Clear Sky Science · en
Quemliclustat and chemotherapy with or without zimberelimab in metastatic pancreatic adenocarcinoma: a randomized phase 1 trial
Why this study matters
Pancreatic cancer is one of the deadliest cancers, in part because it is usually found after it has spread and because standard chemotherapy buys patients only a short amount of extra time. This study tests a new drug strategy that aims not only to attack tumor cells directly, but also to lift a kind of chemical "fog" that keeps the immune system from helping. By pairing a targeted medicine called quemliclustat with routine chemotherapy, and sometimes with an immune‑boosting antibody, the researchers ask whether they can safely extend survival and better harness the body’s own defenses.
A chemical shield around the tumor
Pancreatic tumors create a harsh neighborhood that makes it hard for immune cells to do their job. When chemotherapy kills cancer cells, they spill a molecule called ATP into the surrounding tissue. An enzyme on cell surfaces, known as CD73, quickly converts ATP into adenosine, a substance that normally helps calm inflammation and protect healthy tissue. Inside tumors, however, high adenosine levels act like a chemical shield, dampening immune attack and allowing cancer cells to persist. The new pill‑like drug quemliclustat blocks CD73, aiming to reduce adenosine and make the tumor environment less welcoming to cancer and more inviting to immune cells. 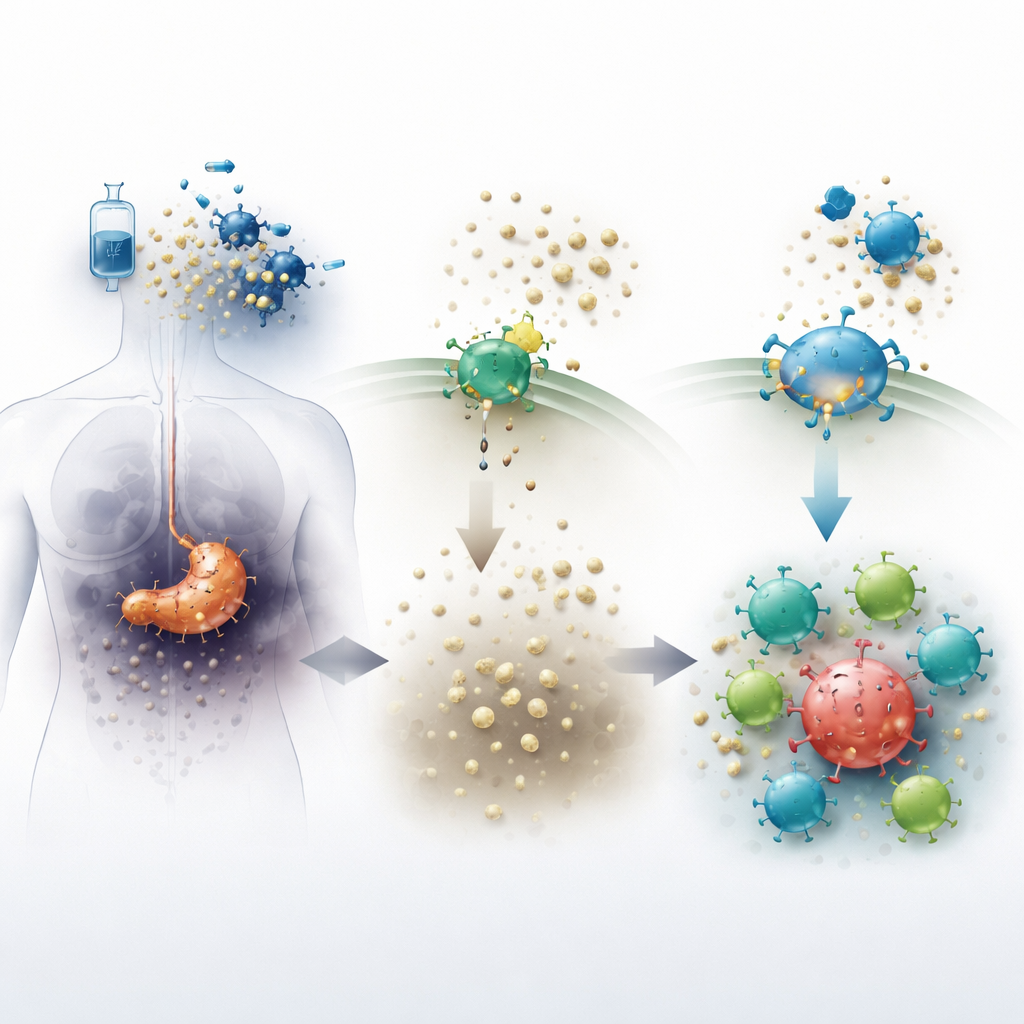
Testing a new combination in patients
The ARC‑8 trial enrolled people whose pancreatic cancer had already spread and who had not yet received treatment for metastatic disease. All participants received a standard chemotherapy mix of gemcitabine and nab‑paclitaxel. In early dose‑finding groups, patients also received quemliclustat together with an immune‑checkpoint antibody called zimberelimab, which can release molecular brakes on immune cells. After