Clear Sky Science · en
TGF-β mediates epigenetic control of innate antiviral responses and SIV reservoir size
Why this research matters for people living with HIV
Even with today’s powerful HIV medicines, a small pool of virus hides silently inside immune cells and can rekindle infection if treatment stops. This study in rhesus macaques explores a new way to shrink that hidden reservoir by retraining the body’s first line of defense—the innate immune system—rather than focusing only on classic virus-fighting T cells and antibodies. The work also helps explain why a tiny fraction of people, known as elite controllers, can keep HIV in check without drugs.
A tug-of-war inside immune cells
The researchers started from an important clinical problem: when antiretroviral therapy is paused, HIV usually roars back from long-lived infected cells. Earlier work showed that combining two antibody drugs that block the immune brakes IL-10 and PD-1 helped most infected macaques control viral rebound, and a subset even showed a marked drop in the amount of viral DNA hidden in their CD4 T cells. In this paper, the team asked what distinguished animals whose reservoirs shrank from those whose reservoirs stayed large, focusing on the behavior and “programming” of innate immune cells and helper T cells in lymph nodes and blood.
Turning up the body’s natural antiviral alarm
Using broad gene activity profiling and single-cell analysis, the scientists found that animals with shrinking reservoirs had switched on powerful antiviral circuits well before therapy was interrupted. Genes typically triggered by interferons—the body’s antiviral alarm molecules—were highly active in many immune cell types, including monocytes, dendritic cells, and multiple T cell subsets. These genes encode “restriction factors” that block viruses at different steps of their life cycle. Laboratory experiments with human CD4 T cells showed that pretreatment with interferon made both directly exposed cells and their neighbors far more resistant to HIV infection, underscoring how a primed innate response can shield bystander cells when virus reappears.
How a calming signal can backfire
In stark contrast, animals whose viral reservoirs remained large had high blood levels of the immune-dampening molecule TGF-β. Detailed pathway analysis linked TGF-β to activation of SMAD proteins and a suite of enzymes, especially certain histone deacetylases (HDACs), that tighten DNA packaging inside cells. This epigenetic tightening made key antiviral genes harder to access and turn on. When human memory CD4 T cells were exposed to plasma rich in TGF-β, or to TGF-β itself, they ramped up HDAC11, became poorer at responding to interferon signals, and were more readily infected by HIV in vitro. Blocking TGF-β or inhibiting HDACs reversed these effects, reopening access to antiviral genes and reducing infection.
