Clear Sky Science · en
CSN5i-3 is an orthosteric molecular glue inhibitor of COP9 signalosome
Why a New Kind of Drug Matters
Many modern medicines work by turning off overactive enzymes, but they often hit too many targets and cause side effects. This study uncovers a surprising way for a small molecule to shut down a key cellular control machine, the COP9 signalosome, much more precisely. By acting both as a conventional blocker and as a “molecular glue,” the compound CSN5i-3 points to a new strategy for designing safer, more selective drugs.
The Cell’s Recycling Switchboard
Inside our cells, a huge network of machines tags unwanted or damaged proteins so they can be destroyed. At the center of this system are cullin–RING ligases, scaffolds that bring together the tools needed to mark proteins for disposal. Their activity is turned on and off by attaching and removing a small protein called NEDD8. The COP9 signalosome (CSN) acts as the off-switch, trimming NEDD8 off cullins so the system can reset. Because this switchboard touches hundreds of cellular processes, chemists have long wanted to control CSN with drugs, but doing so safely is challenging.
A Small Molecule with a Big Twist
The compound CSN5i-3 was designed to plug the working site of CSN’s cutting subunit, CSN5, like a cork in a bottle. In simple tests, it binds this site only moderately well, in the micromolar range, which would not normally make a potent drug. Yet in cellular reactions CSN5i-3 shuts down CSN at nanomolar levels—around a thousand times stronger than its binding strength would suggest. Earlier work also hinted that it behaves like an “uncompetitive” inhibitor, preferring to bind the enzyme only when the substrate is already in place, a pattern rarely seen for direct active-site blockers. These oddities suggested that something more subtle was going on.
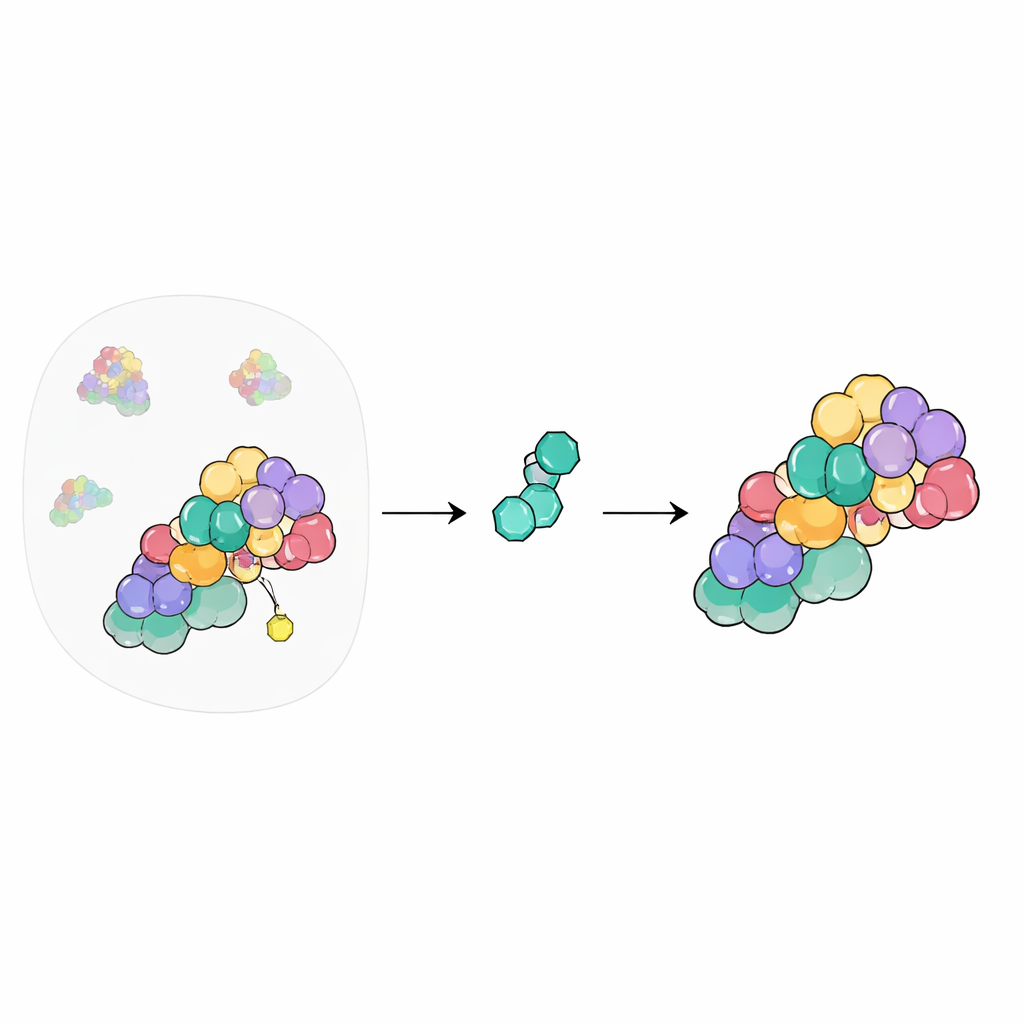
Seeing the Molecular Embrace
To understand this, the authors used high-resolution cryo–electron microscopy to visualize CSN in several states: alone, with its NEDD8‑modified cullin partners, and with CSN5i-3 present or absent. They first captured a “pre‑cut” state in which the tail of NEDD8 and the cullin attachment point are snugly positioned in the groove of CSN5, held in place by a chain of protein–protein contacts that extends from NEDD8 through the cullin and a helper protein called RBX to the rest of CSN. This structure explains how CSN recognizes its targets across many different cullins while maintaining the right geometry to break the NEDD8–cullin link.
