Clear Sky Science · en
Distinct origins and niches determine the cellular responsiveness of CNS macrophages after repopulation
Why brain border guardians matter
Our brains are not only made of nerve cells. They are also patrolled by immune cells that sit either deep inside the tissue or at its borders, where the brain meets its protective membranes and blood vessels. This study explores how different groups of these immune sentinels disappear and then grow back, and shows that the way they are replaced can change how the brain responds to inflammation and stroke. Understanding this regrowth opens the door to therapies that tweak border immune cells without disturbing the rest of the brain.
Different neighborhoods, different brain sentinels
Immune cells called macrophages occupy distinct “neighborhoods” in the central nervous system. Microglia live within the brain tissue itself, while so‑called border macrophages sit along the brain’s surfaces and around blood vessels. Among these border cells, subdural macrophages lie just above the brain tissue, hugging the surface and perivascular spaces, whereas another group populates the tough outer membrane called the dura. Although these populations all help monitor and shape immune responses in the brain, they have different origins and lifespans, and earlier work suggested that microglia can maintain themselves locally without help from blood‑borne cells.
Resetting the system and watching it regrow
To see how each macrophage group recovers after being wiped out, the researchers used a drug that blocks a key survival signal and temporarily depletes these cells in mice. They then tracked how and where the cells came back using a combination of genetic “barcode” mice, single‑cell RNA sequencing, chromatin profiling and high‑resolution microscopy. Microglia in the brain tissue bounced back quickly and almost completely by dividing from the rare survivors that escaped depletion, and there was no sign that circulating immune cells moved in to replace them. In contrast, subdural macrophages at the brain surface repopulated slowly and unevenly across regions, hinting that they were drawing on additional sources beyond the original resident cells. 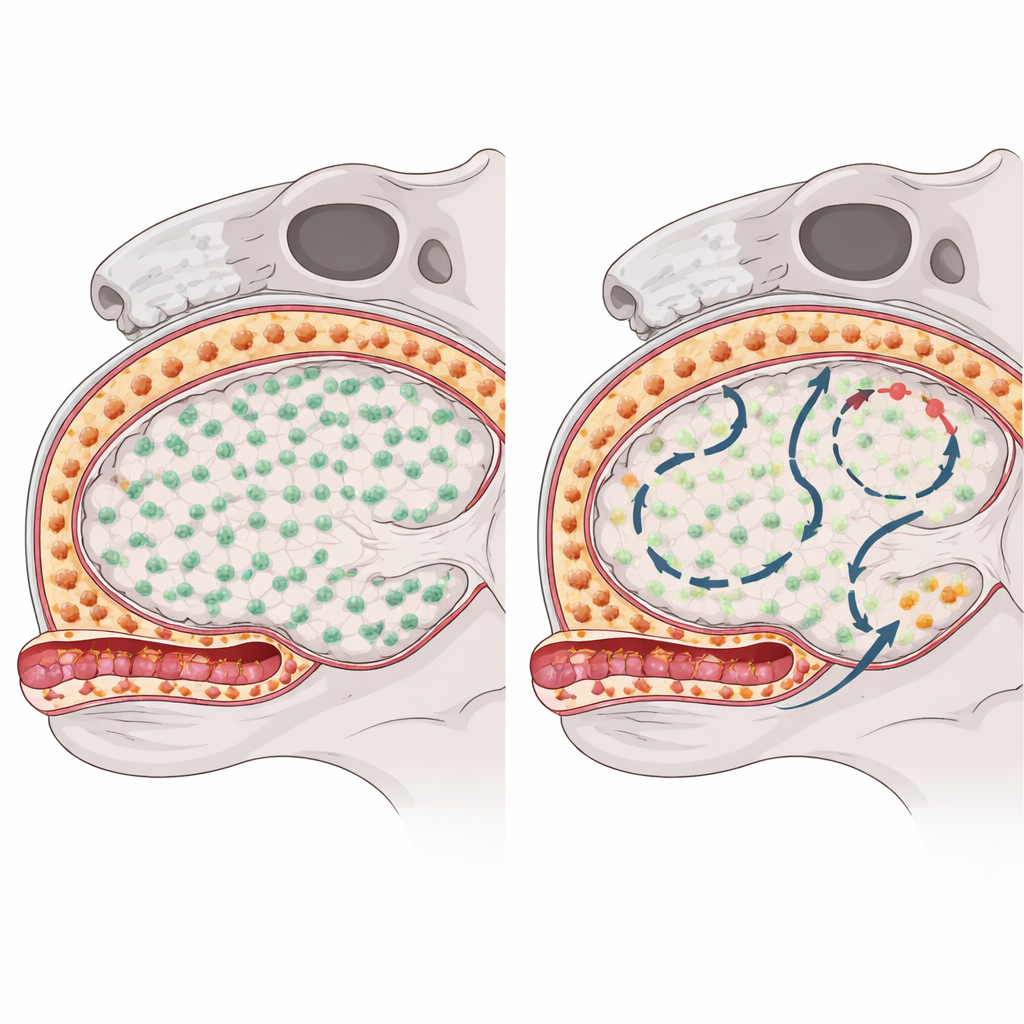
