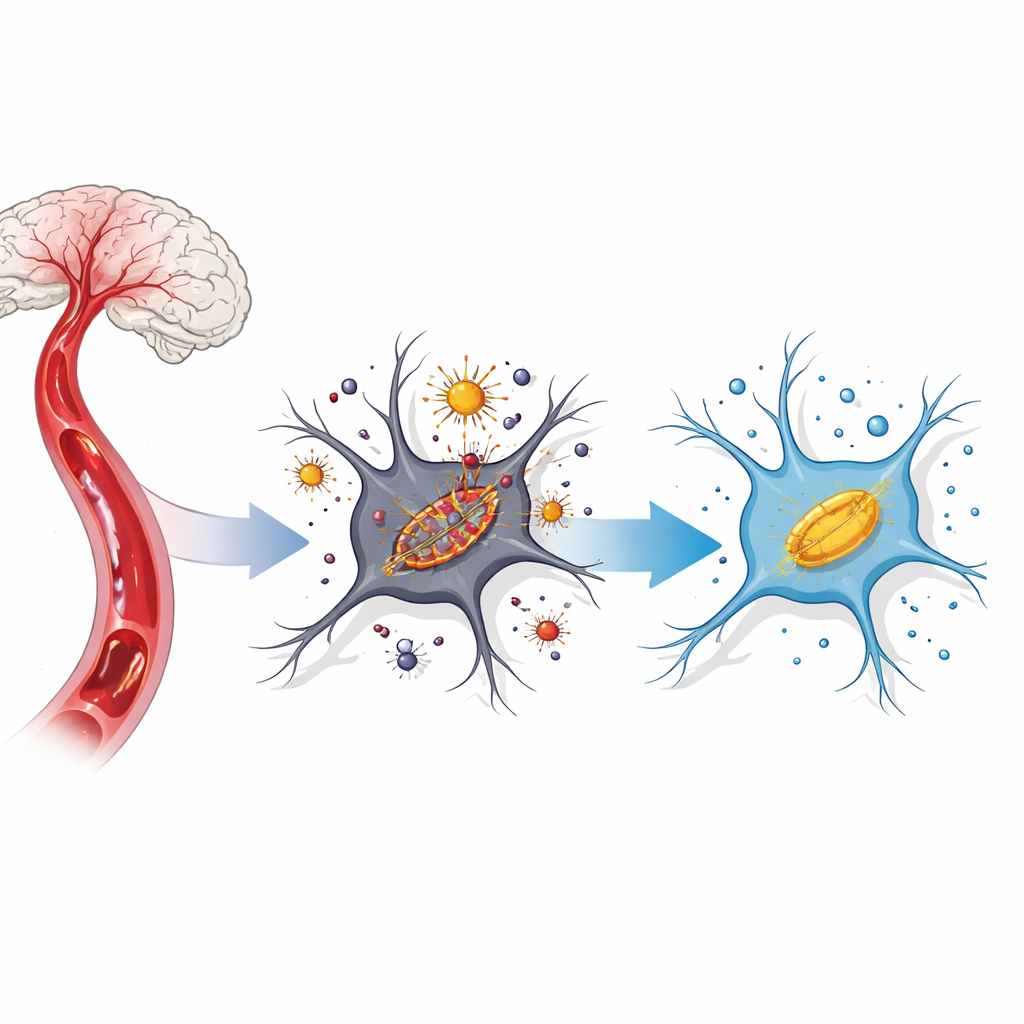
A harmful chain reaction inside brain cells
After blood flow is cut off and then restored, brain cells experience a storm of chemical stress. Iron that is normally locked away becomes loose inside the cell and reacts with oxygen to generate aggressive molecules known as reactive oxygen species. These attack cell membranes rich in fats, especially in tiny powerhouses called mitochondria. Scientists now call this iron‑driven, membrane‑focused cell death “ferroptosis.” In stroke models, ferroptosis appears when iron builds up, protective antioxidants drop, and mitochondria become shrunken and dysfunctional.
Testing pramipexole in a stroke-like lab model
To mimic what happens during a stroke and subsequent treatment, the researchers used mouse hippocampal nerve cells (HT22 cells) and briefly deprived them of both oxygen and glucose before restoring normal conditions. This oxygen–glucose deprivation/reoxygenation model reproduces many features of ischemia–reperfusion injury. The team exposed cells to different doses and timings of pramipexole and measured survival, mitochondrial health, iron levels, reactive oxygen species, and key antioxidant systems. They also used erastin, a chemical that specifically triggers ferroptosis, to see whether pramipexole could counteract this process directly.
How the drug restores balance inside cells
Without protection, the stroke-like treatment reduced cell survival, disrupted mitochondrial membrane potential, increased iron and reactive oxygen species, and depleted the antioxidant molecule glutathione along with the enzyme GPX4 that depends on it. Under the electron microscope, mitochondria appeared smaller and darker, matching hallmarks of ferroptosis. Pramipexole, particularly at a moderate dose given during the reoxygenation phase, reversed many of these changes: cells survived better, mitochondria retained their shape and electrical charge, iron and oxidants declined, and glutathione and GPX4 activity rebounded. Similar benefits were seen when cells were damaged by erastin, strengthening the case that pramipexole acts by restraining ferroptosis rather than only by general antioxidation.