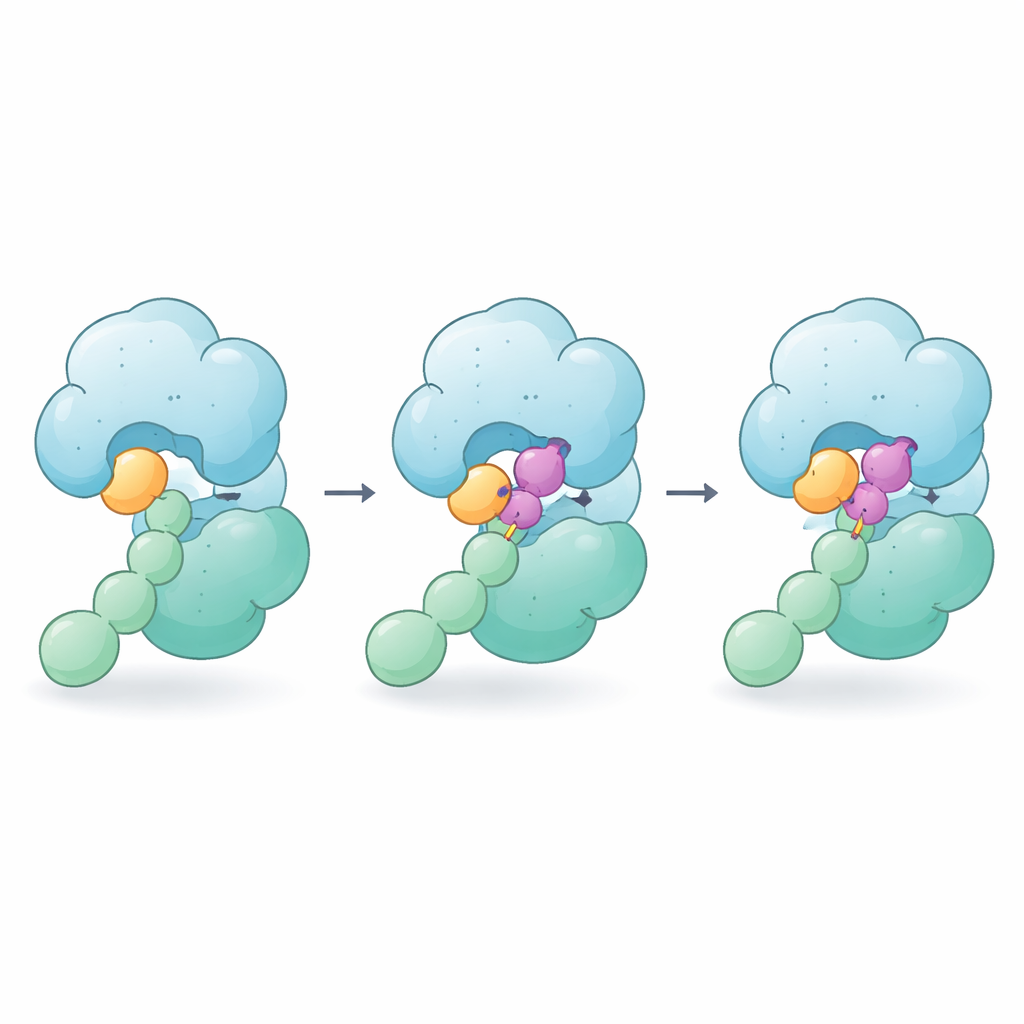
How the Glue Holds Without Cutting
When CSN5i-3 is added, it wedges into the same groove where the NEDD8–cullin bond would sit, so it still works as a classic blocker. But the pictures reveal that the molecule does more: one end nestles against the tail of NEDD8 while the other presses on the nearby region of the cullin. In effect, CSN5i-3 becomes a bridge that strengthens the weak contacts between NEDD8 and CSN5. Biophysical measurements show that, in the presence of the compound, free NEDD8 suddenly binds tightly to CSN, and the whole enzyme–substrate complex becomes much more stable. The authors call this behavior “orthosteric molecular glue” because the drug sits in the normal active site (orthosteric) yet also glues together enzyme and substrate.
Ripple Effects Across the Cell
By locking CSN onto its NEDD8‑decorated partners, CSN5i-3 changes how long these complexes linger in cells. Using cross-linking and mass spectrometry, the team mapped which proteins stay associated with CSN before and after treatment. They found that cullins and many of their adaptors are more often found bound to CSN when the compound is present, consistent with reaction “traffic jams” caused by the glue effect. Strikingly, some substrate receptors become less stable and are degraded, while others are trapped together with CSN. This diversity of outcomes suggests that subtle differences in how individual receptors touch CSN can determine whether they are protected or eliminated when an orthosteric molecular glue is applied.
What This Means for Future Medicines
The key message for non-specialists is that CSN5i-3 shows how a drug can be both a plug and a glue at the same time. Instead of needing to bind its target extremely tightly on its own, the molecule borrows strength from the very substrate it helps trap, achieving high real-world potency from modest basic affinity. Because the glue contacts can be tuned to recognize particular substrates, this approach could, in principle, be used to design medicines that block harmful enzyme actions while sparing others, reducing side effects. The authors propose “orthosteric molecular glue inhibitors” as a new class of compounds that could be applied to many enzymes involved in cutting or modifying proteins, opening a path toward more selective control of the cell’s busiest machines.
Citation: Shi, H., Wang, X., Yu, C. et al. CSN5i-3 is an orthosteric molecular glue inhibitor of COP9 signalosome. Nature 652, 1375–1383 (2026). https://doi.org/10.1038/s41586-026-10129-y
Keywords: molecular glue, COP9 signalosome, protein degradation, enzyme inhibitors, drug discovery