establishing a safe dose of quemliclustat, the study expanded into larger groups in which patients were randomly assigned to receive chemotherapy plus quemliclustat either with or without zimberelimab. The main goals were to assess safety and to look for early hints of benefit, such as tumor shrinkage and survival.
Safety and survival signals
Across all treatment arms, side effects looked very similar to what doctors expect from this chemotherapy regimen alone, such as fatigue, nausea and low blood counts. Serious immune‑related complications were uncommon, and most severe problems were linked to chemotherapy rather than to the new drugs. When the researchers pooled patients who received the recommended dose of quemliclustat, they found that tumors shrank in roughly a third of patients and remained under control in about three‑quarters. Median overall survival reached about 16 months in this group, notably longer than the roughly 9 to 12 months seen in several recent trials of chemotherapy alone. A careful comparison with a “synthetic” control group built from previous studies suggested a survival advantage of almost six months for patients receiving the quemliclustat combination.
Cracking how the drug reshapes the tumor neighborhood
To understand why some patients benefited more than others, the team looked at gene activity patterns in tumor biopsies and in lab‑grown cells. They focused on a set of genes called the NR4A family, which light up when adenosine levels are high. In dishes, adding adenosine or its precursors boosted NR4A genes in multiple cell types found in pancreatic tumors, while blocking CD73 or adenosine receptors kept this signal down. In patient samples, chemotherapy itself increased NR4A activity in many tumor‑associated cells, consistent with a surge of adenosine after cell damage. Strikingly, patients whose tumors started out with higher NR4A levels tended to live longer on quemliclustat therapy, but this pattern did not hold in two outside trials where patients received similar chemotherapy without quemliclustat. 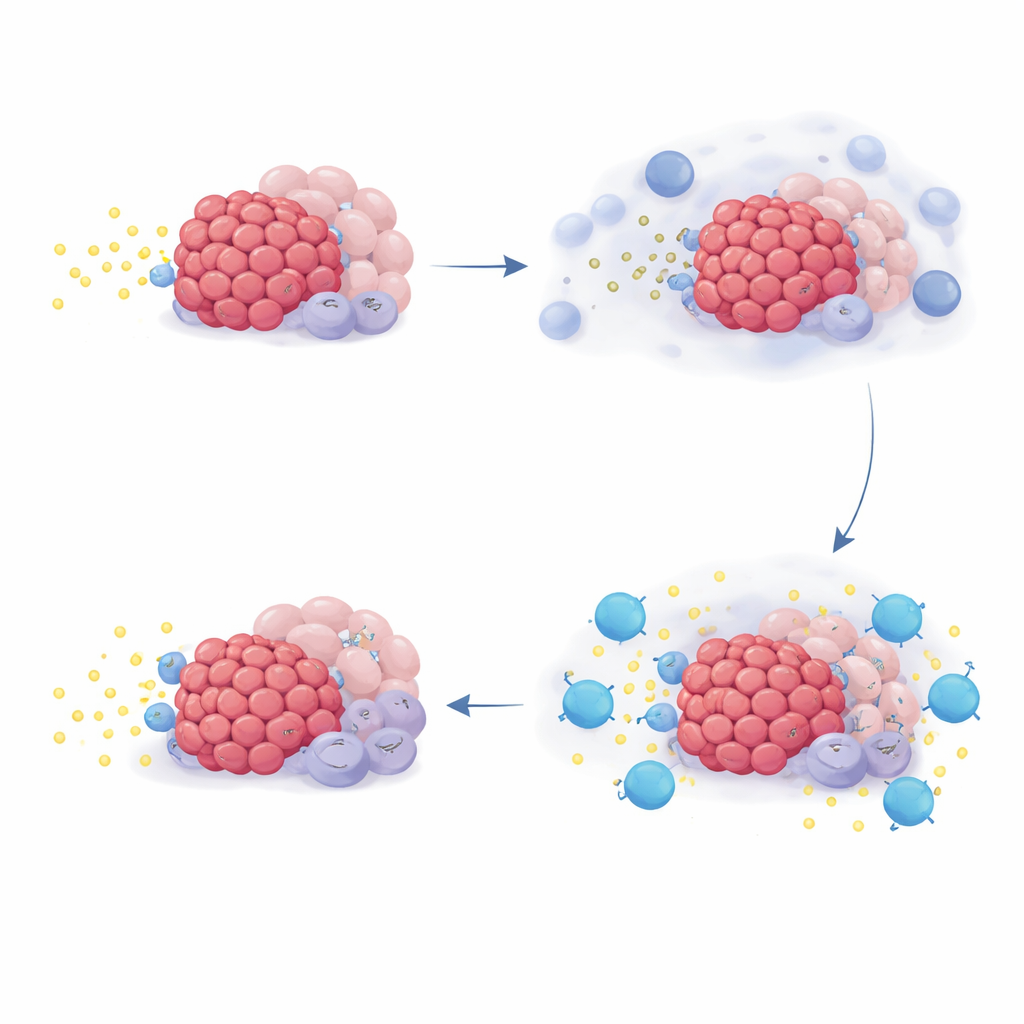
