Clear Sky Science · en
Trafficking of a nitrogenase FeMo-cofactor assembly intermediate
How Microbes Help Feed the World
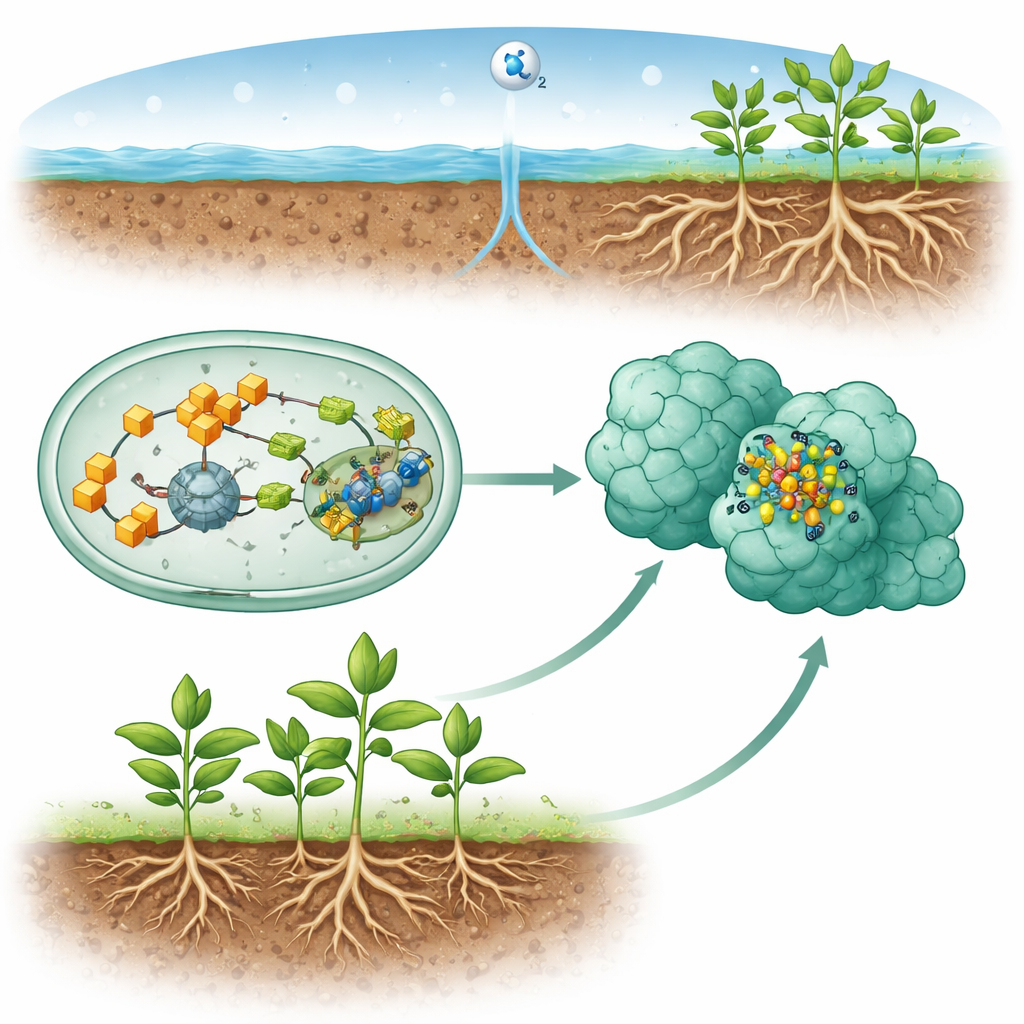
Nitrogen is a basic ingredient of DNA and proteins, yet most living things cannot use the huge reservoir of nitrogen gas in the air. Instead, they depend on specialized microbes that convert this gas into forms plants can digest, silently propping up global agriculture and natural ecosystems. This article peeks into one of the most intricate molecular machines behind that trick and asks a simple question: how do cells carefully build and hand off the tiny metal cluster at the heart of the nitrogen-fixing enzyme without dropping it along the way?
The Tiny Engine Inside Nitrogen-Fixing Bacteria
Many soil and aquatic microbes use an enzyme called nitrogenase to convert nitrogen gas into ammonia, which plants then use as fertilizer. The most common version of this enzyme relies on a complex metal cluster called the FeMo-cofactor, built from iron, sulfur, carbon, molybdenum, and an organic side piece known as homocitrate. This cofactor does not assemble itself inside the finished enzyme. Instead, cells construct it step by step on helper proteins and then deliver it into the working site of nitrogenase. Understanding how this assembly line works is crucial not only for basic biology but also for long-term goals such as designing crops or industrial systems that fix nitrogen more efficiently and with less energy than current fertilizer production.
A Molecular Relay for a Fragile Cargo
The study focuses on a key late step in this assembly line. At this stage, another enzyme, NifB, has already forged a large iron–sulfur cluster called NifB-co. This fragile cluster must be moved to a scaffold protein pair, NifEN, where it will be converted into the final FeMo-cofactor. A small carrier protein, NifX, acts like a courier that picks up NifB-co from its birthplace and brings it to NifEN. Using high-resolution cryo–electron microscopy, the authors captured NifEN together with NifX and NifB-co, effectively freezing the handoff in mid-motion. They found that NifX is anchored to NifEN by its tail but keeps its main body flexible, allowing it to swing its cargo into position.