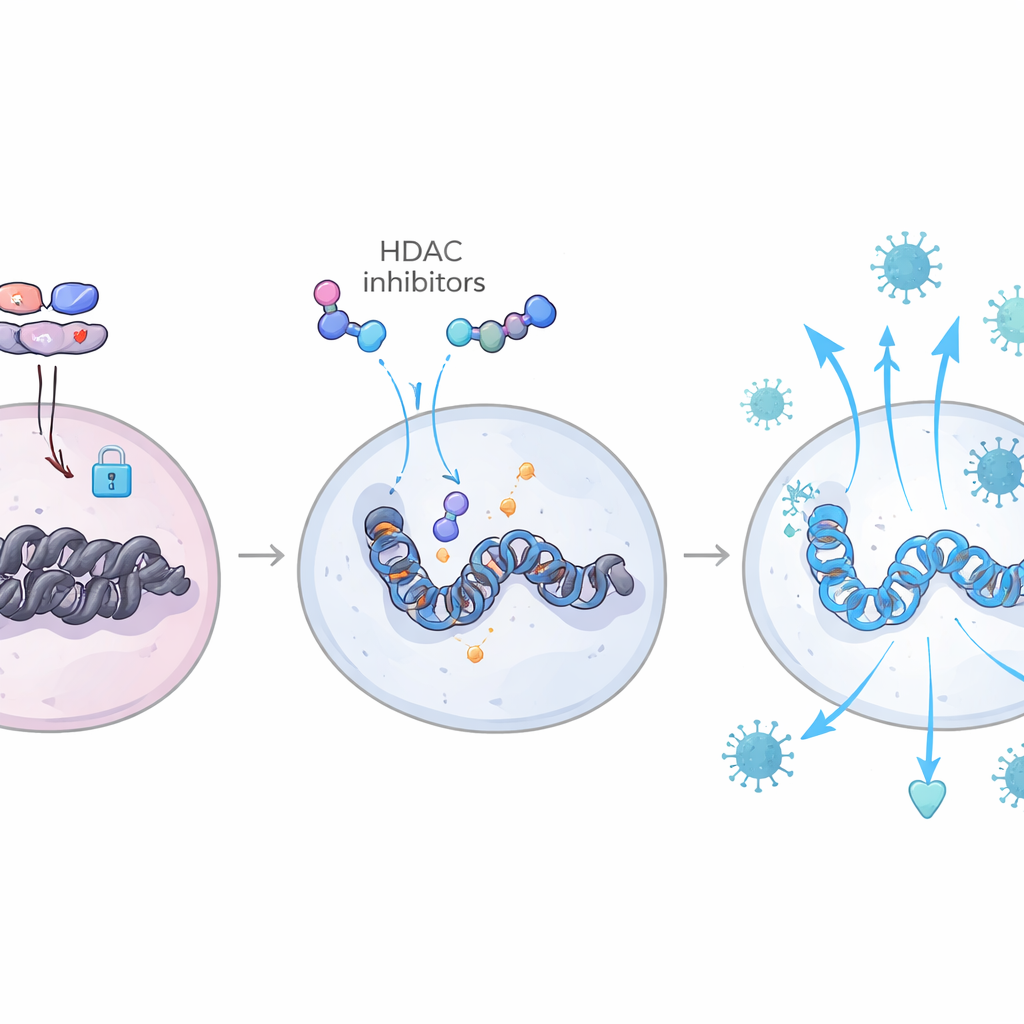
Rewriting the immune playbook through epigenetics
By combining measurements of gene activity with maps of chromatin openness in individual cells, the team could see these tug-of-war forces directly in the genome. In animals with shrinking reservoirs, binding sites for interferon-linked transcription factors such as IRF and STAT families were more accessible across T cells and myeloid cells, while binding sites used by TGF-β–associated AP-1 and SMAD complexes were relatively closed. These animals also showed increased activity of a pathway involving IL-6 and the factor C/EBP-β, which further amplifies interferon responses and helps keep TGF-β in a restrained, inactive form. Together, this created a durable, “trained” antiviral state that limited new rounds of infection when drugs were stopped. Importantly, many of the same gene signatures appeared in blood cells from human HIV elite controllers, linking the macaque findings to a naturally occurring form of viral control.
What this means for future HIV cure strategies
To a non-specialist, the main message is that the size of the hidden HIV reservoir is not fixed—it is strongly influenced by how innate immune cells are wired at the level of their DNA packaging. When TGF-β dominates, it recruits epigenetic enzymes that lock down antiviral genes, leaving cells vulnerable and allowing the viral reservoir to persist. When interferon-driven programs and IL-6–mediated amplification win out, many potential target cells become harder to infect, and the reservoir shrinks. Because drugs that block PD-1 and HDACs, and agents that boost interferon pathways, already exist in cancer and other fields, these results point toward combination therapies that could reprogram innate immunity, curb TGF-β’s suppressive influence, and gradually erode HIV’s last strongholds in the body.
Citation: Ghneim, K., ten-Caten, F., Santana, A.C. et al. TGF-β mediates epigenetic control of innate antiviral responses and SIV reservoir size. Nat Immunol 27, 686–699 (2026). https://doi.org/10.1038/s41590-026-02458-x
Keywords: HIV reservoir, innate immunity, interferon signaling, TGF-beta, epigenetic reprogramming