Bringing immune cells back to the fight
Advanced imaging of tumor slices revealed that areas with high NR4A levels had fewer activated killer T cells nearby, suggesting that adenosine creates a gradient that keeps these cells at a distance. In patients who had biopsies taken before and after treatment, quemliclustat generally reduced NR4A signals in tumors. Those with the greatest drop showed the strongest rise in gene markers of T cell presence and activity, and they also enjoyed the best overall survival, with more than half still alive beyond two years. Together, these findings support the idea that blocking CD73 thins the adenosine “fog,” allowing immune cells to move closer to cancer cells and attack them more effectively.
What this could mean for patients
This early‑phase trial was designed mainly to test safety, and it lacks a traditional randomized chemotherapy‑only control group, so the results cannot yet prove that quemliclustat extends life. Still, the combination appeared tolerable and produced survival times that compare favorably with past studies. The biological analyses link drug exposure, changes in tumor chemistry and immune activation to patient outcomes, strengthening the case that targeting adenosine is a promising strategy. A larger phase 3 trial now underway will test whether adding quemliclustat to standard chemotherapy truly improves survival for people facing metastatic pancreatic cancer.
Citation: Wainberg, Z.A., Manji, G.A., Bahary, N. et al. Quemliclustat and chemotherapy with or without zimberelimab in metastatic pancreatic adenocarcinoma: a randomized phase 1 trial. Nat Med 32, 1267–1277 (2026). https://doi.org/10.1038/s41591-026-04283-z
Keywords: pancreatic cancer, tumor microenvironment, immunotherapy, adenosine pathway, clinical trial