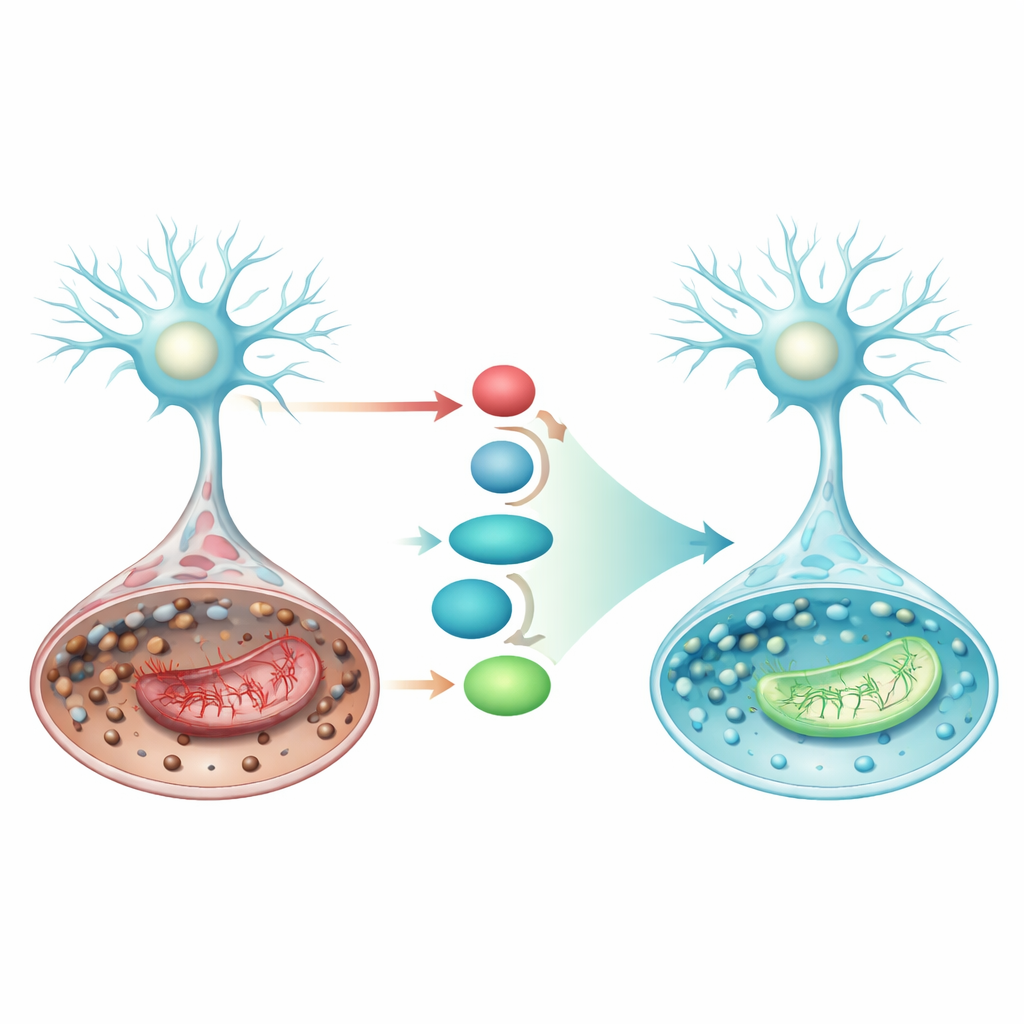
Switching on an internal defense pathway
The study also focused on a built‑in cellular defense line involving the protein Nrf2 and its downstream partners. When activated, Nrf2 moves into the cell nucleus and boosts the production of SLC7A11, a transporter that brings in raw material for glutathione, and GPX4, the enzyme that uses glutathione to neutralize lipid peroxides. In the stroke-like condition, SLC7A11 and GPX4 levels fell, tilting cells toward ferroptosis. Pramipexole strongly increased Nrf2, SLC7A11, and GPX4 at both the protein and gene levels and, at the same time, dampened p53, a protein that can push cells further toward ferroptosis and mitochondrial failure. These coordinated shifts suggest that pramipexole strengthens the Nrf2/SLC7A11/GPX4 axis, rebuilding the cell’s antioxidant shield exactly where it is most needed.
What this could mean for future stroke care
Taken together, the findings show that pramipexole helps nerve cells weather the oxidative storm that follows restored blood flow, mainly by blocking ferroptosis and preserving mitochondrial function. Although this work was done in cultured cells rather than patients, it points to a promising new way to repurpose an existing brain drug for stroke: using it not just to affect dopamine signaling, but to stabilize iron handling and membrane integrity through the Nrf2/SLC7A11/GPX4 pathway. If similar protection can be confirmed in animal models and clinical trials, pramipexole or related compounds might one day be added to reperfusion therapies to reduce lasting brain damage after ischemic stroke.
Citation: Zhang, L., Kang, X., Wang, Q. et al. Pramipexole alleviates ferroptosis in HT22 cells induced by oxygen–glucose deprivation/reoxygenation via the Nrf2/SLC7A11/GPX4 pathway. Sci Rep 16, 11134 (2026). https://doi.org/10.1038/s41598-026-40289-w
Keywords: ischemic stroke, ferroptosis, pramipexole, neuronal protection, oxidative stress