When blood cells move in and stay
Fate‑mapping experiments showed that, unlike microglia, subdural macrophages partially refilled their niches using short‑lived monocytes that circulate in the blood. After depletion, signals from blood vessel and stromal cells created a temporary window in which these monocytes could stick to activated vessel walls, crawl across them with the help of adhesion molecules such as ICAM‑1 and VCAM‑1, and settle into perivascular and surface niches. Blocking these adhesion pathways reduced the number of incoming cells, confirming their importance. Once engrafted, monocyte‑derived border macrophages did not simply mimic the original embryo‑derived cells: they kept a distinct gene expression and chromatin pattern, were enriched for molecules involved in antigen presentation and cell adhesion, and remained as a stable part of the population long after the initial injury had apparently healed.
Lasting rewiring of border immunity
Single‑cell and bulk sequencing revealed that microglia largely regained their original molecular profile after repopulation, with minimal long‑term changes. Subdural macrophages, however, were durably rebalanced toward a more “alert” subtype that expressed higher levels of immune activation genes. By separating embryo‑derived and monocyte‑derived cells using genetic labels, the team showed that developmental origin strongly shaped both gene activity and accessible DNA regions in these cells. Monocyte‑derived border macrophages carried epigenetic marks typical of recently differentiated blood cells and showed heightened accessibility at genes involved in sensing pathogens and interacting with other immune cells. When the animals were challenged with bacterial components, these newcomer cells mounted stronger inflammatory and stress responses than their embryo‑derived neighbors, while the reactions of microglia and dural macrophages were much less altered. 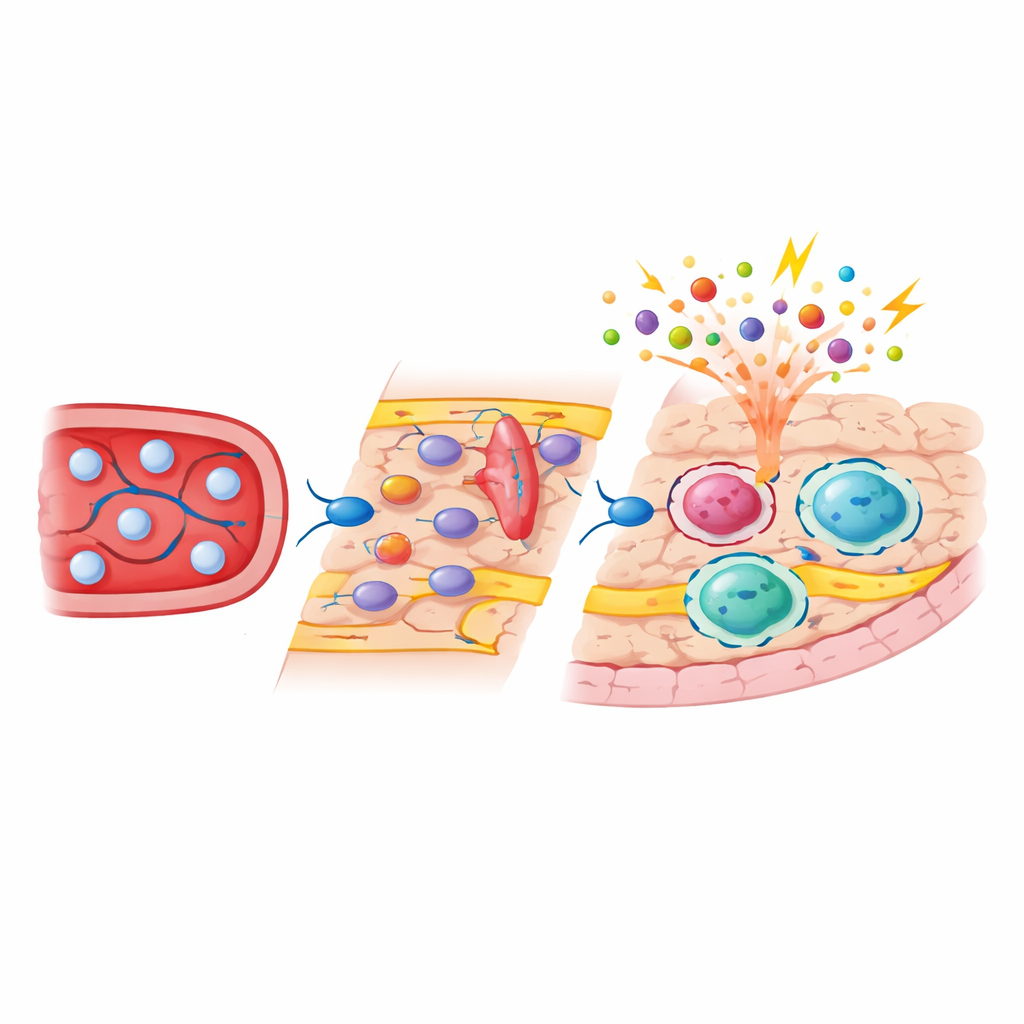