Seeing the Handoff in Atomic Detail
The images reveal that NifB-co first docks on the outer surface of NifEN, where two sulfur-containing amino acids at the beginning of the NifE subunit grip the cluster at opposite ends. In a special “transfer” state, one end of the cluster is still held by a histidine on NifX while the other is captured by NifE. This creates a brief moment when the cargo is literally shared between courier and scaffold, ensuring a safe, direct pass that minimizes exposure to the environment. Biochemical tests using mutant strains of the nitrogen-fixing bacterium Azotobacter vinelandii showed that when this N-terminal gripping loop on NifE is removed, the cells struggle to grow on nitrogen gas alone, especially when NifB-co is scarce, underscoring how vital this docking site is for efficient cofactor production.
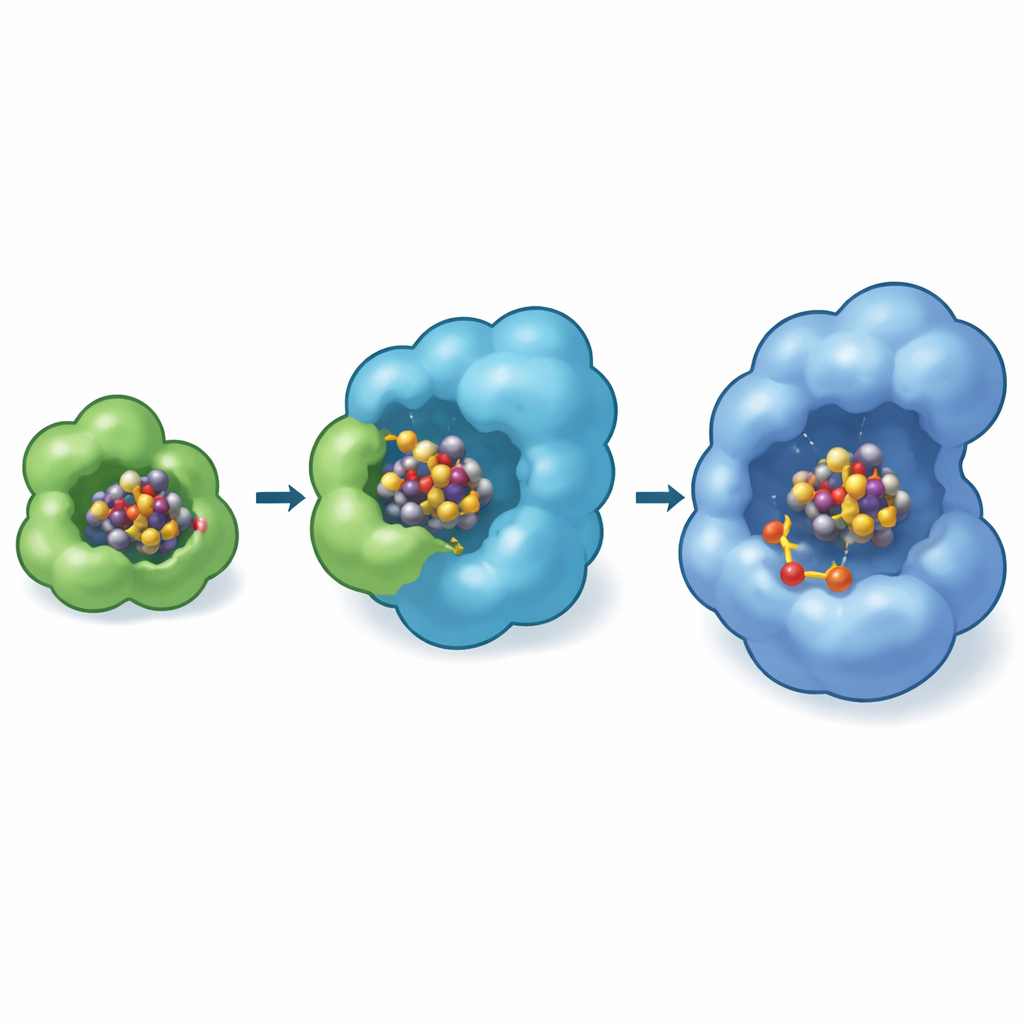
A Hidden Workshop Inside the Scaffold
Curiously, the experimentally observed docking site on NifEN sits near the protein surface, while in the finished nitrogenase enzyme the mature FeMo-cofactor sits deep inside a cavity. To bridge this gap, the authors used advanced computer models (Boltz-2) to predict where the cluster might move next. These simulations repeatedly placed NifB-co in an internal pocket of NifEN near a specific cysteine residue that is known to be essential for cofactor formation. The models further suggested that this inner pocket is where molybdenum and homocitrate are added to the cluster, turning NifB-co into the complete FeMo-cofactor. In the same modeling framework, NifX binds its cargo in a positively charged pocket that matches the experimental structure, reinforcing the idea of a defined route: NifX → surface receiving site on NifE → internal conversion chamber.
Why This Invisible Dance Matters
Taken together, the structural snapshots and simulations outline a choreographed sequence for building the nitrogenase heart: a costly metal cluster is made, handed from carrier to scaffold, moved into an inner chamber, and only then upgraded with molybdenum and homocitrate before being passed on to the final enzyme. For a general reader, the key message is that life invests great care in managing precious chemical parts, using a chain of specialized proteins to protect and refine them. By clarifying how this molecular relay works, the study adds an important piece to our understanding of biological nitrogen fixation, a process that underpins soil fertility and food production worldwide and may one day inspire cleaner, more sustainable ways to make fertilizer.
Citation: Schneider, F.F., Martin del Campo, J.S., Zhang, L. et al. Trafficking of a nitrogenase FeMo-cofactor assembly intermediate. Nat Chem Biol 22, 822–828 (2026). https://doi.org/10.1038/s41589-026-02179-0
Keywords: nitrogen fixation, nitrogenase, metal cofactors, cryo-electron microscopy, enzyme assembly