Consequences for stroke and brain health
The authors then asked whether this hidden reshaping of border macrophages affects disease. In a model of ischemic stroke, mice whose subdural macrophages had been repopulated performed worse in behavioral tests, even though lesion size and blood vessel reopening were similar to controls. Analysis of whole tissue and isolated border macrophages showed stronger induction of genes linked to inflammation, reactive oxygen species and blood–brain barrier disruption in repopulated animals. Monocyte‑derived border cells were especially reactive, producing more chemokines and activation markers after stroke. Imaging of blood vessel activation confirmed a higher signal for P‑selectin, a marker of intense vascular inflammation, which correlated with poorer motor recovery.
What this means for future treatments
This work shows that not all brain macrophages are equal, and that replacing them does not simply reset the system. Microglia can quietly rebuild themselves from surviving cells and return to business as usual. By contrast, border macrophages at the brain surface become a permanent mix of original, embryo‑derived cells and new arrivals from the blood, and this mixed population responds more aggressively to systemic inflammation and stroke. Because the researchers developed a way to selectively exchange these border cells while sparing microglia, their findings point to a future in which therapies could fine‑tune the brain’s immune borders—either damping down harmful inflammation in disorders like stroke and neurodegeneration or boosting protective responses—without globally disrupting the brain’s internal immune network.
Citation: Fliegauf, M., Levard, D., Cardamone, F. et al. Distinct origins and niches determine the cellular responsiveness of CNS macrophages after repopulation. Nat Immunol 27, 961–974 (2026). https://doi.org/10.1038/s41590-026-02457-y
Keywords: brain border macrophages, microglia repopulation, monocyte engraftment, neuroinflammation, ischemic stroke